The diagnosis of vulvodynia is made after taking a careful history, ruling out infectious or dermatologic abnormalities, and eliciting pain in response to light pressure on the labia, introitus, or hymenal remnants. Several treatment options have been used, although the evidence for many of these treatments is incomplete. Treatments include oral medications that decrease nerve hypersensitivity (e.g., tricyclic antidepressants, selective serotonin reuptake inhibitors, anticonvulsants), pelvic floor biofeedback, cognitive behavioral therapy, local treatments, and (rarely) surgery. Most women experience substantial improvement when one or more treatments are used.
Vulvodynia is characterized by chronic discomfort in the vulvar region; the discomfort may range from mild to severe and debilitating. The diagnosis depends on a consistent history, lack of a documented infectious or dermatologic cause, and in most women, tenderness when gentle pressure is applied by a cotton swab to the vulva, introitus, or hymenal areas. The pain usually is present during and after intercourse, and other factors may exacerbate the pain (e.g., bicycle riding, tampon insertion, prolonged sitting, wearing tight clothes) (Table 1).1 In some women the pain is spontaneous.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Vulvodynia should be suspected in any female with a history of more than three months of pain at the introitus or vulva. | B | 6,7 |
| Tricyclic antidepressants should be considered for the treatment of vulvodynia. | B | 9,39,42,47 |
| Selective serotonin reuptake inhibitors and gabapentin (Neurontin) should be considered for symptomatic relief of vulvodynia. | B | 9,63 |
| Cognitive behavioral therapy should be used to decrease vulvar pain with intercourse. | B | 47 |
| Biofeedback and physical therapy should be considered to help patients regain control of the pelvic floor musculature. | B | 14,47,51 |
| Perineoplasty should be reserved for women with severe vulvodynia. | B | 47 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 1135 orhttps://www.aafp.org/afpsort.xml.
TABLE 1 Factors Affecting Pain in Women with Vulvodynia
| Factors that exacerbate pain (%) | Factors that relieve pain (%) |
|---|---|
| Intercourse (91) | Loose clothing (38) |
| Tight clothes (57) | Not wearing underwear (32) |
| Partner touch (56) | Applying ice to the area (25) |
| Riding a bicycle (42) | Being distracted (19) |
| Use of tampons (40) | Lying down (19) |
| Prolonged sitting (28) |
Information from reference 1.
Although vulvodynia was described in 1889 as “excessive sensitivity” of the vulva,2 it rarely was referred to in the medical literature until the 1980s. Recognition of this disorder and its effects on the lives of women worldwide led to the adoption of the term “vulvodynia” by the International Society for the Study of Vulvovaginal Diseases (ISSVD) in 1983. At the time, it was defined as “chronic vulvar discomfort that is characterized by the complaint of burning, stinging, irritation, or rawness” in the absence of skin disease or infection.3 The ISSVD recently revised the definition to include two subgroups: localized and generalized vulvar dysesthesia.4 Each of these subgroups is further categorized as provoked, spontaneous, or mixed. It is unclear whether these groups are separate disorders or different presentations of the same disorder.1 The term “vulvar vestibulitis” is no longer used because inflammation is not a prominent component of the disorder; it is now referred to as localized vulvar dysesthesia (or vestibulodynia).4
Prevalence
Three studies5–7 that systematically addressed prevalence in different settings found vulvar pain to be much more common than previously thought, with rates of 15 percent in one gynecologist’s practice,5 1.7 percent in an Internet survey,6 and 8.6 percent in a population-based study of symptomatic women in the Boston area.7 These findings would extrapolate to more than 2.4 million women in the United States and approximately 15 affected women in a family practice of 2,000 patients.
Characteristics of Women with Vulvodynia
Women presenting with vulvodynia typically are white; are in stable, long-term relationships; have had the pain for several years; and have been examined several times by multiple physicians before receiving the diagnosis.6–9 The age range is broad, from children (rarely) to women 80 years and older,6 but most women with this disorder are between 20 and 50 years of age.
Vulvodynia is not associated with sexually transmitted diseases (STDs) or STD risk factors,8,10 but affected women often have been treated repeatedly for candidal vulvovaginitis.8,10,11 In the past, it was theorized that the pain of vulvodynia was due to psychological issues.12,13 However, recent data indicate that women with vulvodynia are psychologically comparable to women without the disorder14–16 and are no more likely to have been abused.8,14,17 Marital satisfaction levels also are similar.14
Although women with vulvodynia report that the quality and quantity of their sexual activity has decreased since the onset of symptoms, more than one half have had intercourse and have had an orgasm in the previous month.18 These women were just as likely as women without pain to participate in other sexual activities (e.g., masturbation, receiving oral sex).18
Pathophysiology
Although research is ongoing, little is known about the causes of vulvodynia. Affected women are more likely to have altered contractile characteristics of the pelvic floor musculature19; biofeedback therapy designed to address these alterations often results in improved muscle function and decreased vulvar pain.20,21 Although women with vulvodynia were known to be sensitive to touch in the vestibular region, it has only recently become clear that women with vulvodynia also have increased sensitivity at peripheral sites, such as the upper arm or leg.22,23 Whether these muscular changes and increased systemic sensitivity are primary or secondary to the pain disorder is unknown.
Several studies have identified minor immunologic changes in women with vulvodynia, such as altered levels of interleukin-1 and tumor necrosis factor-α in vestibular tissue24; increased production of interleukin-1β and decreased production of interleukin-1 receptor antagonist by lymphocytes following stimulation25; decreased production of interferon-α26; and changes in the gene associated with interleukin-1 receptor antagonist.27,28 These changes could result in a decreased ability to downregulate the inflammatory response, which in turn may be associated with neuropathic changes.
There is controversy about whether changes exist in the inflammatory infiltrate in vulvar tissue of women with vulvodynia. Some studies29–31 found an increase in inflammatory cells or mast cells, whereas others32,33 found that inflammatory cell infiltrates were similar in patients with vulvodynia and control patients. More recently, vulvar biopsy specimens have shown increased neuronal proliferation and branching in the vulvar tissue of women with vulvodynia compared with tissue of asymptomatic women.34–36 The cause of this increased neuronal density and its role in vulvodynia remain unclear.
Clinical Presentation
Symptoms of vulvodynia may have been present since childhood or the time of first intercourse, or they may appear after several years of painless sex.10,18 The pain often is described as “burning,” but it may be irritating, sharp, prickly, or, occasionally, pruritic, and it can be mild to severe.23 Factors that exacerbate the symptoms and those that lessen the pain are listed in Table 1.1 The pain can begin suddenly when provoked, and it tends to dissipate gradually; women with vulvodynia often report hours to days of discomfort after intercourse or a pelvic examination.
Allodynia (i.e., pain elicited by a nonpainful stimulus) and hyperpathia (i.e., when a stimulus causes greater pain than expected) suggest a neuropathic cause of the pain of vulvodynia.36 This characterization has led to improved understanding of why certain therapies often are not useful (e.g., corticosteroids, estrogen therapy), and why others that have been effective in the treatment of other neuropathic disorders may be of use in patients with vulvodynia (e.g., amitriptyline, gabapentin [Neurontin]).
Diagnosis
The diagnosis of vulvodynia depends on a careful history, followed by a confirmatory physical examination (Figure 1).37 The history should include information about the onset and character of the pain, provoking and relieving factors, medical evaluations to date, and attempted treatments and their effects on the pain. Occasionally, the patient may not be aware that the sensitivity is in the area of the introitus, and she may describe the pain as deeper in the vagina or pelvis. Confirmation during the examination often will clarify this issue.
Figure 1. Diagnosis of Vulvar Pain and Dyspareunia
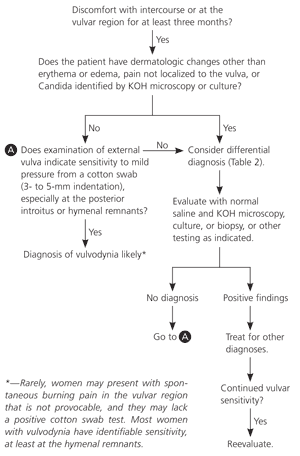
Algorithm for the diagnosis of vulvar pain and dyspareunia. (KOH = potassium hydroxide.)
Adapted with permission from Reed BD. Vulvodynia. Female Patient 2005;30:48–54.
The physical examination is an important part of the diagnostic process. The vulva may be erythematous in women with vulvodynia, but the presence of a rash or altered mucosa or skin is not consistent with vulvodynia and requires further evaluation or biopsy. A cotton swab is used to gently indent (approximately 5 mm) several locations on the labia, introitus, and hymenal remnants. This pressure will elicit discomfort in almost all women with vulvodynia; the posterior introitus and the posterior hymenal remnants are the most common sites of increased sensitivity. Although some women have spontaneous pain that may not be provoked with a cotton swab, a lack of sensitivity in all of these areas is unusual among women with provocable pain. Other diagnoses that may be suggested by the examination and history are listed in Table 2.
TABLE 2 Differential Diagnosis of Dyspareunia at the Vaginal Introitus
| Diagnosis | Clinical findings |
|---|---|
| Allergic vulvitis | Pruritus, irritation, burning |
| History consistent with allergen exposure; lack of infectious cause | |
| Chronic candidal vulvovaginitis | Variable erythema, edema, or thick white discharge; pruritus is common |
| Potassium hydroxide microscopy or culture positive | |
| Lichen planus | White reticulate lesions, vaginal discharge, pruritus, burning, dyspareunia, and bleeding with intercourse; may have erosions, erythema, buccal lesions, or papulosquamous plaques |
| Lichen sclerosis | Vulvar thinning, whitening, and wrinkling; agglutination of labia. Pruritus may be severe. |
| Pudendal canal syndrome | Unilateral genital pain, often increased with sitting |
| Vaginismus | Pelvic floor muscle spasm present and accentuated with examination |
| Vulvar atrophy | Pale, thinning mucosa; possible tears or petechiae. White blood cells and parabasal cells present in vaginal discharge |
| Vulvar intraepithelial neoplasia | White or multicolored, elevated lesions, possibly warty. Patient may be asymptomatic or have pruritus. |
Vaginal secretions should be evaluated for the presence of an active candidal vulvovaginal infection. If an infection is diagnosed, treatment with antifungal medication should precede treatment for vulvodynia. Treatment for a possible yeast infection without confirmation by strongly positive potassium hydroxide microscopy (i.e., budding spores or hyphae) is not recommended, and a yeast culture should be performed if there is uncertainty about infection. Although women with vulvodynia may carry Candida, eradication of Candida usually will not alleviate the patient’s symptoms.
Treatment
Data are lacking about the effectiveness of various treatment options for vulvodynia, and many of the commonly recommended treatments have not been systematically studied in randomized controlled trials (RCTs). Nevertheless, several treatments have been used with varying degrees of success (Table 39,21,38–53),54 and the responses to these treatments suggest that improvement should be expected in most women with vulvodynia.
TABLE 3 Treatment Options for Women with Vulvodynia
| Therapy | Proposed mechanism | Beginning dosage | Continuing dosage | Side effects | Precautions | Evidence |
|---|---|---|---|---|---|---|
| Oral therapies | ||||||
| Amitriptyline | Decreases neuronal hypersensitivity | 25 mg at bedtime for 10 days, then 50 mg at bedtime daily | 50 to 100 mg at bedtime (higher dosages to 225 mg occasionally are successful) | Dry mouth, fatigue (often transient), constipation, weight gain (uncommon) | Start at lower dosages in older patients. | Case reports9,38,39 |
| Retrospective reports9,39 | ||||||
| Calcium citrate | Decreases oxalate deposition in tissues | 2 tablets twice daily | 2 to 4 tablets twice daily | Minimal | Used in conjunction with other treatments. | Case reports40 |
| Anecdotal evidence when used in conjunction with a low-oxalate diet41 | ||||||
| Desipramine (Norpramin) | Decreases neuronal hypersensitivity | 25 mg at bedtime for 10 days, then 50 mg at bedtime daily | 50 to 100 mg at bedtime (higher dosages to 225 mg occasionally are successful) | Same as amitriptyline but less common | Start at lower dosages in older patients. | None. Based on similarity to amitriptyline and use in other painful neuropathies42 |
| Gabapentin (Neurontin) | Decreases neuronal hypersensitivity | 300 mg daily, increasing every five days by 300 mg per day (to three-times-per-day dosing) | 300 mg three times daily, to maximum of 900 mg three times daily (2,700 mg) | Headaches, nausea, vomiting, fatigue, and dizziness (often transient or mild)43 | — | Case reports suggest benefit.43 |
| Paroxetine (Paxil) | Decreases neuronal hypersensitivity | 10 mg daily | 20 to 60 mg daily | Rarely fatigue, anorgasmia, or weight gain | Discontinuation should be gradual. | Case reports9 |
| Venlafaxine (Effexor) | Decreases neuronal hypersensitivity | 37.5 mg daily | 75 to 150 mg daily | Anorgasmia, gastrointestinal side effects, anxiety | Blood pressure, electrolyte levels, and lipid levels should be monitored periodically. | Used in other painful neuropathies |
| Dietary changes | ||||||
| Low-oxalate diet | Decreases the possible role of oxalate deposition in vulvar tissue | Ranges from a highly oxalate-restrictive diet to avoidance of a short list of foods that increase oxalate levels significantly44 | — | A low-oxalate diet is very restrictive. If initiated, it should be continued only if clear benefits are noted. | — | The role of oxalates in vulvodynia is controversial.40,41 A case report40 suggested benefit when used in conjunction with calcium citrate supplementation. |
| Surgical therapy | ||||||
| Perineoplasty and vestibulectomy | Removal of hypersensitive tissue and replacement with vaginal mucosa advancement | — | — | Acute discomfort and job absenteeism. Rarely, bleeding, infection, hematoma, wound separation, vaginismus, vaginal stenosis | Only useful in localized (vestibular) cases. Most physicians reserve surgical treatment for patients who have not had success with other therapies. | Case series45,46 |
| Contolled treatment trial47 (70 percent response) | ||||||
| Topical therapies | ||||||
| Lidocaine gel or cream (5%) | Local anesthetic to decrease hypersensitivity | Apply topically to introitus. | Apply as needed before intercourse or nightly on a cotton ball in the introitus. | — | — | Case series48 |
| Cromolyn cream (4%) | Decreases possible mast cell degranulation in vulvar tissue | Apply three times daily to introitus. | Continue three-times-daily application to introitus. | — | Not commercially available; must be formulated in a bland cream or ointment. | Case report suggested benefit.49 |
| Blinded treatment trial49 suggested no improvement. | ||||||
| Avoidance of irritants (e.g., perfumes, harsh soaps, colored underwear, nylon) | Decreases exposure to foreign antigens and heat | — | — | — | — | Unknown efficacy50 |
| Other therapies | ||||||
| Biofeedback and/or physical therapy | Improves the electrodiagnostic pattern of pelvic floor musculature via biofeedback and muscle conditioning | Evaluation by physical therapist and instruction in home biofeedback exercises, with or without other physical therapy modalities and portable electromyographic feedback instrumentation | Continue home biofeedback daily for 16 weeks.47,51 Six to 16 physical therapy sessions, if used | Discomfort during treatment | Biofeedback requires home electromyographic monitor. Physical therapy typically uses instrumentation during therapy and exercises and stretching at home. | Nonblinded case series21,51 of biofeedback with six-month follow-up indicated benefit. |
| Controlled trial47 of biofeedback indicated 30 percent improvement. | ||||||
| Retrospective case series52 of physical therapy suggested improvement in most patients. | ||||||
| Cognitive behavioral therapy | Increases understanding of the disorder and encourages patients to find ways to minimize symptoms | Eight two-hour group sessions over 12 weeks | — | — | Recommended duration can be up to 1.5 years.53 | Randomized treatment trial47 (30 percent response) |
note:None of the medications listed have been approved by the U.S. Food and Drug Administration for the treatment of vulvodynia.
SYSTEMIC THERAPY
Because the pain of vulvodynia seems to be neuropathic, many medications that have been used effectively in the treatment of other neuropathic disorders have been used in patients with vulvodynia. Tricyclic antidepressants (TCAs) often are used as a first-line therapy. Many patients tolerate TCAs well after approximately one week of use, but prolonged fatigue, constipation, and weight gain may require a change in the type of medication or dosage. Dry mouth is common when taking TCAs but rarely necessitates discontinuation. Amitriptyline has been used primarily, but other TCAs with less severe side effects, such as desipramine (Norpramin), may be effective as well. Although selective serotonin reuptake inhibitors (SSRIs) are not commonly considered effective for treatment of neuropathic pain, many patients who cannot tolerate TCAs have responded well to SSRIs. Venlafaxine (Effexor) is being used more commonly for the treatment of vulvodynia. Gabapentin may be helpful for various types of neuropathic pain. However, current recommendations for three-times-per-day dosage limit the ease with which this drug can be used.
There are few data on which to base recommendations for the use of calcium citrate, and this therapy has been studied only in conjunction with a low-oxalate diet. However, some patients find it helpful, and side effects are minimal. Prolonged treatment with oral fluconazole (Diflucan) has been recommended based on the supposition that chronic candidal infections may contribute to vulvodynia, but results have been inconsistent.38,55 This therapy should be reserved for patients with documented candidal infection. Narcotics and analgesics, such as nonsteroidal anti-inflammatory drugs and acetaminophen, have not been helpful.
PSYCHOLOGICAL APPROACHES
One RCT47 has shown that cognitive behavioral therapy is associated with a 30 percent decrease in reported vulvar pain with intercourse. Although psychological predispositions have not been proven to be associated with vulvodynia, the pain may be associated with interpersonal or individual psychological difficulties. Sexual, individual, and marital counseling also should be considered in patients with ongoing difficulties in these areas.
LOCAL THERAPY
Biofeedback and physical therapy have been used to reverse changes in the pelvic floor musculature and to help women regain control of the muscles, including improving strength and relaxation. Studies21,47,51 suggest that these therapies can improve symptoms of vulvodynia.
Although the value of most local treatment measures has not been determined, women with vulvodynia often are counseled to avoid the use of harsh soaps and perfumed products in the vulvar region50 and to wear all-cotton underwear.56 Some experts think that dermatologic diagnoses (e.g., atopy, dermatographism, irritant contact dermatitis) and candidiasis are responsible for many of the symptoms of vulvodynia, and they recommend treatment with antihistamines or antifungal medications, as well as avoidance of potentially irritating contactants.57
Topical corticosteroids11 and estrogens45 generally have not been successful in decreasing the pain of vulvodynia, and these treatments are used primarily in patients with specific indications (e.g., estrogen deficiency, lichen sclerosus). Similarly, the injection of steroids58 and interferon46,59,60 in women with localized symptoms has met with conflicting results and recommendations. Topical lidocaine (Xylocaine) has been used as needed (up to three or four vulvar applications per day) and recently has been used on a nightly basis in the introitus to minimize vulvar pain, with encouraging results.48 Topical cromolyn sodium appeared useful in a case series, but an RCT suggested that results were similar to those in the placebo group.49
SURGERY
Surgery is one of the oldest therapies for localized vulvar vestibulodynia, but this therapy typically is reserved for women with severe, debilitating symptoms that are localized to the introitus. Several studies47,61 have found subjective improvement in pain in 60 to 80 percent of women who underwent surgery. Until recently, most of the studies were case series, with the inherent limitations of selection bias, reporting bias, and poor controlling for confounders.46,62 A three-armed treatment trial47 comparing surgery with biofeedback and cognitive behavioral therapy found that women who underwent perineoplasty had greater reductions in pain than those who were treated with biofeedback or cognitive behavioral therapy. Nevertheless, most experts do not recommend surgery for women with this disorder unless symptoms are severe and other treatments have not been effective. In light of recent evidence6,7 suggesting that vulvodynia may be transient in a substantial percentage of women, reserving surgical approaches when possible seems prudent.
Carbon dioxide laser surgery for the vulva and vestibule was used for several years, but this procedure is no longer recommended because of the scarring and worsening of symptoms that can follow. The use of other dye laser protocols is controversial.50 Other therapies have been tried, with inconsistent results. Isolated case reports suggest that hypnosis,63 inosine pranobex (Isoprinosine; not available in the United States),64 and acupuncture65,66 may be effective treatments for vulvodynia, but further study is warranted.
A large proportion of women with chronic vaginal symptoms (some of whom have vulvodynia) are using or have used complementary therapies.67 Few data are available on which to base recommendations for or against these treatments. Physicians should ask patients about other therapies they are using or considering.
WHEN TO REFER
Family physicians can diagnose and treat women with vulvodynia, and substantial improvement should be expected. Referral to a subspecialist should be considered for any patient who is unresponsive to treatment.
Prognosis
The natural history of vulvodynia is not clear. Many women have had this disorder for years, and it traditionally has been considered chronic. However, recent data6,7 suggest that approximately one half of women who report that they have had prolonged vulvar pain no longer have symptoms of vulvodynia. Hence, it is reasonable to think that symptoms in a substantial proportion of women with this disorder may abate. The prognosis for women who receive appropriate treatment is quite good. A follow-up study9 of 104 women seen in two referral clinics at the University of Michigan suggests that most women with vulvodynia experience substantial improvement with medical treatment.

