Pharmacologic treatment of hyperlipidemia in conjunction with therapeutic lifestyle changes can be used for both primary and secondary prevention of cardiovascular disease. Statins have the most convincing data for primary prevention, especially for higher risk patients. Therefore, risk stratification is essential. Statin therapy is also recommended for secondary prevention in all patients with known cardiovascular disease or the risk equivalent. High-dose statins should be initiated in patients with acute coronary syndrome. Omega-3 fatty acids may be a good alternative after myocardial infarction for patients who cannot tolerate statins. Fibrates and niacin have not been shown to reduce all-cause mortality in secondary prevention, but may be useful adjuncts when statins alone cannot adequately control lipid levels. Other cholesterol-lowering medications used for primary or secondary prevention of cardiovascular disease have not been shown to consistently improve patient-oriented outcomes. There is good evidence for using statins in the secondary prevention of stroke and peripheral arterial disease.
Cardiovascular disease (CVD) is the leading cause of mortality in the United States, accounting for 33.6 percent of all deaths in 2007.1 Hyperlipidemia is a common risk factor for CVD, with 53.4 percent of adults in the United States having abnormal cholesterol values and 32 percent having elevated low-density lipoprotein (LDL) cholesterol levels.1
SORT: KEY RECOMMENDATIONS FOR PRACTICE
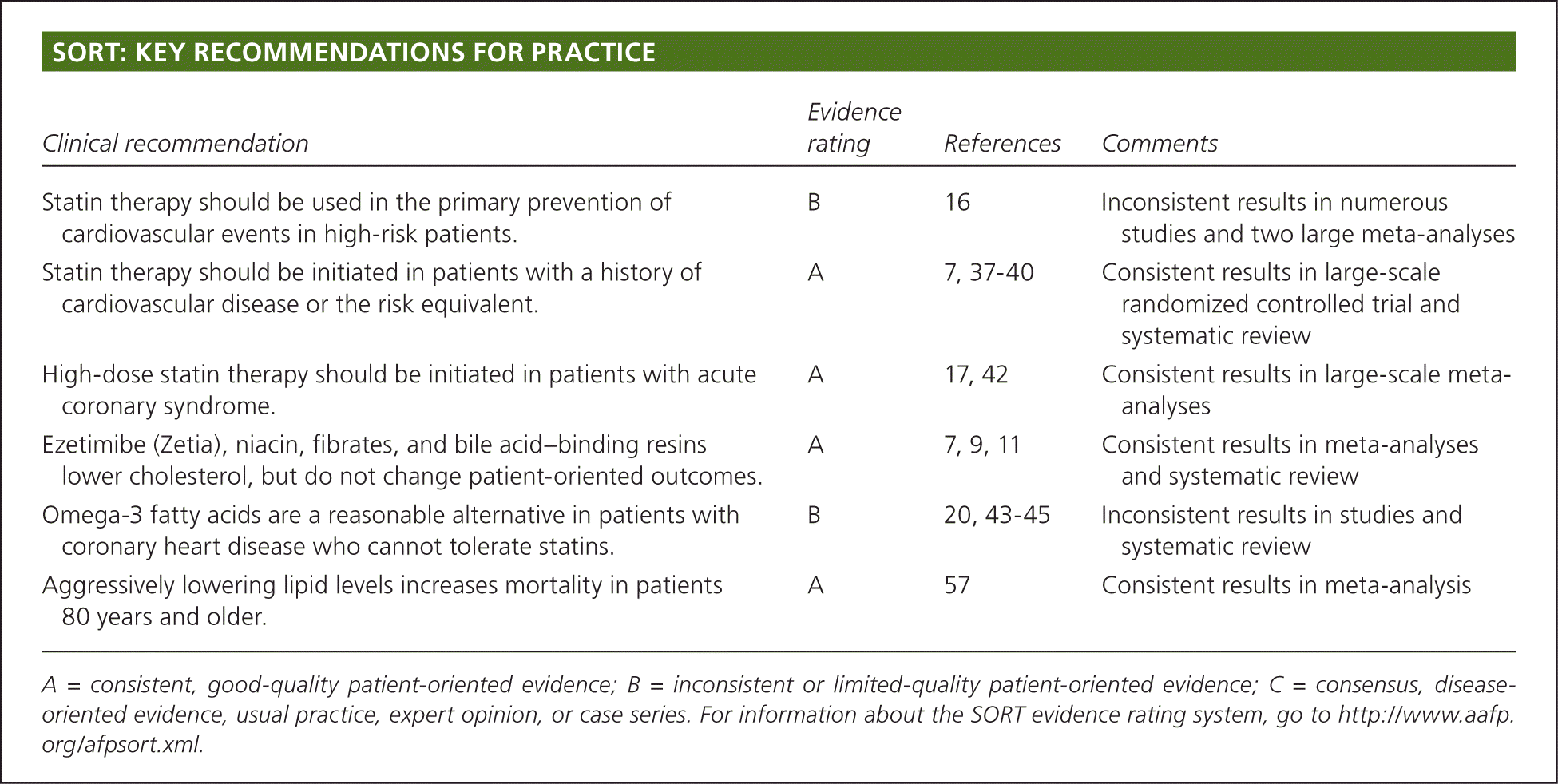
| Clinical recommendation | Evidence rating | References | Comments |
|---|---|---|---|
| Statin therapy should be used in the primary prevention of cardiovascular events in high-risk patients. | B | 16 | Inconsistent results in numerous studies and two large meta-analyses |
| Statin therapy should be initiated in patients with a history of cardiovascular disease or the risk equivalent. | A | 7, 37–40 | Consistent results in large-scale randomized controlled trial and systematic review |
| High-dose statin therapy should be initiated in patients with acute coronary syndrome. | A | 17, 42 | Consistent results in large-scale meta-analyses |
| Ezetimibe (Zetia), niacin, fibrates, and bile acid–binding resins lower cholesterol, but do not change patient-oriented outcomes. | A | 7, 9, 11 | Consistent results in meta-analyses and systematic review |
| Omega-3 fatty acids are a reasonable alternative in patients with coronary heart disease who cannot tolerate statins. | B | 20, 43–45 | Inconsistent results in studies and systematic review |
| Aggressively lowering lipid levels increases mortality in patients 80 years and older. | A | 57 | Consistent results in meta-analysis |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
National Practice Guidelines
U.S., U.K., and Canadian guidelines are available to help physicians manage hyperlipidemia (Table 1).2–4 These guidelines agree that therapeutic lifestyle changes are the mainstay of hyperlipidemia management, and that LDL cholesterol should be the primary target of therapy. Treatment of hyperlipidemia improves outcomes for patients with known coronary heart disease (CHD) or the risk equivalent, and for high-risk patients (i.e., those with a 10-year CHD risk of greater than 20 percent) without known CHD or the risk equivalent.
Table 1. Summary of Major Hyperlipidemia Guidelines
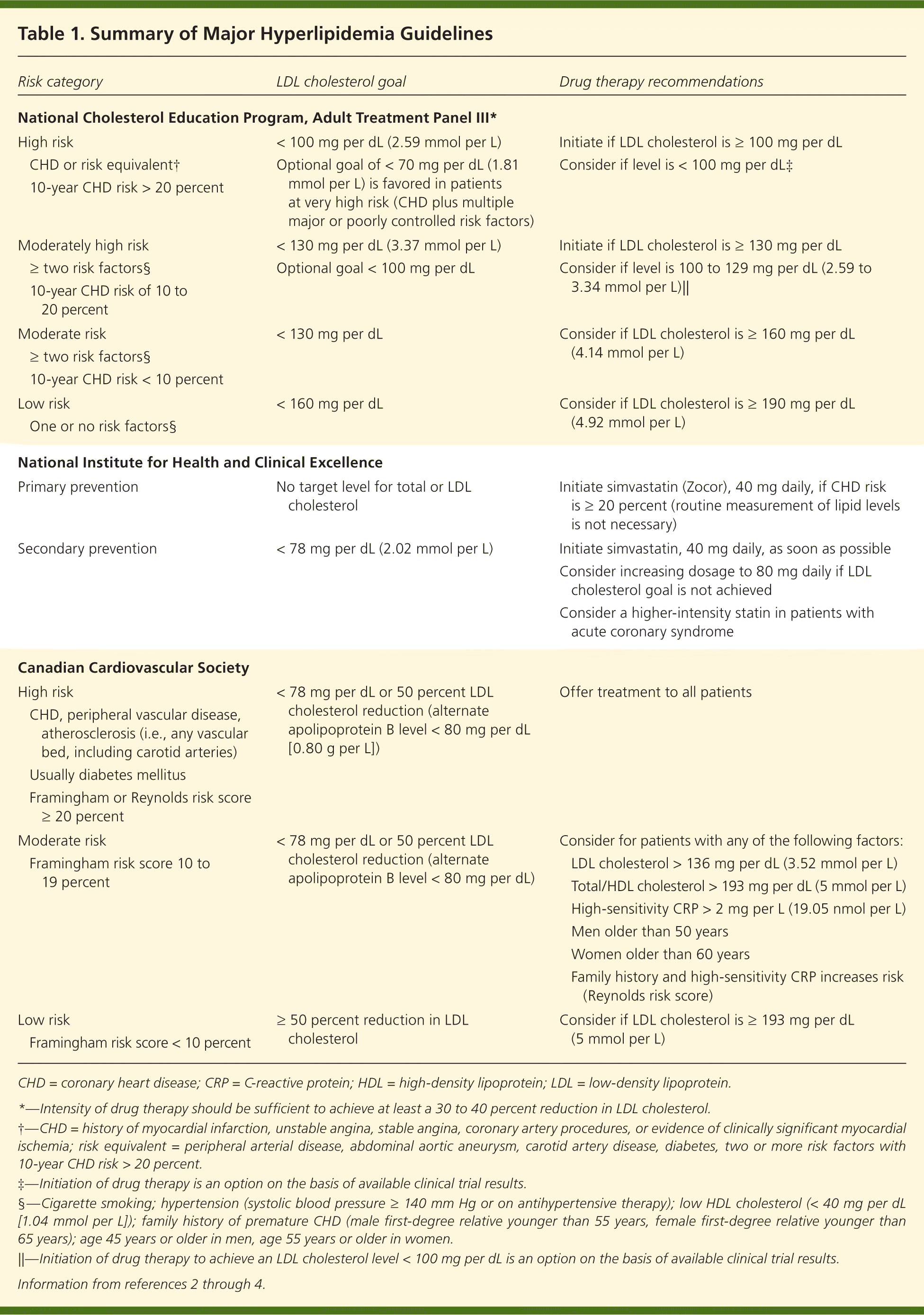
| Risk category | LDL cholesterol goal | Drug therapy recommendations |
|---|---|---|
| National Cholesterol Education Program, Adult Treatment Panel III* | ||
High risk
| < 100 mg per dL (2.59 mmol per L) |
|
| Optional goal of < 70 mg per dL (1.81 mmol per L) is favored in patients at very high risk (CHD plus multiple major or poorly controlled risk factors) | ||
Moderately high risk
| < 130 mg per dL (3.37 mmol per L) |
|
| Optional goal < 100 mg per dL | ||
Moderate risk
| < 130 mg per dL |
|
Low risk
| < 160 mg per dL |
|
| National Institute for Health and Clinical Excellence | ||
| Primary prevention | No target level for total or LDL cholesterol |
|
| Secondary prevention | < 78 mg per dL (2.02 mmol per L) |
|
| Canadian Cardiovascular Society | ||
High risk
| < 78 mg per dL or 50 percent LDL cholesterol reduction (alternate apolipoprotein B level < 80 mg per dL [0.80 g per L]) |
|
Moderate risk
| < 78 mg per dL or 50 percent LDL cholesterol reduction (alternate apolipoprotein B level < 80 mg per dL) |
|
Low risk
| ≥ 50 percent reduction in LDL cholesterol |
|
CHD = coronary heart disease; CRP = C-reactive protein; HDL = high-density lipoprotein; LDL = low-density lipoprotein.
*—Intensity of drug therapy should be sufficient to achieve at least a 30 to 40 percent reduction in LDL cholesterol.
†—CHD = history of myocardial infarction, unstable angina, stable angina, coronary artery procedures, or evidence of clinically significant myocardial ischemia; risk equivalent = peripheral arterial disease, abdominal aortic aneurysm, carotid artery disease, diabetes, two or more risk factors with 10-year CHD risk > 20 percent.
‡—Initiation of drug therapy is an option on the basis of available clinical trial results.
§— [ corrected] Cigarette smoking; hypertension (systolic blood pressure ≥ 140 mm Hg or on antihypertensive therapy); low HDL cholesterol (< 40 mg per dL [1.04 mmol per L]); family history of premature CHD (male first-degree relative younger than 55 years, female first-degree relative younger than 65 years); age 45 years or older in men, age 55 years or older in women.
∥—Initiation of drug therapy to achieve an LDL cholesterol level < 100 mg per dL is an option on the basis of available clinical trial results.
The U.S. National Cholesterol Education Program, Adult Treatment Panel (ATP) III guidelines advocate for a treat-to-target approach and are more aggressive than other guidelines.2 The U.K. National Institute for Health and Clinical Excellence (NICE) guidelines recommend offering a fixed-dose statin based on CHD risk stratification, and recommend against checking cholesterol levels after a patient starts statin therapy.3 For secondary prevention, NICE recommends a treat-to-target therapy that is less aggressive than that recommended by the ATP III. The ATP III recommendations are based on the assumption that achieving the mean LDL cholesterol level observed in clinical trials will produce similar results in practice, but no clinical trial has assessed a treat-to-target strategy.
In theory, when comparing the treat-to-target and fixed-dose approaches, essentially the same number of patients would be treated with statin therapy.5 A theoretical modeling study comparing approaches for primary CHD risk reduction suggests that a fixed-dose approach based on individual risk would prevent more events and save more quality-adjusted life-years.5 Although neither approach is clearly superior at this time, it is reasonable for physicians to engage patients in a shared, informed decision-making process and to determine treatment goals based on the patient's overall CHD risk, risk reduction that could be expected with treatment, and values and preferences.
Primary Prevention
Primary prevention of CVD consists of treating patients with hyperlipidemia before clinical CHD manifests (e.g., myocardial infarction). The evidence supporting treatment of hyperlipidemia for primary prevention is inconsistent. Patients with the highest baseline risk are most likely to benefit. Medications should be chosen based on a favorable balance between the likelihood of benefits (e.g., patient-oriented outcomes, mortality, CVD events, functional status, quality of life) and harm (adverse effects), as well as cost.6 Table 2 summarizes medications used to treat lipid disorders.7–20
Table 2. Characteristics of Medications to Treat Lipid Disorders
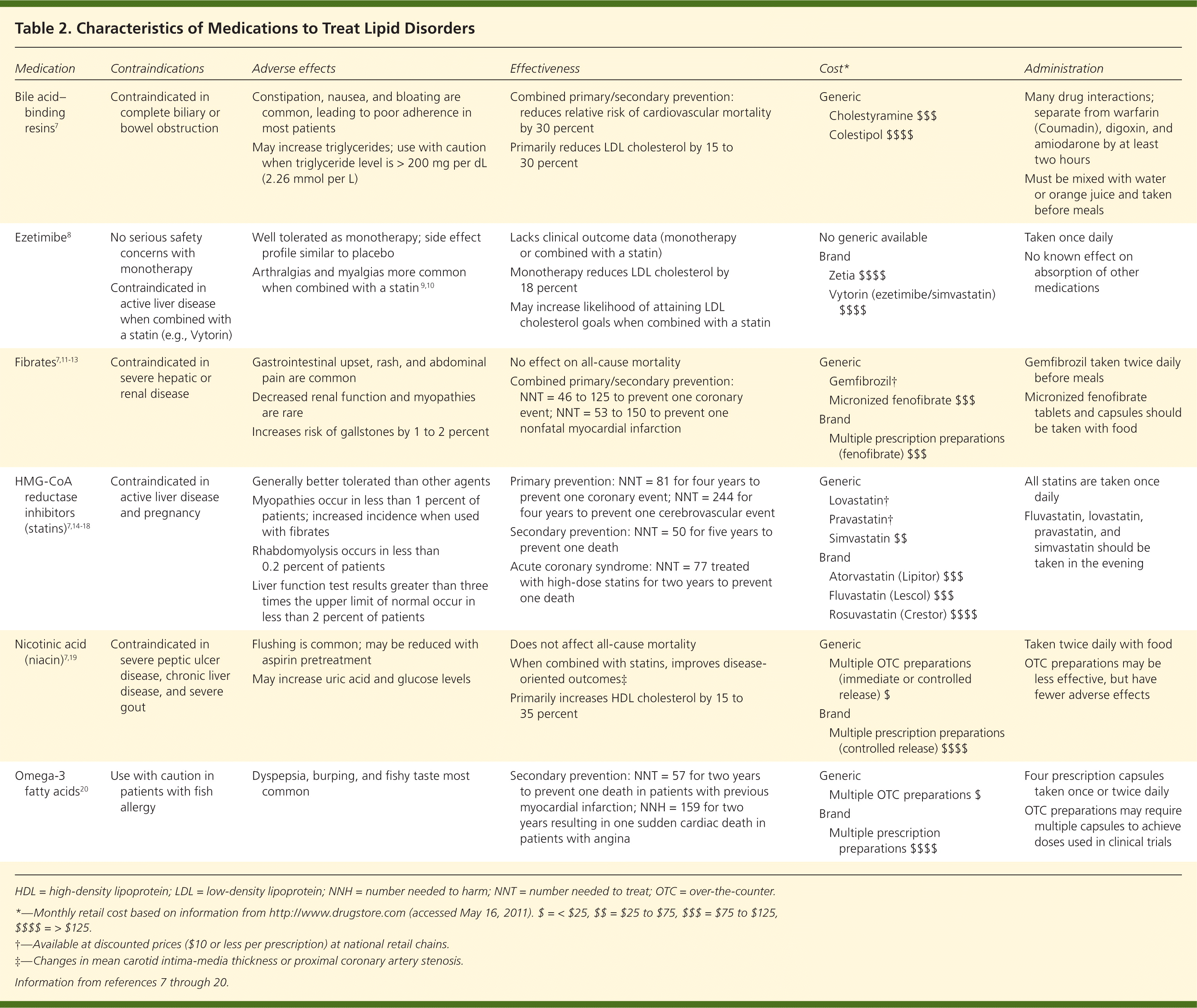
| Medication | Contraindications | Adverse effects | Effectiveness | Cost* | Administration |
|---|---|---|---|---|---|
| Bile acid–binding resins7 | Contraindicated in complete biliary or bowel obstruction |
|
|
|
|
| Ezetimibe8 | No serious safety concerns with monotherapy |
|
|
| |
| Contraindicated in active liver disease when combined with a statin (e.g., Vytorin) | |||||
| Fibrates7,11–13 | Contraindicated in severe hepatic or renal disease |
|
|
|
|
| HMG-CoA reductase inhibitors (statins)7,14–18 | Contraindicated in active liver disease and pregnancy |
|
|
| |
| Nicotinic acid (niacin)7,19 | Contraindicated in severe peptic ulcer disease, chronic liver disease, and severe gout |
|
|
|
|
| Omega-3 fatty acids20 | Use with caution in patients with fish allergy |
|
|
|
|
HDL = high-density lipoprotein; LDL = low-density lipoprotein; NNH = number needed to harm; NNT = number needed to treat; OTC = over-the-counter.
*—Monthly retail cost based on information from http://www.drugstore.com (accessed May 16, 2011). $ = < $25, $$ = $25 to $75, $$$ = $75 to $125, $$$$ = > $125.
†—Available at discounted prices ($10 or less per prescription) at national retail chains.
‡—Changes in mean carotid intima-media thickness or proximal coronary artery stenosis.
STATINS
Two large meta-analyses have evaluated statins for primary prevention of CVD.16,21 The first analysis pooled 10 trials and found that statins were superior to placebo for all-cause mortality (number needed to treat [NNT] = 172), major coronary events (NNT = 81), and major cerebrovascular events (NNT = 244).16 However, the second meta-analysis was conducted because the first did not exclusively evaluate primary prevention, including trials with up to 20 percent of patients with CHD or the equivalent. The second analysis, which excluded such groups and included newer studies, showed no difference between statins and placebo for all-cause mortality.21
Studies have evaluated statin therapy based on C-reactive protein level. The JUPITER (Justification for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin) study included participants with LDL cholesterol levels of less than 130 mg per dL (3.37 mmol per L) and elevated high-sensitivity C-reactive protein levels who received rosuvastatin (Crestor) or placebo for two years.22 Compared with placebo, statin therapy reduced the risk of the primary composite end point (myocardial infarction, stroke, hospitalization for unstable angina, arterial revascularization, and cardiovascular death; NNT = 84). However, the benefit of statin therapy on individual outcomes varied widely, with NNT ranging from 150 for arterial revascularization to 244 for myocardial infarction.
Critics argue against using the JUPITER study results to justify broadly expanding statin use in primary prevention.23–25 The study ended early and excluded non-adherent patients, which could exaggerate the positive effects of treatment without allowing sufficient time for adverse effects to develop.23,24 Furthermore, if “hard cardiac” clinical outcomes are taken out, the study included only 240 events.23 The JUPITER study also failed to compare rosuvastatin with other interventions, such as lifestyle changes, aspirin, or less-expensive statins, and did not evaluate cost-effectiveness. Several authors had financial ties to the sponsor, and one held the patent to the high-sensitivity C-reactive protein assay used to determine study eligibility.
Treatment with statins as primary prevention should be offered to those at the highest risk of developing CHD based on validated prediction models, such as the Reynolds risk score (http://www.reynoldsriskscore.org/) or the Framingham risk assessment tool (http://hp2010.nhlbihin.net/atpiii/calculator.asp). No statin has been proven superior at preventing CHD and, at equivalent doses, all statins substantially reduce LDL cholesterol.26
NONSTATINS
Some patients will have low levels of high-density lipoprotein (HDL) cholesterol or high levels of triglycerides even after meeting their LDL cholesterol target. Evidence for treating these secondary lipid abnormalities with medications other than statins for primary prevention is not convincing.
For example, there is no evidence that fibrates have mortality benefit in primary prevention, and there is some evidence that they are harmful.11 None of the four trials that have compared fibrates with placebo in patients without a history of CHD showed that fibrates reduce overall mortality.27–30 In three of the trials, fibrates reduced the risk of coronary events compared with placebo (NNT = 125 with clofibrate [not available in the United States]; NNT = 72 with gemfibrozil [Lopid]; NNT = 8 with bezafibrate [not available in the United States]).27–29 Clofibrate was associated with an increased risk of overall mortality (number needed to harm [NNH] = 9 for 13.2 years).27 Combining fibrate with statin therapy does not appear to be beneficial.31
Niacin is the only drug consistently proven to raise HDL cholesterol levels32–34; however, there is no evidence that it reduces all-cause or cardiovascular mortality in primary prevention.7 In the JUPITER study, patients taking rosuvastatin had fewer CHD outcomes regardless of HDL cholesterol level.22 This implies that significantly lowering LDL cholesterol levels may be just as effective at protecting against CHD as artificially raising HDL cholesterol levels.22
When used in primary prevention, bile acid–binding resins (e.g., cholestyramine [Questran], colestipol [Colestid], colesevelam [Welchol]) reduce LDL cholesterol levels, but do not affect mortality.7 There are no studies of resin therapy to reduce CHD risk in patients at low risk (less than 0.6 percent annual risk) or at high risk (1.5 percent or greater annual risk) of a primary event. The one study evaluating resins in patients with moderate risk (0.6 to 1.4 percent annual risk) did not show a clear mortality benefit.35
Although omega-3 fatty acids were previously thought to reduce CHD risk, a 2004 systematic review concluded that it is unclear if dietary or supplemental omega-3 fatty acids reduce the risk of total mortality or cardiovascular events in persons at high risk of CVD or in the general population.36 However, there is also no evidence to support advising patients to stop consuming rich sources of omega-3 fats.
Secondary Prevention
STATINS
Statins are indicated in virtually all patients with a history of CHD, reducing the risk of all-cause mortality (NNT = 50 for five years) and cardiovascular mortality.7 Most patients with a CHD risk equivalent also benefit from statin therapy. The ATP III guidelines recommend initiating statins in patients with a history of CHD, and adjusting the intensity of therapy to achieve at least a 30 to 40 percent reduction in LDL cholesterol or an absolute LDL cholesterol level below 70 mg per dL (1.81 mmol per L) or 100 mg per dL (2.59 mmol per L).2 The Canadian guidelines recommend a similar treat-to-target approach.4 The NICE guidelines recommend simvastatin (Zocor), 40 mg, for all patients with clinical evidence of CHD and a higher-intensity statin for patients with acute coronary syndrome.3
Statins may benefit patients with CHD independent of baseline cholesterol levels or age.37–40 The effectiveness of statin therapy on reducing mortality, myocardial infarction, and stroke does not appear to differ among atorvastatin (Lipitor), pravastatin (Pravachol), and simvastatin.41 No study has directly compared equivalent dosages of two different statins for secondary prevention. The ideal starting dose in patients with CHD depends on the presence of acute coronary syndrome.
A meta-analysis including patients with recent acute coronary syndrome compared high-dose statin therapy (40 mg of simvastatin daily for one month followed by 80 mg daily, or 80 mg of atorvastatin daily) with moderate-dose therapy (placebo for four months followed by 20 mg of simvastatin daily, or 40 mg of pravastatin daily).17 High-dose statins reduced overall mortality (NNT = 77 for two years), primarily from a decrease in cardiovascular mortality (NNT = 112 for two years).
A second meta-analysis compared the effectiveness of high-dose statin therapy (80 mg of simvastatin daily) with lower-dose statin therapy (up to 20 mg of simvastatin daily) in patients with recent acute coronary syndrome or stable CHD.42 Higher-dose statin therapy reduced overall mortality (NNT = 91 for two years) in patients with acute coronary syndrome. High-dose statins did not reduce mortality in patients with stable CHD, but decreased adverse cardiovascular events by 1.9 percent over 4.7 years (NNT = 53). Based on these findings, it is reasonable to initiate lower doses of statins in patients with stable CHD and reserve initial high-dose statins for those with recent acute coronary syndrome.
NONSTATINS
A meta-analysis demonstrated that fibrates reduced the risk of subsequent coronary events (NNT = 35), but not overall mortality, compared with placebo.11 Combination statin-fibrate therapy has not been shown to improve all-cause mortality compared with statins alone.9 A statin/ezetimibe combination may increase the likelihood of attaining LDL cholesterol goals, but has no known effect on all-cause mortality.9
Niacin monotherapy for secondary prevention does not reduce the risk of overall mortality.7 When added to statin therapy in patients with CHD, niacin improves disease-oriented outcomes (e.g., changes in mean carotid intima-media thickness or proximal coronary artery stenosis) compared with ezetimibe (Zetia) and antioxidants.10,19
Individual trials have yielded conflicting evidence for omega-3 fatty acids in secondary risk reduction.43–45 The optimal quantity and type of omega-3 fatty acid are unclear.46 In a recent systematic review comparing omega-3 fatty acids with control diets or placebo in patients with CHD, omega-3 fatty acids were associated with a reduced risk of sudden cardiac death (NNT = 98), cardiac death (NNT = 66), and all-cause mortality (NNT = 57) in the subgroup with a history of CHD and myocardial infarction.20 In the baseline subgroup with only a history of CHD (e.g., angina), omega-3 fatty acids increased the risk of sudden cardiac death (NNH = 159) and did not alter the risk of cardiac death or all-cause mortality. Although their benefit is small, using omega-3 fatty acids may be a reasonable alternative after myocardial infarction in patients who cannot tolerate statin therapy.
Bile acid–binding resins have no statistically significant effect on overall mortality when used for secondary prevention (relative risk = 0.84; 95% confidence interval, 0.66 to 1.08).7
CEREBROVASCULAR DISEASE
A Cochrane review of statins for secondary prevention of cerebrovascular disease reported that statins reduced recurrent strokes (NNT = 76), but not all-cause mortality.47 Other lipid-lowering medications, including fibrates, increased the risk of subsequent strokes. In an acute setting, initiating statin therapy may reduce stroke severity and disability, with data supporting 40 to 80 mg of atorvastatin daily over 20 to 80 mg of simvastatin daily.48 However, initiating high-dose atorvastatin after ischemic stroke has been shown to increase the risk of hemorrhagic stroke (NNH = 106 over five years of treatment), while reducing recurrent ischemic strokes (NNT = 42 over five years of treatment) and not affecting overall mortality.49 Thus, initiating a moderate-dose statin is an option to reduce the risk of recurrent strokes.
PERIPHERAL ARTERIAL DISEASE
A Cochrane review concluded that statin use did not reduce mortality in patients with peripheral arterial disease.50 However, using statins in these patients increased maximal walking distance by 499 ft (152 m) and pain-free walking distance by 295 ft (90 m). Lipid therapy did not change ankle-brachial index scores. Statin therapy for patients with abdominal aortic aneurysms has not been shown to change the rate of expansion, but patients who are taking statins at the time of a rupture have a lower mortality rate (NNT = 3.5).51,52
Statin Therapy in Specific Populations
Women
All women with a history of CHD should be offered treatment with a statin. There are arguments for and against recommending statins in women to reduce primary CHD risk.53,54 Some believe that women may respond differently to statins than men. Women also have a different baseline risk than men and therefore may not derive the same benefit. A systematic review concluded that lipid-lowering therapy in women does not change overall mortality, but may reduce the risk of coronary events in secondary, but not primary, prevention.55
OLDER PERSONS
Clinical trials rarely include older patients, and extrapolating data from younger to older populations may be problematic. A meta-analysis found that in patients 62 to 85 years of age with CHD and hyperlipidemia, statins reduced overall mortality risk (NNT = 28), nonfatal myocardial infarction (NNT = 38), and stroke (NNT = 58) over five years.56 A meta-analysis of patients 80 years or older receiving lipid-lowering therapy found that low cholesterol levels were associated with an increase in overall mortality compared with high cholesterol levels.57 The decision to use statins for prevention of CHD in older patients should depend on individual baseline cardiovascular risk, expected degree of risk reduction, life expectancy, potential risks of therapy, and patient preference.
Data Sources: A PubMed search was completed in Clinical Queries using the key terms primary cardiovascular risk reduction, secondary cardiovascular risk reduction, hyperlipidemia, cholesterol, HMG CoA reductase inhibitors, fibric acid derivatives, bile-acid binding agents, ezetimibe, niacin, and omega-3 fatty acids. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Also searched were the Cochrane database, Dynamed, Clinical Evidence, Essential Evidence Plus, National Guideline Clearinghouse database, and U.S. Preventive Services Task Force. Search dates: June 3, 2010; September 10, 2010; and February 11, 2011.

