Venous ulcer, also known as stasis ulcer, is the most common etiology of lower extremity ulceration, affecting approximately 1 percent of the U.S. population. Possible causes of venous ulcers include inflammatory processes resulting in leukocyte activation, endothelial damage, platelet aggregation, and intracellular edema. The primary risk factors for venous ulcer development are older age, obesity, previous leg injuries, deep venous thrombosis, and phlebitis. On physical examination, venous ulcers are generally irregular, shallow, and located over bony prominences. Granulation tissue and fibrin are typically present in the ulcer base. Associated findings include lower extremity varicosities, edema, venous dermatitis, and lipodermatosclerosis. Venous ulcers are usually recurrent, and an open ulcer can persist for weeks to many years. Severe complications include cellulitis, osteomyelitis, and malignant change. Poor prognostic factors include large ulcer size and prolonged duration. Evidence-based treatment options for venous ulcers include leg elevation, compression therapy, dressings, pentoxifylline, and aspirin therapy. Surgical management may be considered for ulcers that are large in size, of prolonged duration, or refractory to conservative measures.
Venous ulcers, or stasis ulcers, account for 80 percent of lower extremity ulcerations.1 Less common etiologies for lower extremity ulcerations include arterial insufficiency; prolonged pressure; diabetic neuropathy; and systemic illness such as rheumatoid arthritis, vasculitis, osteomyelitis, and skin malignancy.2 The overall prevalence of venous ulcers in the United States is approximately 1 percent.1 Venous ulcers are more common in women and older persons.3–6 The primary risk factors are older age, obesity, previous leg injuries, deep venous thrombosis, and phlebitis.7
Venous ulcers are often recurrent, and open ulcers can persist from weeks to many years.8–10 Severe complications include cellulitis, osteomyelitis, and malignant change.3 Although the overall prevalence is relatively low, the refractory nature of these ulcers increase the risk of morbidity and mortality, and have a significant impact on patient quality of life.11,12 The financial burden of venous ulcers is estimated to be $2 billion per year in the United States.13,14
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Compression therapy has been proven beneficial for venous ulcer treatment and is the standard of care. | A | 2, 7, 10, 22–26, 45 |
| Leg elevation minimizes edema in patients with venous insufficiency and is recommended as adjunctive therapy for venous ulcers. The recommended regimen is 30 minutes, three or four times per day. | C | 27 |
| Dressings are beneficial for venous ulcer healing, but no dressing has been shown to be superior. | A | 28, 29 |
| Pentoxifylline (Trental) is effective when used with compression therapy for venous ulcers, and may be useful as monotherapy. | A | 31 |
| Aspirin (300 mg per day) is effective when used with compression therapy for venous ulcers. | B | 32 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Pathophysiology
The pathophysiology of venous ulcers is not entirely clear. Venous incompetence and associated venous hypertension are thought to be the primary mechanisms for ulcer formation. Factors that may lead to venous incompetence include immobility; ineffective pumping of the calf muscle; and venous valve dysfunction from trauma, congenital absence, venous thrombosis, or phlebitis.14 Subsequently, chronic venous stasis causes pooling of blood in the venous circulatory system triggering further capillary damage and activation of inflammatory process. Leukocyte activation, endothelial damage, platelet aggregation, and intracellular edema contribute to venous ulcer development and impaired wound healing.2,14
Clinical Presentation and Diagnosis
Determining etiology is a critical step in the management of venous ulcers. Characteristic differences in clinical presentation and physical examination findings can help differentiate venous ulcers from other lower extremity ulcers (Table 1).2 The diagnosis of venous ulcers is generally clinical; however, tests such as ankle-brachial index, color duplex ultrasonography, plethysmography, and venography may be helpful if the diagnosis is unclear.15–18
Table 1. Common Lower Extremity Ulcers
| Ulcer type | General characteristics | Pathophysiology | Clinical features | Treatment options |
|---|---|---|---|---|
| Venous | Most common type; women affected more than men; often occurs in older persons | Venous hypertension | Shallow, painful ulcer located over bony prominences, particularly the gaiter area (over medial malleolus); granulation tissue and fibrin present | Leg elevation, compression therapy, aspirin, pentoxifylline (Trental), surgical management |
| Associated findings include edema, venous dermatitis, varicosities, and lipodermatosclerosis | ||||
| Arterial | Associated with cardiac or cerebrovascular disease; patients may present with claudication, impotence, pain in distal foot; concomitant with venous disease in up to 25 percent of cases | Tissue ischemia | Ulcers are commonly deep, located over bony prominences, round or punched out with sharply demarcated borders; yellow base or necrosis; exposure of tendons Associated findings include abnormal pedal pulses, cool limbs, femoral bruit and prolonged venous filling time | Revascularization, antiplatelet medications, management of risk factors |
| Neuropathic | Most common cause of foot ulcers, usually from diabetes mellitus | Trauma, prolonged pressure | Usually occurs on plantar aspect of feet in patients with diabetes, neurologic disorders, or Hansen disease | Off-loading of pressure, topical growth factors; tissue-engineered skin |
| Pressure | Usually occurs in patients with limited mobility | Tissue ischemia and necrosis secondary to prolonged pressure | Located over bony prominences; risk factors include excessive moisture and altered mental status | Off-loading of pressure; reduction of excessive moisture, sheer, and friction; adequate nutrition |
Adapted with permission from de Araujo T, Valencia I, Federman DG, Kirsner RS. Managing the patient with venous ulcers. Ann Intern Med. 2003;138(4):327.
Venous stasis commonly presents as a dull ache or pain in the lower extremities, swelling that subsides with elevation, eczematous changes of the surrounding skin, and varicose veins.2 Venous ulcers often occur over bony prominences, particularly the gaiter area (over the medial malleolus). The recurrence of an ulcer in the same area is highly suggestive of venous ulcer.
On physical examination, venous ulcers are generally irregular and shallow (Figure 1). Granulation tissue and fibrin are often present in the ulcer base. Other findings include lower extremity varicosities; edema; venous dermatitis associated with hyperpigmentation and hemosiderosis or hemoglobin deposition in the skin; and lipodermatosclerosis associated with thickening and fibrosis of normal adipose tissue under skin.
Figure 1.
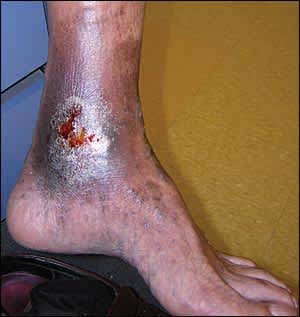
Venous leg ulcer. Wounds are generally irregular and shallow.
Reprinted with permission from Thomas Jefferson University Clinical Image Database.
A clinical severity score based on the CEAP (clinical, etiology, anatomy, and pathophysiology) classification system can guide the assessment of chronic venous disorders. The highest CEAP severity score is applied to patients with ulcers that are active, chronic (greater than three months' duration, and especially greater than 12 months' duration), and large (larger than 6 cm in diameter).19,20 Poor prognostic factors for venous ulcers include large size and prolonged duration.21,22
Treatment
Treatment options for venous ulcers include conservative management, mechanical treatment, medications, and surgical options (Table 2).1,2,7,10,19,22–44 In general, the goals of treatment are to reduce edema, improve ulcer healing, and prevent recurrence. Although numerous treatment methods are available, they have variable effectiveness and limited data to support their use.
Table 2. Treatment Options for Venous Ulcers
| Treatment options | Comments | References |
|---|---|---|
| Conservative management | ||
| Compression therapy (inelastic, elastic, intermittent pneumatic) | Standard of care; proven benefit (benefit of intermittent pneumatic therapy is less clear); associated with decreased rate of ulcer recurrence | 2, 7, 10, 22–26 |
| Leg elevation | Standard of care when used with compression therapy; minimizes edema; recommended for 30 minutes, three or four times a day | 27 |
| Dressings | No one type of dressing is superior | 28, 29 |
| Mechanical treatment | ||
| Topical negative pressure (vacuum-assisted closure) | No robust evidence regarding its use for venous ulcers | 30 |
| Medications | ||
| Pentoxifylline (Trental) | Effective when used with compression therapy; may be useful as monotherapy | 31 |
| Aspirin | Effective when used with compression therapy; dosage of 300 mg once per day | 32 |
| Iloprost | Intravenous administration (not available in the United States) may be beneficial, but data are insufficient to recommend its use; high cost limits use | 33 |
| Oral zinc | Not shown to be beneficial | 34 |
| Antibiotics/antiseptics | Oral antibiotic treatment is warranted in cases of suspected cellulitis; routine use of systemic antibiotics provides no healing benefit; benefit of adding the topical antiseptic cadexomer iodine (not available in the United States) is unclear | 1 |
| Hyperbaric oxygen therapy | No proven benefit | 35 |
| Surgical management | ||
| Debridement | More studies needed to establish benefit | 36–40 |
| Human skin grafting | More studies needed to establish benefit | 41 |
| Artificial skin (human skin equivalent) | May be beneficial when used with compression therapy; concern about infection transmission | 42 |
| Surgery for venous insufficiency | May be beneficial for severe or refractory cases; associated with decreased rate of ulcer recurrence | 19, 35, 40, 42–44 |
Information from references 1, 2, 7, 10, 19, and 22 through 44.
Conservative Management
COMPRESSION THERAPY
Compression therapy is the standard of care for venous ulcers and chronic venous insufficiency.23,45 A recent Cochrane review found that venous ulcers heal more quickly with compression therapy than without.45 Methods include inelastic, elastic, and intermittent pneumatic compression. Compression therapy reduces edema, improves venous reflux, enhances healing of ulcers, and reduces pain.23 Success rates range from 30 to 60 percent at 24 weeks, and 70 to 85 percent after one year.22 After an ulcer has healed, lifelong maintenance of compression therapy may reduce the risk of recurrence.12,24,25 However, adherence to the therapy may be limited by pain; drainage; application difficulty; and physical limitations, including obesity and contact dermatitis.19 Contraindications to compression therapy include clinically significant arterial disease and uncompensated heart failure.
Inelastic. Inelastic compression therapy provides high working pressure during ambulation and muscle contraction, but no resting pressure. The most common method of inelastic compression therapy is the Unna boot, a zinc oxide–impregnated, moist bandage that hardens after application. The Unna boot improves healing rates compared with placebo or hydroactive dressings.22,26 However, a 2009 Cochrane review found that adding a component of elastic compression therapy is more effective than inelastic compression therapy alone.45 Also, because of its inelasticity, the Unna boot does not conform to changes in leg size and may be uncomfortable to wear. In addition, the Unna boot may lead to a foul smell from the accumulation of exudate from the ulcer, requiring frequent reapplications.2
Elastic. Unlike the Unna boot, elastic compression therapy methods conform to changes in leg size and sustain compression during both rest and activity. Stockings or bandages can be used; however, elastic wraps (e.g., Ace wraps) are not recommended because they do not provide enough pressure.45 Compression stockings are graded, with the greatest pressure at the ankle and gradually decreasing pressure toward the knee and thigh (pressure should be at least 20 to 30 mm Hg, and preferably 30 to 44 mm Hg). Compression stockings are removed at night, and should be replaced every six months because they lose pressure with regular washing.2
Elastic bandages (e.g., Profore) are alternatives to compression stockings. A recent meta-analysis showed that elastic compression therapy is more effective than inelastic therapy.46 In addition, high compression has been proven more effective than low compression, and multilayer bandages are more effective than single layer.23,45,47 The disadvantage of multilayer compression bandages is that they require skilled application in the physician's office one or two times per week, depending on drainage.
Intermittent Pneumatic Compression. Intermittent pneumatic compression therapy comprises a pump that delivers air to inflatable and deflatable sleeves that embrace extremities, providing intermittent compression.7,23 The benefits of intermittent pneumatic compression are less clear than that of standard continuous compression. It also is expensive and requires immobilization of the patient; therefore, intermittent pneumatic compression is generally reserved for bedridden patients who cannot tolerate continuous compression therapy.24,40
LEG ELEVATION
Leg elevation when used in combination with compression therapy is also considered standard of care. Leg elevation requires raising lower extremities above the level of the heart, with the aim of reducing edema, improving microcirculation and oxygen delivery, and hastening ulcer healing. In one small study, leg elevation increased the laser Doppler flux (i.e., flow within veins) by 45 percent.27 Although leg elevation is most effective if performed for 30 minutes, three or four times per day, this duration of treatment may be difficult for patients to follow in real-world settings.
DRESSINGS
Dressings are often used under compression bandages to promote faster healing and prevent adherence of the bandage to the ulcer. A wide range of dressings are available, including hydrocolloids (e.g., Duoderm), foams, hydrogels, pastes, and simple nonadherent dressings.14,28 A meta-analysis of 42 randomized controlled trials (RCTs) with a total of more than 1,000 patients showed no significant difference among dressing types.29 Furthermore, the more expensive hydrocolloid dressings were not shown to have a healing benefit over the lower-cost simple nonadherent dressings. Without clear evidence to support the use of one dressing over another, the choice of dressings for venous ulcers can be guided by cost, ease of application, and patient and physician preference.29
Mechanical Treatment
Topical negative pressure, also called vacuum-assisted closure, has been shown to help reduce wound depth and volume compared with a hydrocolloid gel and gauze regimen for wounds of any etiology.30 However, clinically meaningful outcomes, such as healing time, have not yet been adequately studied. There is currently insufficient high-quality data to support the use of topical negative pressure for venous ulcers.30 In addition, the therapy generally has not been used in clinical practice because of the challenge in administering both topical negative pressure and a compression dressing on the affected leg.
Medications
PENTOXIFYLLINE
Pentoxifylline (Trental) is an inhibitor of platelet aggregation, which reduces blood viscosity and, in turn, improves microcirculation. Pentoxifylline (400 mg three times per day) has been shown to be an effective adjunctive treatment for venous ulcers when added to compression therapy.31,40 Pentoxifylline may also be useful as monotherapy in patients who are unable to tolerate compression bandaging.31 The most common adverse effects are gastrointestinal (e.g., nausea, vomiting, diarrhea, heartburn, loss of appetite). Despite a number of studies to support its effectiveness as adjunctive therapy, and possibly as monotherapy, the cost-effectiveness of pentoxifylline has not been established.
ASPIRIN
Like pentoxifylline therapy, aspirin (300 mg per day) combined with compression therapy has been shown to increase ulcer healing time and reduce ulcer size, compared with compression therapy alone.32 In general, adding aspirin therapy to compression bandages is recommended in the treatment of venous ulcers as long as there are no contraindications to its use.
ILOPROST
The synthetic prostacyclin iloprost is a vasodilator that inhibits platelet aggregation. In one study, intravenous iloprost (not available in the United States) used with elastic compression therapy significantly reduced healing time of venous ulcers compared with placebo.33 However, the medication is very costly and there are insufficient data to recommend its use.40
ORAL ZINC
Zinc is a trace metal with potential anti-inflammatory effects. Oral zinc therapy has been shown to decrease healing time in patients with pilonidal sinus. However, in a recent meta-analysis of six small studies, oral zinc had no beneficial effect in the treatment of venous ulcers.34
ANTIBIOTICS/ANTISEPTICS
Bacterial colonization and superimposed bacterial infections are common in venous ulcers and contribute to poor wound healing. However, a recent Cochrane review of 22 RCTs of systemic and topical antibiotics and antiseptics for venous ulcer treatment found no evidence that routine use of oral antibiotics improves healing rates.1 Studies comparing topical antibiotics and antiseptics, such as povidone-iodine solution (Betadine), peroxide-based preparations, ethacridine lactate (not available in the United States), and mupirocin (Bactroban), have found some evidence to support the use of the topical antiseptic agent cadexomer iodine (not available in the United States; a pooled estimate from two trials suggests an increased healing rate at four to six weeks compared with placebo).1 More high-quality data are needed to better evaluate the effectiveness of topical preparations, however.1
Oral antibiotics are recommended to treat venous ulcers only in cases of suspected cellulitis. Suspected osteomyelitis warrants an evaluation for arterial disease and consideration of intravenous antibiotics to treat the underlying infection.
HYPERBARIC OXYGEN THERAPY
Hyperbaric oxygen therapy has also been proposed as an adjunctive therapy for chronic wound healing because of potential anti-inflammatory and antibacterial effects, and its benefits in healing diabetic foot ulcers. However, data to support its use for venous ulcers are limited.35
Surgical Management
Overall, acute ulcers (duration of three months or less) have a 71 to 80 percent chance of healing, whereas chronic ulcers have only a 22 percent chance of healing after six months of treatment.7 Given the poor healing rates associated with chronic ulcers, surgical evaluation and management should be considered in patients with venous ulcers that are refractory to conservative therapies.48
DEBRIDEMENT
Removal of necrotic tissue and bacterial burden through debridement has long been used in wound care to enhance healing. Debridement may be sharp (e.g., using curette or scissors), enzymatic, mechanical, biologic (i.e., using larvae), or autolytic. However, there are few high-quality studies that directly evaluate the effect of debridement versus no debridement or the superiority of one type of debridement on the rate of venous ulcer healing.36–40 In addition, most wounds with significant necrotic tissue should be evaluated for arterial insufficiency because purely venous ulcers rarely need much debridement.
SKIN GRAFTING
Human skin grafting may be used for patients with large or refractory venous ulcers. It is performed with autograft (skin or cells taken from another site on the same patient), allograft (skin or cells taken from another person), or artificial skin (human skin equivalent).41,42,49 However, skin grafting generally is not effective if there is persistent edema, which is common with venous insufficiency, and the underlying venous disease is not addressed.40 A recent Cochrane review found few high-quality studies to support the use of human skin grafting for the treatment of venous ulcers.41
SURGERY FOR VENOUS INSUFFICIENCY
The role of surgery is to reduce venous reflux, hasten healing, and prevent ulcer recurrence. Surgical options for treatment of venous insufficiency include ablation of the saphenous vein; interruption of the perforating veins with subfascial endoscopic surgery; treatment of iliac vein obstruction with stenting; and removal of incompetent superficial veins with phlebectomy, stripping, sclerotherapy, or laser therapy.19,35,40
In one study, ablative superficial venous surgery reduced the rate of venous ulcer recurrence at 12 months by more than one half, compared with compression therapy alone.42 In another study, surgical management led to an ulcer healing rate of 88 percent, with only a 13 percent recurrence rate over 10 months.43 There is no evidence demonstrating the superiority of surgery over medical management; however, evaluation for possible surgical intervention should occur early.44

