Parkinson’s disease is a common neurodegenerative disorder that can cause significant disability and decreased quality of life. The cardinal physical signs of the disease are distal resting tremor, rigidity, bradykinesia, and asymmetric onset. Levodopa is the primary treatment for Parkinson’s disease; however, its long-term use is limited by motor complications and drug-induced dyskinesia. Dopamine agonists are options for initial treatment and have been shown to delay the onset of motor complications. However, dopamine agonists are inferior to levodopa in controlling motor symptoms. After levodopa-related motor complications develop in advanced Parkinson’s disease, it is beneficial to initiate adjuvant therapy with dopamine agonists, catecholO-methyltransferase inhibitors, or monoamine oxidase-B inhibitors. Deep brain stimulation of the subthalamic nucleus has been shown to ameliorate symptoms in patients with advanced disease. Depression, dementia, and psychosis are common psychiatric problems associated with Parkinson’s disease. Psychosis is usually drug induced and can be managed initially by reducing antiparkinsonian medications. The judicious use of psychoactive agents may be necessary. Consultation with a subspecialist is often required.
Parkinson’s disease is a progressive neurodegenerative disorder with an estimated prevalence of 0.3 percent in the U.S. population.1 The prevalence increases to 4 to 5 percent in those older than 85 years.1 Characteristic neuropathologic features of the disease are dopaminergic neuron degeneration in the substantia nigra and the presence of eosinophilic intracytoplasmic inclusions (Lewy bodies) in the residual dopaminergic neurons.2 Family physicians should have a good understanding of Parkinson’s disease because of its increasing prevalence as the population ages. Treatment should be individualized to decrease symptoms while minimizing motor and nonmotor complications (Figure 1).3,4 As the disease progresses, treatment can be increasingly complicated, and comanagement with a subspecialist may be required. The most important goal is to help patients maintain maximal autonomy and quality of life.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Levodopa is the most effective pharmacologic treatment for Parkinson’s disease symptoms, especially bradykinesia and rigidity. | A | 14,15 |
| Dopamine agonists effectively treat early Parkinson’s disease. | A | 15,17–19,21,24,25 |
| Motor complications in patients with advanced Parkinson’s disease can be treated by adding a dopamine agonist, monoamine oxidase-B inhibitor, or catecholO-methyltransferase inhibitor to levodopa therapy. | A | 15,24–27 |
| Deep brain stimulation of the subthalamic nucleus can improve Parkinson’s disease symptoms. | A | 15 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 2008 orhttps://www.aafp.org/afpsort.xml.
Figure 1. Diagnosis and Management of Parkinson's Disease
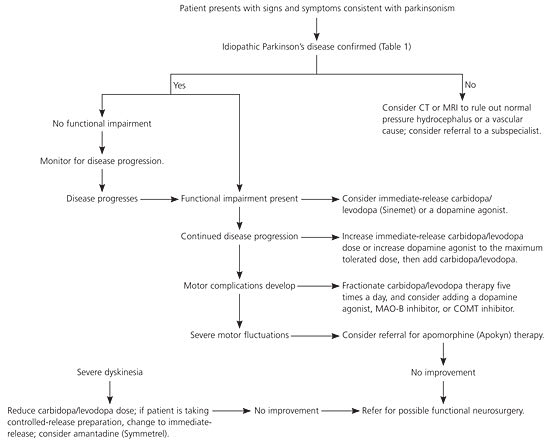
Algorithm for the diagnosis and management of Parkinson’s disease. (CT = computed tomography; MRI = magnetic resonance imaging; MAO-B = monoamine oxidase-B; COMT = catechol O-methyltransferase.)
Information from references 3 and 4.
Diagnosis
Despite advances in radiologic testing, the diagnosis of idiopathic Parkinson’s disease remains a clinical one. A diagnosis requires the presence of the following cardinal signs: distal resting tremor of 3 to 6 Hz, rigidity, bradykinesia, and asymmetrical onset.5 Other well-known signs of Parkinson’s disease include late-onset postural instability, decreased olfaction, and micrographia. Patients must also respond to an adequate therapeutic challenge of levodopa or a dopamine agonist.5 Symptoms and signs associated with Parkinson’s disease are summarized in Table 1.6
TABLE 1 Signs and Symptoms of Parkinson’s Disease
| Sign/symptom | LR+ | LR- | Description |
|---|---|---|---|
| History of tremor | 1.3 to 17 | 0.24 to 0.60 | Observed as patient rests hands in his or her lap; often described as pill-rolling in quality; must be distinguished from postural tremor (as limb is held against gravity) or kinetic tremor (occurs with movements) |
| Distal resting tremor as a sign | 1.3 to 1.5 | 0.47 to 0.61 | |
| History of bradykinesia and rigidity | 4.5 | 0.12 | Difficulty with rapidly and sequentially tapping the fingers of one hand and then the other on a table top; difficulty tapping the heel rapidly; difficulty twiddling or circling the hands rapidly around each other in front of the body; reduced arm swing on affected side during ambulation |
| Difficulty turning over in bed | 13 | 0.56 | |
| Difficulty opening jars | 6.1 | 0.26 | |
| Difficulty rising from a chair | 1.9 to 5.2 | 0.39 to 0.58 | |
| Rigidity as a sign | 0.53 to 2.8 | 0.38 to 1.6 | The physician feels resistance as he or she places a finger within the patient’s antecubital fossa and repeatedly flexes and extends the patient’s arm at the elbow; resistance can be cogwheel rigidity (catching and releasing) or lead-pipe rigidity (continuously rigid); rigidity must be distinguished from spasticity, which has only increased flexor tone; rigidity also can be tested at wrist supination or pronation |
| Poor heel-to-toe gait | 2.9 | 0.32 | Small, shuffling steps may be observed, with difficulty initiating ambulation; patients may have a festinating gait (involuntary acceleration of gait); heel-to-toe ambulation is impaired; arms often are stationary; posture often is stooped; patients may have difficulty turning and have poor balance |
| History of shuffling gait | 3.3 to 15 | 0.32 to 0.50 | |
| Loss of balance | 1.6 to 6.6 | 0.29 to 0.35 | |
| History of micrographia | 2.8 to 5.9 | 0.30 to 0.44 | Handwriting is small and often indecipherable |
LR+ = positive likelihood ratio; NPV = negative likelihood ratio.
Adapted with permission from Rao G, Fisch L, Srinivasan S, D’Amico F, Okada T, Eaton C, et al. Does this patient have Parkinson’s disease? JAMA 2003;289:251.
The clinical presentation of Parkinson’s disease is similar to that of diverse neurologic disorders called parkinsonisms (Table 2).5–8 Symptoms that suggest a diagnosis other than Parkinson’s disease include lack of response to levodopa, hallucinations, prominent and early dementia, early postural instability, severe and early autonomic dysfunction, upward gaze paralysis, and involuntary movements other than tremor.5
TABLE 2 Differential Diagnosis of Parkinsonism
| Diagnosis | Historical features | Signs/symptoms | Radiographic finding | Comments |
|---|---|---|---|---|
| Idiopathic Parkinson’s disease | Difficulty with tasks, rigidity, tremor | SeeTable 1 | No specific CT or MRI findings | — |
| Drug-induced parkinsonism | Previous use of a causative drug such as an antipsychotic, reserpine (Serpalan), or metoclopramide (Reglan) | Tremor, rigidity, bradykinesia; often bilateral symptoms | Normal | May persist for up to one year after discontinuation of the drug |
| Vascular parkinsonism | Stepwise progression; CVA or TIA, comorbid cardiovascular disease | Fixed deficits from previous events | Lesions in white matter with or without basal ganglia5 | Common because of the prevalence of cerebrovascular disease5 |
| Essential tremor | History in multiple family members, little evolution5 | Tremor often is action-based; absence of extrapyramidal symptoms (except possible mild rigidity); no response to levodopa5; tremor often is bilateral and can be attenuated by alcohol6 | SPECT shows normal dopaminergic system5 | Common5 |
| Normal-pressure hydrocephalus | Ataxia, dementia, urinary incontinence | Ataxic gait, change in mental status | CT or MRI shows hydrocephalus5 | Distinctive clinical features |
| Progressive supranuclear palsy | Onset after 40 years of age; frequent falls5 | Vertical gaze paralysis (or slowed vertical saccadic movements); marked postural instability (increased fall risk within the first year of disease)5; resting tremor (rare)6; nuchal dystonia; normal olfaction7 | MRI shows mesencephalon-brainstem atrophy involving the superior colliculi5 | — |
| Multiple system atrophy (e.g., Shy Drager syndrome, olivopontocerebellar atrophy, nigrostriatal degeneration) | Autonomic and urinary dysfunction, parkinsonism with poor levodopa response, cerebellar dysfunction5 | Resting tremor (rare), transient response to levodopa (25 percent of patients)6; possible mild impaired olfaction but less severe than Parkinson’s disease8 | MRI shows nigrostriatal degeneration with gliosis in the lateral putamen with hyperintense proton density bands and hypointense bands in the T2 images5 | Decreased sphincter EMG results; changes in urodynamic and sympathocutaneous testing; altered cardiovascular responses5 |
| Corticobasal degeneration | Cortical and cognitive impairment with involuntary movements5 | Apraxia, aphasia, sensory disorders, positive Babinski test, asymmetrical parkinsonism without impaired deambulation (difficulty in stopping gait), myoclonus, dystonia5 | MRI shows asymmetric atrophy of frontal and parietal regions, there may be atrophy of basal ganglia and/or corpus callosum; PET shows decreased glucose metabolism in frontoparietal cortex, thalamus, and basal ganglia | Commonly Underdiagnosed because of its heterogeneity5 |
| Dementia with Lewy bodies | Cognitive impairment, hallucinations, episodes of delirium, parkinsonism5 | Impaired attention and visuospatial abilities, increased falls, episodes of syncope5 | PET shows low glucose metabolism in cortex5 | Marked intolerance to neuroleptic drugs5 |
CT = computed tomography; MRI = magnetic resonance imaging; CVA = cerebrovascular accident; TIA = transient ischemic attack; SPECT= single-photon emission computed tomography; EMG = electromyography; PET = positron emission tomography.
Advances in radiologic technologies have aided in distinguishing the etiologies of parkinsonism and in more accurately diagnosing idiopathic Parkinson’s disease. Although head computed tomography (CT) and magnetic resonance imaging (MRI) show no specific Parkinson’s disease patterns, they can help rule out or confirm other diseases. Rapidly emerging technologies (e.g., positron emission tomography, single-photon emission CT) likely will impact Parkinson’s disease diagnoses; however, the evidence currently does not show that specific tests improve diagnostic accuracy.7 New data suggest that pronounced loss of olfaction can distinguish Parkinson’s disease from other parkinsonisms.9
Neuroprotection
Neuroprotection includes secondary prevention strategies aimed at slowing, blocking, or reversing disease progression. Preliminary studies suggest that coenzyme Q1010 and some dopamine agonists11,12 may slow disease progression. Despite these studies, no neuroprotective agents have been proven effective.
Symptom Management
Symptomatic therapy for Parkinson’s disease should be initiated at the onset of functional impairment. Several factors determine whether a patient has functional impairment. For instance, involvement of the dominant hand and bradykinesia tend to most greatly affect a patient’s ability to work and perform activities of daily living.3 The Unified Parkinson’s Disease Rating Scale (UPDRS) is a standardized assessment tool that facilitates accurate documentation of disease progression and treatment response.13 The four-part scale measures mental effects, limitations in activities of daily living, motor impairment, and treatment or disease complications. The UPDRS is available athttp://www.mdvu.org/pdf/updrs.pdf.
Early-Stage Treatment
Early-stage Parkinson’s disease includes patients who have had the disease for less than five years or those who have not developed motor complications from levodopa use.4 Treatment with monoamine oxidase-B (MAO-B) inhibitors, amantadine (Symmetrel), or anticholinergics may modestly improve mild symptoms; however, most patients need levodopa or a dopamine agonist. The American Academy of Neurology (AAN) recommends levodopa or a dopamine agonist, when dopaminergic treatment is required, depending on the need to improve motor disability (levodopa is better) or decrease motor complications (dopamine agonists cause fewer motor complications).14 Table 315,16 summarizes medications approved for Parkinson’s disease. In general, a dopamine agonist is initiated in patients with mild disease with onset at a younger age, whereas levodopa is initiated for older patients with severe motor symptoms.
TABLE 3 FDA-Approved Medications for Parkinson’s Disease
| Medication | Adverse effects | Indications and comments |
|---|---|---|
| Anticholinergics | ||
| Benztropine (Cogentin), trihexyphenidyl (Artane) | Dry mouth, dry eyes, constipation, hypotension, cognitive impairment, urinary retention | Useful for symptomatic control of Parkinson’s disease (benefits are mild to moderate); associated with more adverse effects than other drugs |
| Carbidopa/levodopa | ||
| Immediate- and sustained-release carbidopa/levodopa (Sinemet) | Nausea, somnolence, dyskinesia, hypotension, hallucinations | Levodopa is the most effective medication and remains the primary treatment for symptomatic Parkinson’s disease; no added benefit for motor complications with sustained-release versus immediate-release preparations |
| COMT inhibitors | ||
| Entacapone (Comtan) | Diarrhea; exacerbates levodopa adverse effects; bright orange urine | Useful for managing motor fluctuations (“wearing-off” effect) in patients taking levodopa; levodopa dose may need to be reduced if dyskinesia appears |
| Tolcapone (Tasmar) | Diarrhea; exacerbates levodopa adverse effects; rare liver failure (liver function monitoring needed) | |
| Dopamine agonists | ||
| Bromocriptine (Parlodel) | Nausea, headache, dizziness | Useful for early and advanced disease |
| Pergolide (Permax) | Somnolence; hallucinations; nausea; edema; fibrosis of cardiac valves, lung, and retroperitoneum; retroperitoneal and pulmonary fibrosis | Useful for the initial treatment of parkinsonism and as adjunct therapy in patients taking levodopa |
| Pramipexole (Mirapex), Ropinirole (Requip) | Nausea, sleep attacks, edema, hallucinations, hypotension | Useful for early disease and in patients with Parkinson’s disease and motor fluctuations |
| MAO-B inhibitors | ||
| Selegiline (Eldepryl) | Nausea, insomnia, drug interactions with other MAO inhibitors/tyramine | Useful for symptomatic control of Parkinson’s disease (benefits are mild to moderate) and as adjuvant therapy for patients with Parkinson’s disease and motor fluctuations |
| Rasagaline (Azilect) | Weight loss, hypotension, dry mouth, drug interactions with other MAO inhibitors/tyramine | |
| NMDA receptor inhibitor | ||
| Amantadine (Symmetrel) | Nausea, hypotension, hallucinations, confusion, edema | Useful for treating akinesia, rigidity, tremor, dyskinesia |
FDA = U.S. Food and Drug Administration; COMT = catechol O-methyltransferase, MAO-B = monoamine oxidase-B; NMDA = N-methyl-d-aspartate.
LEVODOPA
Levodopa is the most effective pharmacologic agent for Parkinson’s disease and remains the primary treatment for symptomatic patients.14,15 Because of its consistent and dramatic beneficial effects, levodopa has not been tested against placebo in therapeutic randomized controlled trials. Levodopa is particularly effective at controlling bradykinesia and rigidity14; however, speech, postural reflex, and gait disturbance are less likely to respond.
Levodopa is always combined with carbidopa, because carbidopa prevents peripheral conversion of levodopa to dopamine by blocking dopa decarboxylase. When combined with levodopa, carbidopa increases cerebral levodopa bioavailability and reduces the peripheral adverse effects of dopamine (e.g., nausea, hypotension). Because dopa decarboxylase is saturated by carbidopa at approximately 70 to 100 mg a day, patients receiving less than this amount of carbidopa are more likely to experience nausea and vomiting. Dosing should start with one 25/100-mg carbidopa/levodopa (Sinemet) tablet three times a day. Sustained-release preparations add no benefit for motor complications compared with immediate-release preparations.14
DOPAMINE AGONISTS
Dopamine agonists directly stimulate dopamine receptors and include bromocriptine (Parlodel), pergolide (Permax), pramipexole (Mirapex), and ropinirole (Requip). Studies have demonstrated that dopamine agonists, alone15,17–19 or combined with levodopa,7 are effective against early Parkinson’s disease. Double-blind controlled trials comparing ropinirole20 or pramipexole21 with levodopa showed that levodopa was more effective at reducing UPDRS scores than dopamine agonists; however, these studies also noted a lower incidence of motor complications with dopamine agonists.
MAO-B INHIBITORS
An evidence-based review showed that the MAO-B inhibitor selegiline (Eldepryl) had a mild symptomatic benefit in patients with early Parkinson’s disease.14 A meta-analysis of 17 trials comparing MAO-B inhibitors with placebo or levodopa in patients with early Parkinson’s disease showed that MAO-B inhibitors reduced disability, the incidence of motor fluctuations, and the need for levodopa without substantial adverse effects or increased mortality.22
OTHER AGENTS
Anticholinergic agents are commonly used to treat Parkinson’s disease. However, low effectiveness and a high incidence of gastrointestinal and neuropsychiatric adverse effects limit their use in older patients. Anticholinergics typically are used in patients younger than 70 years with disabling resting tremors and preserved cognitive function.
The N-methyl-d-aspartate receptor inhibitor amantadine was originally used as an antiviral agent and has been shown to improve akinesia, rigidity, and tremor in patients with Parkinson’s disease. The drug may be clinically useful, although rigorous studies are lacking.15
Late-Stage Treatment
Late-stage Parkinson’s disease includes patients already receiving carbidopa/levodopa treatment who have developed motor complications. After five years of treatment with levodopa, about 40 percent of patients develop motor fluctuations and dyskinesia (i.e., involuntary choreiform or stereotypic movements involving the head, trunk, limbs, and, occasionally, the respiratory muscles).23 Patients may experience a “wearing-off” effect characterized by a shorter duration of benefit from each levodopa dose, causing parkinsonian symptoms to reemerge. Patients can also experience an “on-off” effect characterized by unpredictable, abrupt fluctuations in motor state from when the medication is effective and symptoms are controlled (“on”) to when parkinsonian symptoms worsen (“off”). These motor complications can be treated by adding a dopamine agonist, MAO-B inhibitor, or catecholO-methyltransferase (COMT) inhibitor.15,24–27
DOPAMINE AGONISTS
Systematic reviews have demonstrated that dopamine agonists may significantly reduce “off” time, improving motor impairment and disability and reducing the need for levodopa.15,24,25 However, the reviews have also shown a trend toward increased adverse events (e.g., dizziness, hallucinations, dyskinesia) with dopamine agonists.
Parenteral apomorphine (Apokyn), a powerful dopamine agonist, is useful for patients experiencing a sudden, unexpected, and resistant “off ” period.15 This drug can cause severe adverse effects and should only be prescribed by those experienced in its complex administration.
COMT INHIBITORS
COMT inhibitors (e.g., entacapone [Comtan], tolcapone [Tasmar]) decrease the degradation of levodopa and extend its half-life, thus relieving the end-of-dose wearing-off effect and reducing “off” time. A Cochrane review showed that, compared with placebo, adjuvant COMT inhibitors reduced “off” time and levodopa dose and modestly improved motor symptoms and disability in patients with advanced Parkinson’s disease and motor complications.26 The use of tolcapone requires close monitoring with liver function tests because the drug is rarely associated with potentially fatal hepatotoxicity. Family physicians should consider consultation if a patient is a candidate for tolcapone therapy.26 Carbidopa/levodopa/entacapone (Stalevo) tablets are available for patients currently receiving levodopa who are experiencing the wearing-off effect.
MAO-B INHIBITORS
An 18-week, randomized double-blind trial evaluated the MAO-B inhibitor rasagiline (Azilect), entacapone, and placebo as adjuncts to levodopa in 687 patients with Parkinson’s disease and motor fluctuations. Rasagiline and entacapone reduced “off” time by about 1.2 hours a day compared with 0.4 hours with placebo.27 Both drugs significantly improved Clinical Global Impression scores and reduced the mean daily dose of levodopa. The frequency of adverse effects was similar among all groups, and neither active treatment increased dyskinesia.27
AMANTADINE
A randomized, double-blind trial of amantadine in patients with Parkinson’s disease and dyskinesia showed a 45 percent reduction in dyskinesia with amantadine compared with placebo. However, the benefit lasted for less than eight months, and withdrawal of amantadine caused a 10 to 20 percent rebound increase in dyskinesia.28
SURGERY
Surgical treatment is becoming more common for Parkinson’s disease because of advances in brain imaging and neurosurgical techniques. An evidence-based review concluded that deep brain stimulation of the subthalamic nucleus effectively improves motor function and reduces motor fluctuations, dyskinesia, and antiparkinsonian medication use.15,29 Unilateral pallidotomy is an effective symptomatic adjunct to levodopa and can treat motor complications; however, it is used less often because it causes destructive lesions.15 Referral to specialized centers with surgeons who are experienced in performing this procedure may be considered for patients with advanced Parkinson’s disease and motor complications.
Treatment of Nonmotor Symptoms
Depression, dementia, and psychosis are common psychiatric problems associated with Parkinson’s disease. Treatment of these symptoms is described in Table 4.3 Depression patients currently receiving levodopa who are experiencing the wearing-off effect is usually treated with a selective serotonin reuptake inhibitor. Tricyclic antidepressants should be used with caution in patients with Parkinson’s disease because the drugs can exacerbate orthostatic hypotension and anticholinergic adverse effects.
TABLE 4 Management of Nonmotor Symptoms of Parkinson’s Disease
| Symptom | Management strategies |
|---|---|
| Cognitive impairment | Evaluate for and treat medical problems (e.g., dehydration, metabolic disorders, infection); adjust antiparkinsonian medications; decrease or discontinue anticholinergics, dopamine agonists, amantadine (Symmetrel), and selegiline (Eldepryl); consider a cholinesterase inhibitor. |
| Constipation | Patients should increase fluid and fiber intake; increase physical activity; discontinue anticholinergics; and use stool softeners, lactulose, mild laxatives, or enemas as needed. |
| Depression | Initiate counseling; consider drug therapy with selective serotonin reuptake inhibitors or tricyclic antidepressants (because of side effect profile, use tricyclic antidepressants with caution). |
| Dysphagia | Perform a swallowing evaluation and refer the patient to a subspecialist; increase “on” time (the period when symptoms are decreased), and encourage patients to eat during this time; patient should eat soft foods; consider gastrostomy. |
| Orthostatic hypotension | Discontinue antihypertensive medication; the head of the patient’s bed should be elevated, and patients should rise slowly from a prone position; consider fludrocortisone (Florinef) or midodrine (Proamatine). |
| Psychosis, hallucinations, or delirium | Decrease or discontinue anticholinergics, dopamine agonists, amantadine, and selegiline; decrease levodopa; consider low-dose clozapine (Clozaril) or quetiapine (Seroquel). |
| Sleep disturbance | Daytime somnolence and sleep attacks; discontinue dopamine agonists. |
| Nighttime awakenings because of bradykinesia; consider a bedtime dose of long-acting carbidopa/levodopa (Sinemet), adjuvant entacapone (Comtan), or a dopamine agonist. | |
| Rapid eye movement sleep behavior disorder; decrease or discontinue nighttime use of antiparkinsonian drugs; consider clonazepam (Klonopin). | |
| Urinary urgency | Reduce evening fluid intake; consider tolterodine (Detrol LA) or oxybutynin (Ditropan); refer patient for urology evaluation, if needed. |
Information from reference 3.
Psychosis in patients with Parkinson’s disease is usually drug induced. It can be managed by decreasing doses of anticholinergics or dopamine agonists and by using the lowest possible dose of levodopa. Antipsychotics are indicated for more severe hallucinations. Clozapine (Clozaril) has been shown to effectively manage drug-induced psychosis without worsening parkinsonian symptoms.30 However, clozapine can cause potentially fatal agranulocytosis and requires frequent blood-count monitoring.30 Quetiapine (Seroquel) has not been evaluated in controlled trials. However, it is commonly prescribed because it does not cause hematologic adverse effects and has fewer extrapyramidal adverse effects than other atypical antipsychotics.
About 20 to 40 percent of patients with Parkinson’s disease develop dementia.31 Cholinesterase inhibitors are effective treatments for these patients.30
Nonpharmacologic Treatment
Cochrane reviews on occupational, physical, and speech therapy for patients with Parkinson’s disease have concluded that there is insufficient evidence to support or refute their effectiveness in clinical practice.32–34 Although nonpharmacologic interventions do not improve the cardinal symptoms of Parkinson’s disease, they are helpful in maintaining the overall well-being of patients.
Stretching, strengthening, and balance training may improve gait speed, balance, and participation in activities of daily living.35,36 Specific voice training can effectively treat voice and speech disorders.37 Nutritional interventions (e.g., a high-fiber diet) can help reduce constipation. Dietary amino acids may interfere with levodopa absorption; therefore, protein restriction may be necessary for patients with decreased levodopa response. There is no evidence supporting the use of vitamin E or other antioxidants. Support and counseling are essential for patients with Parkinson’s disease. In one study, patient education was associated with better health-related quality of life.38

