Atopic dermatitis is a common inflammatory skin condition that usually affects children. It is a chronic disease, with periods of remission and flare-ups, that adversely affects the quality of life of patients and their families. Aggressive therapy with emollients is an important intervention for patients with atopic dermatitis. Patients should avoid individual disease triggers and allergens. Topical corticosteroids are the mainstay of treatment for flare-ups and are the standard to which other treatments are compared. Topical calcineurin inhibitors should not be used in patients younger than two years or in those who are immunosuppressed, and should be second-line therapies in other patients. Rarely, systemic agents (e.g., cyclosporine, interferon gamma-1b, oral corticosteroids) may be considered in adults.
Atopic dermatitis is the most common childhood skin disorder in developed countries.1 The prevalence of atopic dermatitis has increased two- to threefold in the last three decades, affecting 15 to 20 percent of young children.2 Clinical findings of atopic dermatitis are variable but can be categorized into three groups of diagnostic features: essential, important, and associated (Table 1).3 Atopic dermatitis is common in flexural areas and on the cheeks and buttocks3 (Figure 1). Prognosis usually is favorable because children commonly outgrow the condition by early adolescence. Persistent cases are associated with early disease onset; severe, widespread disease; concomitant asthma or hay fever; and a family history of atopic dermatitis.4
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Emollients are the mainstay of maintenance therapy for atopic dermatitis. | B | 3, 4, 10 |
| Topical corticosteroids should be first-line treatments for patients with atopic dermatitis flare-ups. | A | 3, 4, 11 |
| Sedating antihistamines are indicated for the treatment of atopic dermatitis when patients have sleep disturbances and concomitant allergic conditions. | A | 11, 13 |
| Antibiotics should be reserved for the treatment of acutely infected lesions associated with atopic dermatitis. | A | 4 |
| Topical calcineurin inhibitors should be second-line treatments for atopic dermatitis flare-ups and maintenance. | A | 25 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 453 or https://www.aafp.org/afpsort.xml.
TABLE 1 Clinical Features of Atopic Dermatitis
| Essential features* | |
| Pruritus | |
| Eczema (acute, subacute, chronic) | |
| Typical morphology and age-specific patterns (i.e., facial, neck, and extensor involvement in children; current or previous flexural lesions in any age group; sparing of groin and axillary regions) | |
| Chronic or relapsing history | |
| Important features† | |
| Onset at early age | |
| Atopy | |
| Personal or family history | |
| Immunoglobulin E reactivity | |
| Xerosis | |
| Associated features‡ | |
| Atypical vascular responses (e.g., facial pallor, white dermatographism, delayed blanch response) | |
| Keratosis pilaris, hyperlinear palms, and ichthyoses | |
| Ocular or periorbital changes | |
| Other regional findings (e.g., perioral changes, periauricular lesions) | |
| Perifollicular accentuation, lichenification, and prurigo lesions | |
note:An atopic dermatitis diagnosis depends on excluding conditions such as scabies, seborrheic dermatitis, allergic contact dermatitis, ichthyosis, cutaneous lymphoma, psoriasis, and immunodeficiency disorders.
*— Essential features must be present for an atopic dermatitis diagnosis.
†— Important features are seen in most patients, supporting the diagnosis.
‡— Clinical associations help to suggest the diagnosis but are too nonspecific to define or detect atopic dermatitis in research or epidemiologic studies.
Adapted with permission from Eichenfield LF, Hanifin JM, Luger TA, Stevens SR, Pride HB. Consensus conference on pediatric atopic dermatitis. J Am Acad Dermatol 2003;49:1088.
Figure 1.
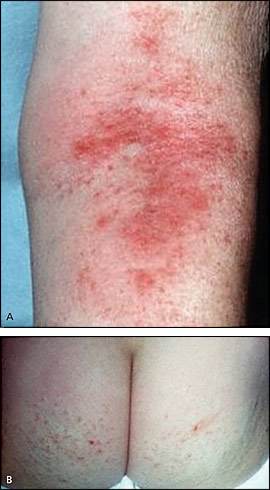
Atopic dermatitis. (A) Flexural areas are common locations for recurrent atopic dermatitis in children and adults. (B) Papular atopic dermatitis of the buttocks is more common in adults.
The cause of atopic dermatitis is multi-factorial, with genetics, environment, and impaired immune response being the most predominant factors. The role of genetics has been demonstrated in studies of families and twins.5 Numerous environmental factors have been associated with atopic dermatitis: exposure to allergens, irritants, bacteria, and hard water; socioeconomic status; and large family size.6 There is evidence that atopic dermatitis is a risk factor for childhood asthma, affecting asthma occurrence, severity, and persistence. Atopic dermatitis is likely related to early immunoglobulin E (IgE) production and later allergen/IgE reactivity.7 The impaired immune response is characterized by activation of T-helper type 2 cells, leading to increased interleukin-4 production, which promotes IgE production.
Atopic dermatitis can have a significant impact on morbidity and quality of life. Children may be affected by itching and associated sleep disturbance, the social stigma of a visible skin condition, and the need for frequent application of topical medications and physician visits. It has been estimated that children with atopic dermatitis lose an average of 1.9 hours of sleep per night, and their parents lose an average of 2.1 hours per night.8 Other significant problems reported in children with atopic dermatitis include irritability, daytime tiredness, dependence, fearfulness, and mood changes.9
Symptom Management
Measures to help prevent and treat atopic dermatitis symptoms should be implemented. Emollient creams can prevent and soothe dry, irritated skin, and antihistamines can treat pruritus from atopic dermatitis.
GENERAL PREVENTIVE MEASURES
Preventing flare-ups with good skin-care practices is an important part of the overall treatment of atopic dermatitis. Dry skin is a feature in nearly all patients with the condition. Emollients are the mainstay of maintenance therapy for atopic dermatitis.3,4,10 Treatment guidelines from the United States and the United Kingdom recommend the use of emollients with or without moisturizers.10,11 Patients should bathe in warm (not hot) water and use mild, unscented soaps or soap-free cleansers.
Liberal amounts of a lubricant or emollient cream should be applied to the skin immediately after bathing. Emollients should be applied once or twice daily to prevent skin dryness and irritation. Patients generally prefer emollient creams over ointments for daytime use because emollients have a nongreasy, cosmetic appearance. Lubricating ointments may be preferred for nighttime use because of their superior hydrating properties. Wearing cotton gloves or socks at night may enhance these properties.
Numerous studies have evaluated a variety of dietary, environmental, and alternative approaches to the prevention of atopic dermatitis flare-ups.3,4,11 Unfortunately, many of these approaches have been shown to be ineffective (Table 2).4 Expert opinion supports the use of comfortable fabrics (e.g., cotton or other smooth fibers) for clothing and bedding.3 Patients should avoid known environmental or dietary triggers. Irritants that cause itching also should be avoided. The development of the “scratch-itch-scratch” behavior that begins with habitual scratching and perpetuates dry, irritated skin can be effectively modified with psychological treatment.12
TABLE 2 Unproven Prevention and Treatment Strategies for Atopic Dermatitis
| Chinese herbal therapy |
| Delayed introduction of solid foods in infants |
| Dietary restrictions |
| Homeopathy |
| Massage therapy |
| Prolonged breastfeeding |
| Reduction of house mite dust |
| Salt baths |
| Use of different diaper materials |
Information from reference 4.
ANTIHISTAMINES
The use of sedating and nonsedating antihistamines to treat pruritus associated with atopic dermatitis has been shown to be ineffective when compared with placebo.13 The use of sedating antihistamines can be beneficial in the treatment of comorbidities (e.g., allergic rhinitis, dermatographism, urticaria) and sleep disturbances.11,13 Additionally, there is no evidence that adding a histamine H2 antagonist is beneficial.4
Treatment
Topical corticosteroids have been the mainstay of treatment for atopic dermatitis flare-ups and are the agents to which other treatments are compared.4,11 Calcineurin inhibitors should be used as second-line agents, and, rarely, systemic therapies may be considered in adults.
TOPICAL CORTICOSTEROIDS
More than 30 topical corticosteroids are available, ranging from low to high potency. Most of these agents are available in varying concentrations and doses; nearly all are available in generic formulations. Unfortunately, there is a paucity of clinical trial data to assist in choosing a corticosteroid. Table 3 provides an overview of common topical corticosteroids.14
TABLE 3 Overview of Common Topical Corticosteroid Therapies
| Therapy | Relative potency class* | Preparation | Cost (generic) for 15 g† |
|---|---|---|---|
| Alclometasone dipropionate 0.05% (Aclovate) | 6 | Cream, ointment | $26 (19 to 20) |
| Betamethasone dipropionate 0.05% (Diprolene) | 3 | Cream | 48 (4 to 8) |
| Betamethasone dipropionate 0.05% | 2 | Ointment | 48 (4 to 8) |
| Betamethasone valerate 0.1% (Beta-Val) | 5 | Cream | 5 (3 to 6) |
| Betamethasone valerate 0.1% | 3 | Ointment | — (4 to 6) |
| Clobetasol propionate 0.05% (Temovate) | 1 | Cream, ointment | 46 (21 to 25) |
| Desonide 0.05% (Desowen) | 6 | Cream | 29 (12 to 15) |
| Desoximetasone 0.25% (Topicort) | 2 | Cream, ointment | 42 (19 to 26) |
| Fluocinolone acetonide 0.01% (Synalar) | 6 | Cream, solution | Cream: 39 (2 to 3) |
| Solution (20 mL): 49 (5 to 11) | |||
| Fluocinonide 0.05% (Lidex) | 2 | Cream, ointment, gel, solution | 47 (9 to 21) |
| Fluticasone propionate 0.005% (Cutivate) | 3 | Ointment | 28 (22 to 23) |
| Fluticasone propionate 0.05% | 5 | Cream | 28 (22 to 23) |
| Halobetasol propionate 0.05% (Ultravate) | 1 | Cream, ointment | 31 to 32 |
| Hydrocortisone 0.5 to 2.5% | 7 | Cream, ointment, lotion | Cream, ointment: — (2 to 4) |
| Lotion (120 mL): — (7 to 10) | |||
| Hydrocortisone butyrate 0.1% (Locoid) | 5 | Cream, ointment, solution | Cream, ointment: — (2 to 4) |
| Lotion (120 mL): — (7 to 10) | |||
| Hydrocortisone valerate 0.2% (Westcort) | 5 | Cream | 18 (15 to 25) |
| Hydrocortisone valerate 0.2% | 4 | Ointment | 18 (15 to 16) |
| Mometasone furoate 0.1% (Elocon) | 4 | Cream, ointment, lotion | Cream, ointment: 31 (24 to 27) |
| Solution (30 mL): 33 (29 to 30) | |||
| Triamcinolone acetonide 0.025 to 0.1% (Kenalog) | 5 | Cream | 11 (4 to 5) |
| Triamcinolone acetonide 0.1% | 4 | Ointment | 13 (3 to 4) |
| Triamcinolone diacetate 0.5% (Aristocort) | 3 | Cream | 37 |
*— Highest potency class is 1 and lowest potency class is 7.
†— Estimated cost to the pharmacist based on average wholesale prices (rounded to the nearest dollar) in Red Book. Montvale, N.J.: Medical Economics Data, 2006. Cost to the patient will be higher, depending on prescription filling fee. Larger quantities generally cost less per gram.
Adapted with permission from Habif TP. Clinical Dermatology: A Color Guide to Diagnosis and Therapy. 3rd ed. St. Louis, Mo.: Mosby, 1996:9–11.
General prescribing practices for topical corticosteroids apply to the treatment of atopic dermatitis. Only low-potency (class 6 or 7) agents should be used on the face, groin, and axillae to minimize local side effects such as acne, striae, telangiectasia, and atrophy. Low-potency agents also are preferred in infants because infants have a relatively higher ratio of skin surface area to body mass than older children and adults and because of the increased potential for systemic absorption with these drugs.
The method of application of a corticosteroid can influence potency of the active ingredient. Ointments generally are more potent than creams but may have a greasy appearance. Ointments should be avoided on open or oozing lesions and in intertriginous folds. They also should not be used in hot, moist climates. Creams may contain preservatives that can precipitate contact dermatitis. Lotions generally lack the hydrating properties necessary for treating atopic dermatitis. An adequate supply of a topical corticosteroid is essential for effective treatment and patient adherence to therapy (Table 414–16 ). Patients generally underestimate the appropriate quantity of topical corticosteroids and emollients needed for long-term therapy.16 Agents with poor cosmetic appeal may interfere with medication adherence.
TABLE 4 Appropriate Quantity of Topical Corticosteroids for the Treatment of Atopic Dermatitis
Compelling evidence regarding the most appropriate frequency of topical corticosteroid application and the role of the vehicle used to deliver the active ingredient generally is lacking. There is no evidence that more frequent application of hydrocortisone butyrate 0.1% (Locoid) or fluticasone propionate 0.05% (Cutivate) cream is more effective than once-daily dosing.17,18 It is difficult to determine if this applies to other topical corticosteroids, although current treatment guidelines do not recommend more than twice-daily application of topical corticosteroids.3,4
Clinical trials have shown that topical corticosteroids are safe and effective for the treatment of atopic dermatitis flare-ups when used for up to four weeks, although many flare-ups may be adequately controlled with a shorter treatment course.19–21 To minimize toxicity, topical corticosteroids should be used for the shortest duration needed to control the flare-up. After the flare-up resolves, maximal preventive strategies should be used to control the disease. Topical corticosteroids do not cure atopic dermatitis.
Long-term topical corticosteroid use is associated with local and systemic adverse effects that may lead to the underutilization of these effective agents.22 Common local adverse effects include striae, petechiae, telangiectasia, skin thinning, atrophy, and worsening acne. These effects are reported infrequently in clinical trials, although trials are primarily designed to assess effectiveness rather than safety and tolerability. Most clinical trials of topical steroids are of short duration and, therefore, are unable to evaluate long-term toxicity.
Systemic adverse effects (primarily hypothalamic-pituitary-adrenal axis suppression, reduced linear growth in children, and bone density changes in adults) are the most worrisome side effects associated with corticosteroids. As with local adverse effects, it is difficult to assess systemic adverse effects based on the current literature. There is no conclusive evidence that properly used topical corticosteroids cause significant systemic adverse effects. At least two randomized trials of long-term (16 to 24 weeks) intermittent dosing showed no clinical evidence of skin thinning, atrophy, or hypothalamic-pituitary-adrenal axis suppression.23,24
TOPICAL CALCINEURIN INHIBITORS
Calcineurin inhibitors (pimecrolimus [Elidel] and tacrolimus [Protopic]) are immunosuppressant agents originally developed for systemic administration to prevent allogeneic transplant rejection. These agents inhibit calcineurin in the skin, which blocks early T-cell activation and the release of cytokines. Topical formulations were developed as alternatives to topical corticosteroids.
A meta-analysis demonstrated that tacrolimus 0.1% is as effective as potent corticosteroids and more effective than mild topical corticosteroids in the treatment of atopic dermatitis.25 The meta-analysis showed that pimecrolimus 0.03% is less effective than the corticosteroid betamethasone valerate 0.1% (Beta-Val).25 The effectiveness of pimecrolimus compared with less potent topical corticosteroids is unknown. Although pimecrolimus has been shown to prevent more flare-ups than vehicle alone, there are no available data comparing low-potency corticosteroids with pimecrolimus to prevent flare-ups.25
Tacrolimus and pimecrolimus also have adverse effects, although they are different than those associated with topical corticosteroids. The most common local adverse effects are skin burning and irritation. Patients using topical calcineurin inhibitors should be counseled on appropriate sun protection, including sunscreen application. Whether these agents induce local or distant malignancy is unclear. However, because of several case reports and additional animal data, the U.S. Food and Drug Administration has approved label revisions for these agents including a second-line indication, enhanced warnings, and a patient education guide.26 The warning statement recommends avoiding long-term use in all patient populations and limits use to children older than two years.26 The complete label information is available at http://www.fda.gov/bbs/topics/news/2006/NEW01299.html.
ANTIBIOTICS
Most patients with atopic dermatitis have Staphylococcus aureus infection.4 The relationship between S. aureus infection and atopic dermatitis flare-ups has been debated but remains unclear. Concerns about resistance limit the use of antibiotics to treating acute skin lesions, rather than decolonization when the skin has not been affected.4 The use of antiseptic baths and washes also should be avoided.3
SYSTEMIC THERAPY
Rarely, systemic therapy is indicated for severe, resistant disease. Systemic corticosteroids are effective at acutely controlling atopic dermatitis in adults, but their use should be restricted to the short term. Rebound flare-ups and diminishing effectiveness severely limit use.27 Agents such as cyclosporine (Sandimmune) and interferon gamma-1b (Actimmune) may be effective for severe atopic dermatitis. Data on the use of mycophenolate mofetil (Cellcept), azathioprine (Imuran), and intravenous immune globulin (human; Baygam) are conflicting, and there is no evidence to support the use of leukotriene inhibitors, methotrexate, desensitization injections, theophylline, or oral pimecrolimus.11
OTHER THERAPIES
Ultraviolet (UV) phototherapy using UVB, narrow-band UVB, UVA, or psoralen plus UVA may be beneficial for the treatment of severe disease if it is used appropriately, depending on the patient's age.4
Figures 1 and 2 printed with permission from Michelle Daffer, M.D.

