Hypertriglyceridemia is associated with an increased risk of cardiovascular events and acute pancreatitis. Along with lowering low-density lipoprotein cholesterol levels and raising high-density lipoprotein cholesterol levels, lowering triglyceride levels in high-risk patients (e.g., those with cardiovascular disease or diabetes) has been associated with decreased cardiovascular morbidity and mortality. Although the management of mixed dyslipidemia is controversial, treatment should focus primarily on lowering low-density lipoprotein cholesterol levels. Secondary goals should include lowering non–high-density lipoprotein cholesterol levels (calculated by subtracting high-density lipoprotein cholesterol from total cholesterol). If serum triglyceride levels are high, lowering these levels can be effective at reaching non–high-density lipoprotein cholesterol goals. Initially, patients with hypertriglyceridemia should be counseled about therapeutic lifestyle changes (e.g., healthy diet, regular exercise, tobacco-use cessation). Patients also should be screened for metabolic syndrome and other acquired or secondary causes. Patients with borderline-high serum triglyceride levels (i.e., 150 to 199 mg per dL [1.70 to 2.25 mmol per L]) and high serum triglyceride levels (i.e., 200 to 499 mg per dL [2.26 to 5.64 mmol per L]) require an overall cardiac risk assessment. Treatment of very high triglyceride levels (i.e., 500 mg per dL [5.65 mmol per L] or higher) is aimed at reducing the risk of acute pancreatitis. Statins, fibrates, niacin, and fish oil (alone or in various combinations) are effective when pharmacotherapy is indicated.
Hypertriglyceridemia as an independent risk factor for coronary heart disease is controversial. Observational studies have shown an association between increased cardiovascular risk and hypertriglyceridemia.1 In addition, metabolic syndrome includes hypertriglyceridemia and low levels of high-density lipoprotein cholesterol (HDL-C).2 Subgroup analysis in trials of lipid-lowering therapy showed that patients with atherogenic dyslipidemia (i.e., those with high triglyceride and apolipoprotein B, low HDL-C, and small low-density lipoprotein particles) had the greatest reduction in coronary events.3,4
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Patients with high serum triglyceride levels should receive counseling about a healthy diet, regular exercise, and tobacco-use cessation. | B | 7,35 |
| After patients with high serum triglyceride levels reach their LDL-C goals, the secondary target should be reaching non–HDL-C goals (30 mg per dL [0.78 mmol per L] higher than the LDL-C goal). | C | 7 |
| Fibrates, niacin, or fish oil can be considered to help lower triglyceride and non–HDL-C levels. | C | 7 |
| Serum triglyceride levels should be lowered in patients with very high triglyceride levels to prevent acute pancreatitis. | C | 7 |
LDL-C = low-density lipoprotein cholesterol; HDL-C = high-density lipoprotein cholesterol.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 1289 orhttps://www.aafp.org/afpsort.xml.
It is unclear whether metabolic syndrome and hypertriglyceridemia are true causal cardiovascular risk factors that can be modified with treatment or are merely biomarkers of future risk. Although research shows that triglyceride reduction is associated with decreased cardiovascular events (particularly among patients with known heart disease),5,6 it is unclear if this is independent of improvement in other lipid parameters, if this also applies to primary prevention, or if all-cause mortality rates are improved. Nonetheless, guidelines from the National Cholesterol Education Program7 and the American Heart Association2 have identified a role for triglyceride control and diagnosis of metabolic syndrome in the management of dyslipidemia.
Pharmacologic Agents
Statins, fibrates, niacin, and fish oil are the main pharmacologic agents for managing hypertriglyceridemia (Table 17–9 ).
Table 1 Selected Therapies for Managing Hypertriglyceridemia
| Therapy | Triglyceride reduction (%) | LDL-C increase/reduction (%) | HDL-C increase (%) | Possible side effects |
|---|---|---|---|---|
| Statins | 20 to 40 | 18 to 55 reduction | 5 to 15 | Myopathy, rhabdomyolysis, elevated liver enzyme levels |
| Atorvastatin (Lipitor), 10 to 80 mg daily | ||||
| Fluvastatin (Lescol), 20 to 80 mg daily at bedtime | ||||
| Lovastatin (Mevacor), 10 to 80 mg daily at bedtime | ||||
| Pravastatin (Pravachol), 10 to 80 mg daily | ||||
| Rosuvastatin (Crestor), 5 to 20 mg daily | ||||
| Simvastatin (Zocor), 5 to 80 mg dailyat bedtime | ||||
| Fibrates | 40 to 60 | 5 to 30 increase | 15 to 25 | Rhabdomyolysis, especially with a gemfibrozil/statin combination |
| Fenofibrate (Tricor), 48 to 145 mg daily | ||||
| Gemfibrozil (Lopid), 600 mg twice daily | ||||
| Niacin | 30 to 50 | 5 to 25 reduction | 20 to 30 | Flushing; worsening glycemic control; elevated liver enzyme levels, especially with OTC sustained-release niacin |
| OTC immediate-release niacin, 0.5 to 2 g two or three times daily | ||||
| OTC sustained-release niacin, 250 to 750 mg once or twice daily | ||||
| Prescription niacin, 500 mg to 2 g daily at bedtime | ||||
| Fish oil, 2 to 4 g total EPA/DHA daily | 30 to 50 | 5 to 10 increase | 5 to 10 | Fishy aftertaste, gastrointestinal upset |
| OTC omega-3 fatty acid capsules | ||||
| Prescription omega-3-acid ethyl esters (Omacor), 1 to 2 g twice daily |
LDL-C = low-density lipoprotein cholesterol; HDL-C = high-density lipoprotein cholesterol; OTC = over the counter; EPA = eicosapentaenoic acid; DHA = docosahexaenoic acid.
STATINS
The primary target of lipid therapy is to reach low-density lipoprotein cholesterol (LDL-C) goals.7,10 Statins have an important role in reducing the risk of cardiovascular events in patients with elevated LDL-C levels, particularly in high-risk patients (e.g., those with cardiovascular disease or diabetes). If hyper-triglyceridemia is a comorbidity, statins can lower triglyceride levels by 20 to 40 percent.7,11
FIBRATES
Fibrates can markedly lower triglyceride levels (40 to 60 percent) and modestly raise HDL-C levels (15 to 25 percent).7 In patients with cardiovascular disease and moderately elevated triglyceride levels and low HDL-C levels, fibrates have been shown to decrease the risk of cardiovascular events (secondary prevention).12,13 Fibrate therapy also has been shown to decrease angiographic progression of coronary heart disease in patients with type 2 diabetes.14
Because data show decreased cardiovascular mortality rates with triglyceride reduction (more than that achieved with LDL-C reduction alone),3 there is increasing interest in fibrate use in patients with hypertriglyceridemia and in combination fibrate/statin therapy in patients with mixed dyslipidemia. However, despite several large studies, no fibrate has been shown to decrease all-cause mortality rates, and some trials have shown an increase in all-cause mortality rates.15–17 A recent primary prevention trial of fenofibrate (Tricor), which included 9,795 patients with type 2 diabetes, did not significantly decrease the primary end points of coronary events or all-cause mortality but decreased the secondary end point of total cardiovascular events (i.e., coronary heart disease events, stroke, or revascularization).15
Combination therapy raises safety concerns. All statins (especially at higher doses) increase the risk of rhabdomyolysis; this risk may be compounded by fibrate use. Cerivastatin (Baycol) was withdrawn from the market because of reports of fatal rhabdomyolysis, often in patients also taking gem-fibrozil (Lopid). An increased risk also has been shown with rosuvastatin (Crestor).18,19 When combined with statins, gemfibrozil may increase serum statin levels by inhibiting statin metabolism.
Compared with gemfibrozil/statin therapy, fenofibrate/statin therapy has a lower incidence and reported rate of rhabdomyolysis and may be safer.20,21 However, long-term safety and outcome data for fibrate/statin combinations are lacking, and combination therapy should be used with caution. Patients should receive the lowest possible statin dosage, be monitored closely for side effects (e.g., muscle pain, brown urine), and be given the opportunity for proper informed consent.
NIACIN
Niacin lowers triglyceride levels by 30 to 50 percent, raises HDL-C levels by 20 to 30 percent, and lowers LDL-C levels by 5 to 25 percent.7,8 Niacin is not as potent as fibrates for lowering triglyceride levels but is more effective at raising HDL-C levels.
Studies evaluating niacin's effect on cardiovascular and all-cause mortality are limited. The largest study (the Coronary Drug Project) revealed that patients treated with niacin had a modest decrease in nonfatal myocardial infarction (8.9 versus 12.2 percent) but no difference in all-cause mortality after five years.22 A nine-year, nonrandomized, nonblinded follow-up study to the Coronary Drug Project revealed a decrease in all-cause mortality rates in the original cohort treated with niacin (52 versus 58 percent;P = .004; number needed to treat = 17 patients for 15 years).23
Concerns over worsening glycemic control may limit the use of niacin in patients with diabetes. However, a study of patients taking lipid-lowering doses of niacin demonstrated that niacin is beneficial without significantly affecting glycemic control.24 A consensus report recommended that niacin therapy be considered for high-risk patients with elevated triglyceride and low HDL-C levels, even with coexistent diabetes.25
The use of niacin is limited because of the risk of vasomotor side effects and elevation of liver enzyme levels.8 Flushing and hepatotoxicity can be minimized by starting with low doses and slowly titrating upward using extended-release formulations and by concurrent use of aspirin taken 30 minutes before niacin. Low-dose niacin combined with a statin has been associated with a significant decrease in cardiovascular events26; however, this combination has not been compared with either agent alone.
FISH OIL
Fish oil contains high amounts of the essential fatty acids docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA); these acids also are known as omega-3 fatty acids. A systematic review showed that fish oil is effective at lowering triglyceride levels.9 Fish oil with 2 to 4 g of total EPA/DHA daily can lower triglyceride levels by 30 to 50 percent.7,27 Randomized controlled trials have shown that, along with statins, fish oil is the only other lipid-lowering agent that can decrease all-cause mortality in patients with known heart disease.17,28
The GISSI-Prevenzione trial showed a 15 percent reduction in all-cause mortality in patients with a recent myocardial infarction who were taking fish oil.29 This reduction was in addition to optimal management with lipid-lowering therapy, antiplatelet agents, beta blockers, and angiotensin-converting enzyme inhibitors.30 These results must be interpreted with caution because a systematic review that combined data from primary and secondary prevention studies showed no mortality benefit.31 If there are benefits to using fish oil in patients with heart disease, they are most likely multifactorial and go beyond triglyceride effects alone.
Studies of fish oil/statin combinations demonstrate an additional 30 percent triglyceride reduction.32,33 Side effects are minimal and include a fishy aftertaste and mild gastrointestinal upset. Bleeding effects are theoretical and have not been shown to be clinically significant even in large doses. Omacor, a newly approved prescription medication for hypertriglyceridemia, is a highly concentrated form of omega-3-acid ethyl esters and is available in 1-g capsules (840 mg EPA/DHA). Over-the-counter capsules are readily available, but physicians should make certain that patients receive 2 to 4 g of total EPA/DHA per day. Most over-the-counter preparations only contain 300 mg of EPA/DHA per capsule.34
Management
Patients should receive initial evaluations and counseling, and further management should be determined based on their risk profiles and the extent of hypertriglyceridemia. Figure 17 presents an algorithm for managing hypertriglyceridemia. Table 27 lists previous and current serum triglyceride classifications.
Figure 1. Management of Hypertriglyceridemia
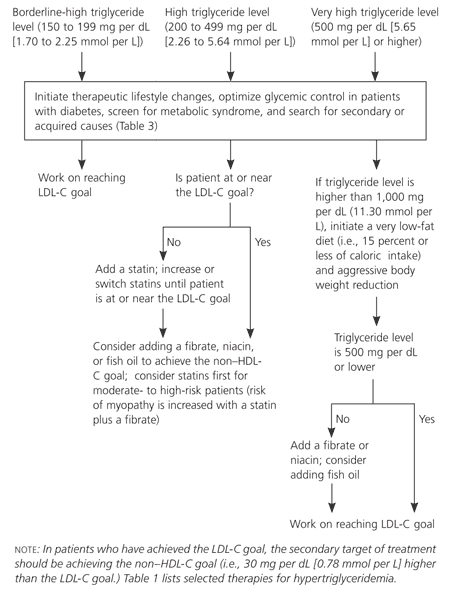
Algorithm for the management of hypertriglyceridemia. (LDL-C= low-density lipoprotein cholesterol; HDL-C= high-density lipoprotein cholesterol.)
Information from reference 7.
Table 2 ATP II and ATP III serum triglyceride classifications
| Triglyceride classification | ATP II levels | ATP III levels |
|---|---|---|
| Normal | Lower than 200 mg per dL (2.26 mmol per L) | Lower than 150 mg per dL (1.70 mmol per L) |
| Borderline high | 200 to 399 mg per dL (2.26 to 4.51 mmol per L) | 150 to 199 mg per dL (1.70 to 2.25 mmol per L) |
| High | 400 to 1,000 mg per dL (4.52 to 11.30 mmol per L) | 200 to 499 mg per dL (2.26 to 5.64 mmol per L) |
| Very high | Higher than 1,000 mg per dL | 500 mg per dL (5.65 mmol per L) or higher |
note:This table compares the older ATP II classification with the current ATP III classification.
ATP = Adult Treatment Panel.
Adapted from Third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III): final report. NIH publication no.: 02-5215. Bethesda, Md.: National Heart, Lung, and Blood Institute, 2002:II–7
INITIAL CONSIDERATIONS
Initial management of hypertriglyceridemia (Table 37 ) should include counseling for therapeutic lifestyle changes (e.g., weight control, including diet and exercise; tobacco-use cessation)7,35 and screening for metabolic syndrome. Patients also should be screened for other acquired or secondary causes.7 If the patient has diabetes, optimizing glycemic control may help lower triglyceride levels without additional medications for hypertriglyceridemia.7
Physicians should stratify the patient's risk to determine a lipid treatment goal. High-risk patients include those with a calculated 10-year coronary heart disease risk above 20 percent and those with known cardiovascular disease or diabetes. Literature on the screening, diagnosis, and nonphar-macologic management of hyperlipidemia is readily available.2,10
In many patients with hypertriglyceridemia, pharmacotherapy is indicated after implementing adequate therapeutic lifestyle changes. The initial goal of pharmacologic therapy is to achieve individual LDL-C goals, which are determined after an assessment of cardiovascular risk. After LDL-C goals are achieved, non–HDL-C goals are the secondary target for therapy.7 Non–HDL-C is calculated by subtracting HDL-C from total cholesterol. The non–HDL-C goal is 30 mg per dL (0.78 mmol per L) higher than the LDL-C goal.
Table 3 Initial Management of Hypertriglyceridemia
| Intervention | Description | Comments |
|---|---|---|
| Counsel patients about therapeutic lifestyle changes | Body weight control, regular physical activity, tobacco- use cessation, avoidance of high-carbohydrate foods, diet low in saturated fat and sugar | Patients with triglyceride levels above 1,000 mg per dL (11.30 mmol per L) should immediately start a very low-fat diet |
| Screen for metabolic syndrome | Constellation of increased abdominal circumference and low HDL-C levels, high triglyceride and blood sugar levels, and elevated blood pressure | Diagnosis and management remain controversial |
| Search for secondary causes | Nephrotic syndrome, diabetes, chronic renal failure, hypothyroidism, various medications | Optimizing glycemic control may improve hypertriglyceridemia |
| Search for acquired causes | Overweight and obesity, excessive alcohol intake, high carbohydrate intake, tobacco use | — |
| Determine cardiac risk profile | Determine cardiac risk factors, and stratify the patient's 10-year risk of coronary heart disease using Framingham risk calculators | — |
HDL-C = high-density lipoprotein cholesterol.
Information from reference 7
BORDERLINE-HIGH TRIGLYCERIDE LEVELS
Drug therapy is not indicated for patients with borderline-high triglyceride levels (i.e., 150 to 199 mg per dL [1.70 to 2.25 mmol per L]). Instead, the physician should consider screening for metabolic syndrome and other acquired or secondary causes of hypertriglyceridemia. LDL-C reduction is the primary goal.
HIGH TRIGLYCERIDE LEVELS
In patients with high triglyceride levels (i.e., 200 to 499 mg per dL [2.26 to 5.64 mmol per L]), lowering triglyceride levels also can lower non–HDL-C levels. Statins with triglyceride-lowering properties are first-line agents for patients who have not reached their LDL-C goals. In patients with high triglyceride levels but no heart disease (or a heart disease equivalent such as peripheral artery or carotid artery disease) who are at or near their LDL-C goals, a fibrate, niacin, or fish oil can be considered to help patients reach their non–HDL-C goals. However, physicians should keep in mind that prospective data from primary prevention trials are lacking.
With newer, more aggressive recommendations for lowering high LDL-C levels in high-risk patients, more patients taking statins likely will require combination therapy to reach LDL-C and non–HDL-C goals.36 Combination therapy with a statin plus fibrate, niacin, or fish oil generally is well-tolerated, although patients should be appropriately monitored. There are no data showing that one combination is superior to others. Which agent to combine with a statin should be determined for individual patients. Niacin may be most appropriate in patients with low HDL-C and high LDL-C levels, whereas a fibrate may be most appropriate in patients at their LDL-C and HDL-C goals but who have elevated triglyceride levels. If a fibrate combination is chosen, fenofibrate may be safer than gemfibrozil.21 Omega-3 fatty acids are recommended for patients with coronary heart disease,37 and fish oil may be better tolerated with less drug-drug interactions than fibrates or niacin.
VERY HIGH TRIGLYCERIDE LEVELS
Patients with very high triglyceride levels (i.e., 500 mg per dL [5.65 mmol per L] or higher) usually require drug therapy in addition to therapeutic lifestyle changes. Fibrates or niacin is a practical first-line choice for these patients.7 The initial goal is to decrease the risk of acute pancreatitis, especially if triglyceride levels are above 1,000 mg per dL (11.30 mmol per L). In addition, patients with a triglyceride level of 1,000 mg per dL or higher should be placed on a very low-fat diet (i.e., 15 percent or less of caloric intake). Normalization of triglyceride levels is rarely achieved in patients with severe hypertriglyceridemia (i.e., triglyceride levels above 2,000 mg per dL [22.60 mmol per L]). Initiating a combination of fibrates, niacin, and/or fish oil to lower triglyceride levels to below 500 mg per dL is the primary goal. Because there is little evidence on preventing pancreatitis with hypertriglyceridemia treatment, combination therapy should be determined for individual patients and used with caution.

