The prevalence of benign anorectal conditions in the primary care setting is high, although evidence of effective therapy is often lacking. In addition to recognizing common benign anorectal disorders, physicians must maintain a high index of suspicion for inflammatory and malignant disorders. Patients with red flags such as increased age, family history, persistent anorectal bleeding despite treatment, weight loss, or iron deficiency anemia should undergo colonoscopy. Pruritus ani, or perianal itching, is managed by treating the underlying cause, ensuring proper hygiene, and providing symptomatic relief with oral antihistamines, topical steroids, or topical capsaicin. Effective treatments for anal fissures include onabotulinumtoxinA, topical nitroglycerin, and topical calcium channel blockers. Symptomatic external hemorrhoids are managed with dietary modifications, topical steroids, and analgesics. Thrombosed hemorrhoids are best treated with hemorrhoidectomy if symptoms are present for less than 72 hours. Grades I through III internal hemorrhoids can be managed with rubber band ligation. For the treatment of grade III internal hemorrhoids, surgical hemorrhoidectomy has higher remission rates but increased pain and complication rates compared with rubber band ligation. Anorectal condylomas, or anogenital warts, are treated based on size and location, with office treatment consisting of topical trichloroacetic acid or podophyllin, cryotherapy, or laser treatment. Simple anorectal fistulas can be treated conservatively with sitz baths and analgesics, whereas complex or nonhealing fistulas may require surgery. Fecal impaction may be treated with polyethylene glycol, enemas, or manual disimpaction. Fecal incontinence is generally treated with loperamide and biofeedback. Surgical intervention is reserved for anal sphincter injury.
Anorectal conditions are a common presentation in the primary care setting. A history and physical examination usually will determine the etiology. Physical examination includes visual inspection, digital rectal examination, and anoscopy. Proper use of an anoscope has been described previously in American Family Physician.1 For any anorectal condition, malignancy must be considered and excluded as indicated from the history and physical examination. Anal cancer can coexist with benign anal conditions. Red flags (i.e., older age, weight loss, iron deficiency anemia, family history of inflammatory bowel disease or colorectal cancer, and persistent anorectal bleeding despite treatment of a suspected benign condition) warrant evaluation with colonoscopy. The U.S. Preventive Services Task Force recommendations on screening for colorectal cancer are available at http://www.uspreventiveservicestaskforce.org/uspstf/uspscolo.htm.2 This article reviews the presentation and physical examination findings of benign anorectal disorders and available treatments. A summary of conditions, history and physical examination findings, and treatment options is provided in Table 1.3–37
SORT: KEY RECOMMENDATIONS FOR PRACTICE
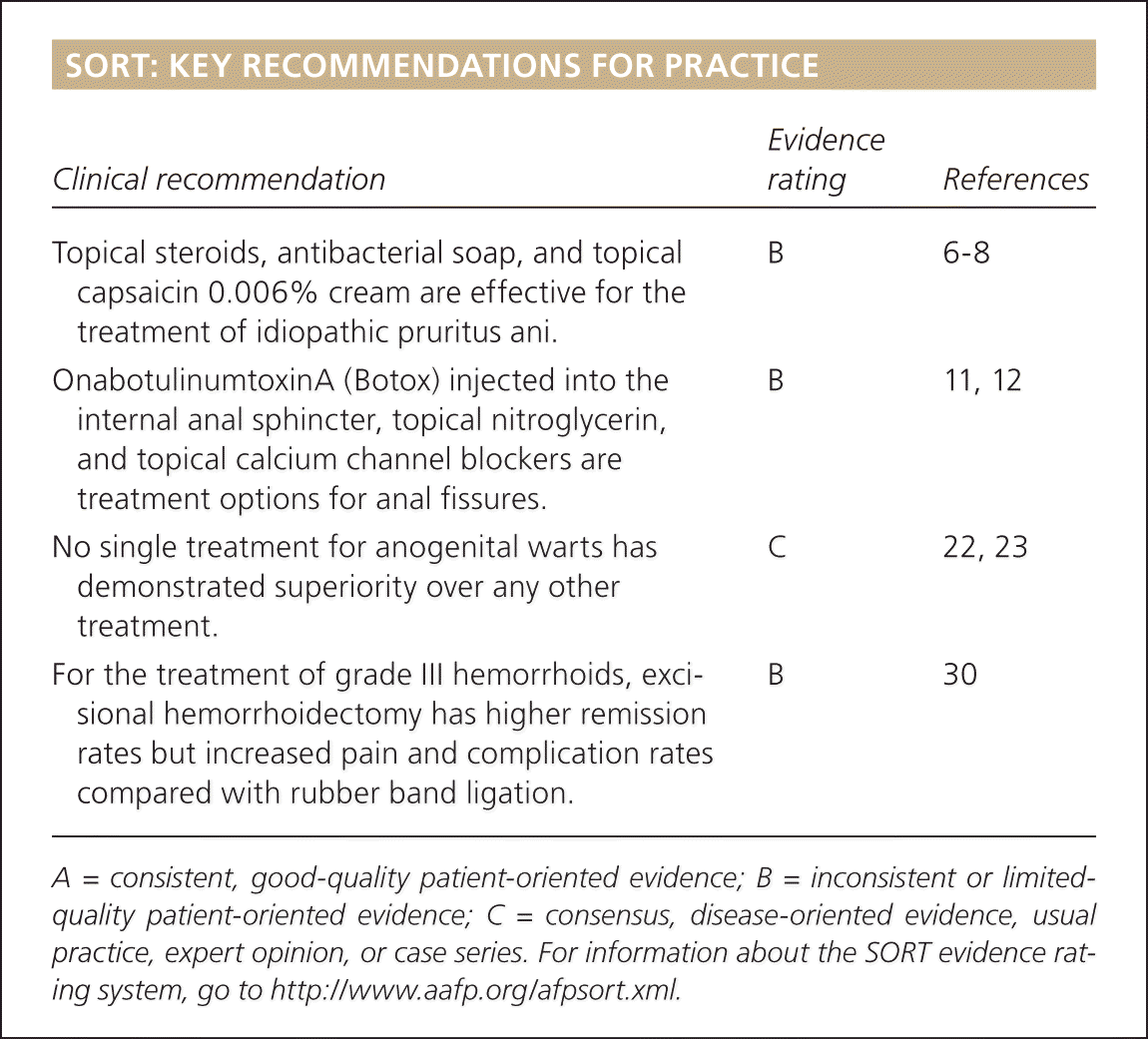
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Topical steroids, antibacterial soap, and topical capsaicin 0.006% cream are effective for the treatment of idiopathic pruritus ani. | B | 6–8 |
| OnabotulinumtoxinA (Botox) injected into the internal anal sphincter, topical nitroglycerin, and topical calcium channel blockers are treatment options for anal fissures. | B | 11, 12 |
| No single treatment for anogenital warts has demonstrated superiority over any other treatment. | C | 22, 23 |
| For the treatment of grade III hemorrhoids, excisional hemorrhoidectomy has higher remission rates but increased pain and complication rates compared with rubber band ligation. | B | 30 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Table 1. Common Anorectal Conditions
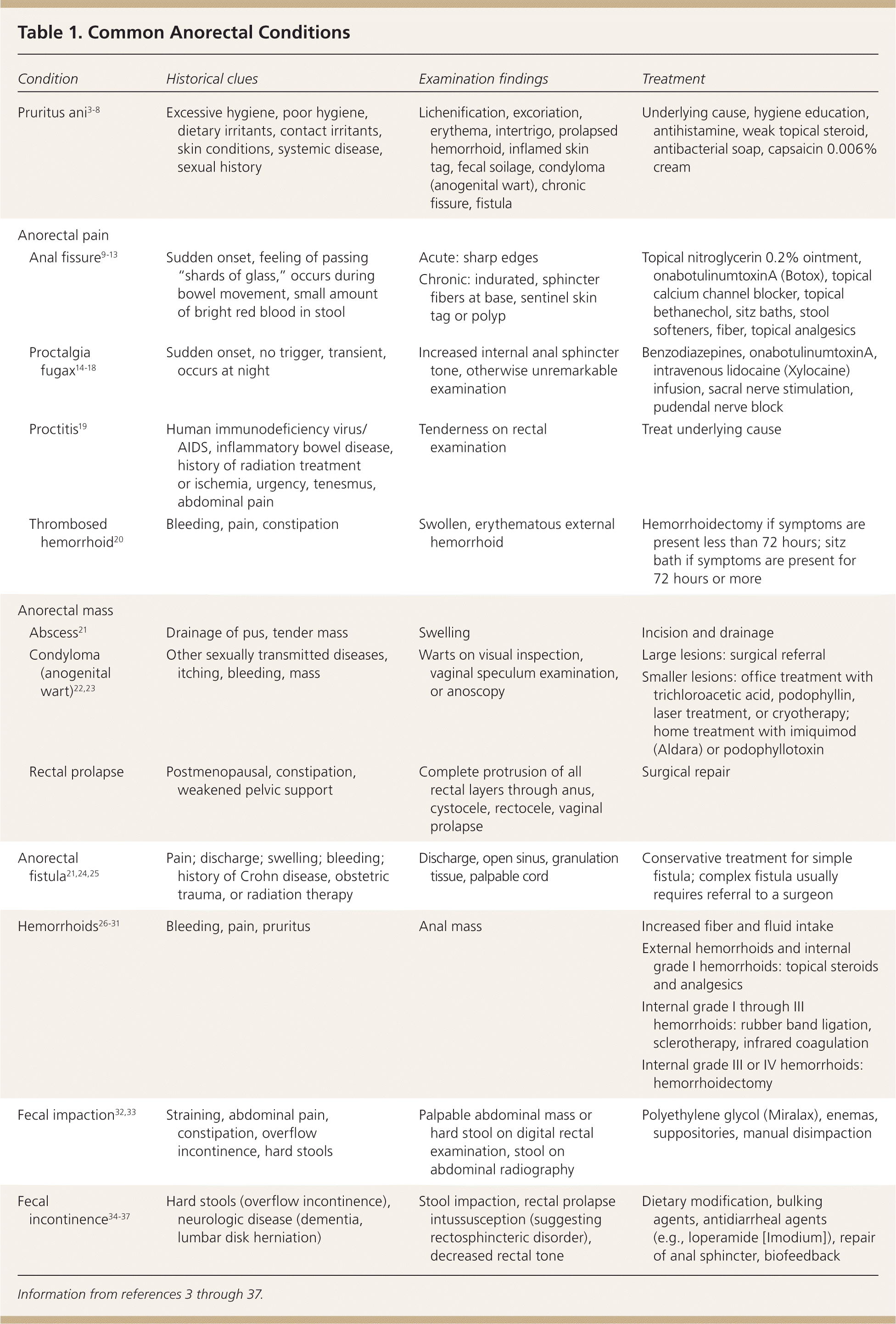
| Condition | Historical clues | Examination findings | Treatment | |
|---|---|---|---|---|
| Pruritus ani3-8 | Excessive hygiene, poor hygiene, dietary irritants, contact irritants, skin conditions, systemic disease, sexual history |
|
| |
| Anorectal pain | ||||
| Anal fissure9-13 | Sudden onset, feeling of passing “shards of glass,” occurs during bowel movement, small amount of bright red blood in stool |
|
| |
| Proctalgia fugax14–18 | Sudden onset, no trigger, transient, occurs at night |
|
| |
| Proctitis19 | Human immunodeficiency virus/AIDS, inflammatory bowel disease, history of radiation treatment or ischemia, urgency, tenesmus, abdominal pain |
|
| |
| Thrombosed hemorrhoid20 | Bleeding, pain, constipation |
|
| |
| Anorectal mass | ||||
| Abscess21 | Drainage of pus, tender mass |
|
| |
| Condyloma (anogenital wart)22,23 | Other sexually transmitted diseases, itching, bleeding, mass |
|
| |
| Rectal prolapse | Postmenopausal, constipation, weakened pelvic support |
|
| |
| Anorectal fistula21,24,25 | Pain; discharge; swelling; bleeding; history of Crohn disease, obstetric trauma, or radiation therapy |
|
| |
| Hemorrhoids26-31 | Bleeding, pain, pruritus |
|
| |
| Fecal impaction32,33 | Straining, abdominal pain, constipation, overflow incontinence, hard stools |
|
| |
| Fecal incontinence34-37 | Hard stools (overflow incontinence), neurologic disease (dementia, lumbar disk herniation) |
|
| |
Pruritus Ani
Pruritus ani, or perianal itching, affects 1 to 5 percent of the population,3 and is the second most common anorectal condition after hemorrhoids.4 Most patients self-treat for 12 months before consulting a physician.5 The etiologies of pruritus ani are listed in Table 2.1,5,38 Pinworm infection is prevalent among young children and institutionalized adults.38 Lumbosacral radiculopathy can manifest as idiopathic pruritus ani and should be considered in older patients with chronic back pain.39 Regardless of the cause, a self-propagating itch-scratch cycle results. The scratching exacerbates the inflammation, causing an irresistible urge to scratch even more. The chronic nature is demonstrated by lichenification of the perianal skin.
Table 2. Causes of Pruritus Ani

| Dermatologic conditions |
| Atopic dermatitis |
| Contact dermatitis |
| Hidradenitis suppurativa |
| Lichen planus |
| Lichen sclerosis |
| Psoriasis |
| Seborrheic dermatitis |
| Dietary irritants |
| Beer |
| Caffeine |
| Chili peppers |
| Citrus |
| Milk |
| Tomatoes |
| Fecal soilage |
| Anal fissure |
| Chronic constipation |
| Chronic diarrhea |
| Fistula |
| Incontinence |
| Prolapsed hemorrhoids |
| Rectal prolapse |
| Skin tags |
| Infections |
| Abscess |
| Candida (intertrigo) |
| Corynebacterium minutissimum (erythrasma) |
| Molluscum contagiosum |
| Parasites (e.g., pinworms, scabies) |
| Sexually transmitted diseases (e.g., herpes simplex virus, human papillomavirus, syphilis) |
| Staphylococcus aureus |
| Malignancy |
| Bowen disease |
| Extramammary Paget disease |
| Squamous cell carcinoma |
| Medications |
| Chemotherapy |
| Colchicine |
| Neomycin |
| Quinidine |
| Miscellaneous |
| Idiopathic |
| Lumbosacral radiculopathy |
| Psychological disorders |
| Systemic disease |
| Aplastic anemia |
| Diabetes mellitus |
| Inflammatory bowel disease |
| Jaundice |
| Leukemia |
| Lymphoma |
| Thyroid disease |
| Topical irritants |
| Deodorants |
| Detergent/fabric softener |
| Hemorrhoid remedies |
| Soaps |
| Suppositories |
| Talcum powder |
| Tight clothing |
| Toilet paper dyes and perfumes |
Treatment of pruritus ani should address the underlying cause. Physicians should educate patients about proper hygiene and avoidance of perfumes, dyes, dietary irritants (especially caffeine), and tight clothing. Patients should keep the area dry and be aware of the itch-scratch cycle. A sedating antihistamine such as hydroxyzine (Vistaril) can reduce scratching during sleep. Patients with hemorrhoidal skin tags that make hygiene difficult can clean the area using a handheld showerhead, squirt bottle, sitz bath, or a bidet.
If conservative measures are ineffective, topical steroids can be used for a maximum of two to four weeks and only in low-potency formulations to minimize the risk of skin atrophy. A small trial found a 68 percent reduction in pruritus ani with topical hydrocortisone.6 Another small study compared a liquid antibacterial soap with topical methylprednisolone (not available in the United States).7 Both treatments were more than 90 percent effective in relieving pruritus ani, and not statistically different. A trial of capsaicin 0.006% cream (not commercially available but can be diluted with white paraffin) for chronic idiopathic pruritus ani showed relief in 31 of 44 patients in the treatment group compared with one of 44 patients in the placebo group.8 If pruritus persists after treatment, the area should be biopsied.
Anorectal Pain
Anorectal pain may be caused by a fissure, an abscess, thrombosed external hemorrhoids, proctitis, perineal sepsis, or proctalgia fugax. Patients liken anal fissure pain to passing “shards of glass,” and may have bright red blood in the stool (Figure 1). Physical examination may not be possible, although the fissure will be visible by gently effacing the anus. The physician should distinguish whether the fissure is acute (i.e., present for less than three months and looks like a laceration) or chronic (i.e., present for three months or more and is indurated, fibrotic, and has an anal skin tag or polyp). Chronic fissures tend not to respond to medical management, and eventually will require a lateral internal sphincterotomy to heal. Fissures located off-midline indicate an atypical etiology, such as Crohn disease, tuberculosis, leukemia, or human immunodeficiency virus. These fissures necessitate referral to a subspecialist.
Figure 1.
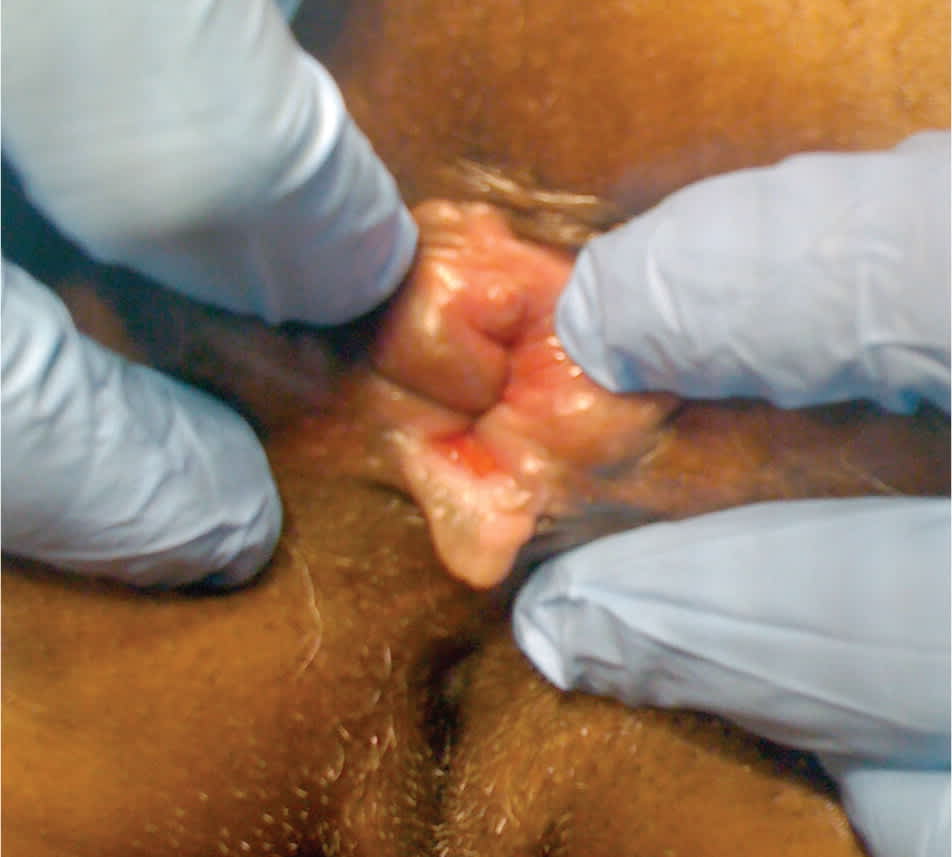
Anal fissure. Patients with anal fissure may experience a feeling of passing “shards of glass.”
Treatment of anal fissure starts with stool softeners, fiber, sitz baths, and topical analgesics. Pathophysiology is linked to high resting sphincter tone plus relative ischemia in the posterior midline,9 so therapies to relax the internal anal sphincter are useful. Topical nitroglycerin 0.2% ointment (compounded by a pharmacist) relaxes the sphincter and enables healing of the fissure.10 Topical nitroglycerin may cause headache that usually resolves within 30 minutes. Patients using it may become orthostatic, although this is rare. In a randomized clinical trial, 20 units of onabotulinumtoxinA (Botox) injected into the internal anal sphincter was shown to be superior to topical nitroglycerin for the treatment of anal fissure (n = 100; 92 versus 70 percent healed at two months). The only adverse effect was transient incontinence of flatus.11 Other studies have found favorable results with low-dose calcium channel blockers, such as topical diltiazem 2% and nifedipine 0.5%, or agents that relax the anal sphincter, such as bethanechol 0.1%.12,13 All of these treatments require compounding by a pharmacist. Fissures often recur and patients may require internal sphincterotomy.
Proctalgia fugax is a sudden severe pain in the rectum lasting seconds or minutes. The pain is sporadic and has no inciting trigger.14 The cause may be spasm of the internal anal sphincter. Various treatments have been described, including benzodiazepines, onabotulinumtoxinA,15 intravenous lidocaine (Xylocaine) infusion,16 sacral nerve stimulation,17 and pudendal nerve block.18 However, there is scant evidence that any of these treatments are effective.
Thrombosed hemorrhoids (Figure 2) are treated with hemorrhoidectomy if symptoms have been present for less than 72 hours, otherwise hemorrhoidectomy is likely to cause more discomfort than it relieves. The hemorrhoidectomy procedure has been described previously in American Family Physician.20 If the patient has symptoms for 72 hours or more, stool softeners and sitz baths will provide symptomatic relief. Any suspicious looking mass or lesion should be biopsied or evaluated by a subspecialist. Family physicians should maintain a high index of suspicion for anal cancer.
Figure 2.
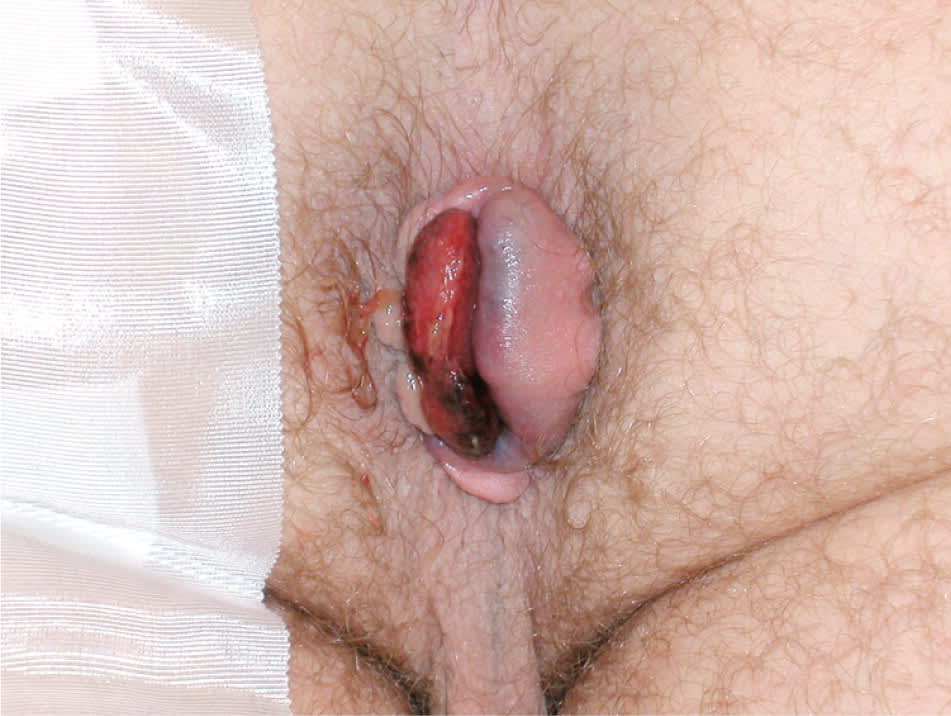
Thrombosed hemorrhoids are best treated by hemorrhoidectomy if present for less than 72 hours.
Proctitis presents as rectal discomfort, tenesmus, purulent discharge, abdominal pain, and urgency. The etiology can be infectious (e.g., herpes simplex virus, gonorrhea, chlamydia, human immunodeficiency virus/AIDS), inflammatory, or secondary to radiation or ischemia. A patient with diabetes mellitus or alcoholism who also has perineal pain, sepsis, crepitus, bullae, and induration may have Fournier gangrene, a medical emergency requiring immediate antibiotics and debridement.19
Anorectal Masses
Condyloma, abscess, polyp, prolapse, hemorrhoid, or anal cancer can present as an anorectal mass. A tender mass that begins insidiously, progressively worsens, and is not exacerbated by defecation may indicate abscess. Patients may report drainage of pus. Acute onset of pain and minimal findings on examination should arouse suspicion for an intersphincteric or deep postanal abscess, which should be addressed immediately by a surgeon before the development of sepsis or a more complicated abscess. A perianal abscess is easily drained with local anesthesia in the office. Antibiotics are often unnecessary after the abscess is drained.21 Immunocompromised patients and patients with diabetes may require hospitalization and intravenous antibiotics.
Rectal prolapse is the complete protrusion of all rectal layers through the anus with straining (Figure 3). Because the underlying condition is caused by weakened pelvic support, patients often have concomitant rectocele, cystocele, or other pelvic organ prolapse. Treatment is surgical.
Figure 3.
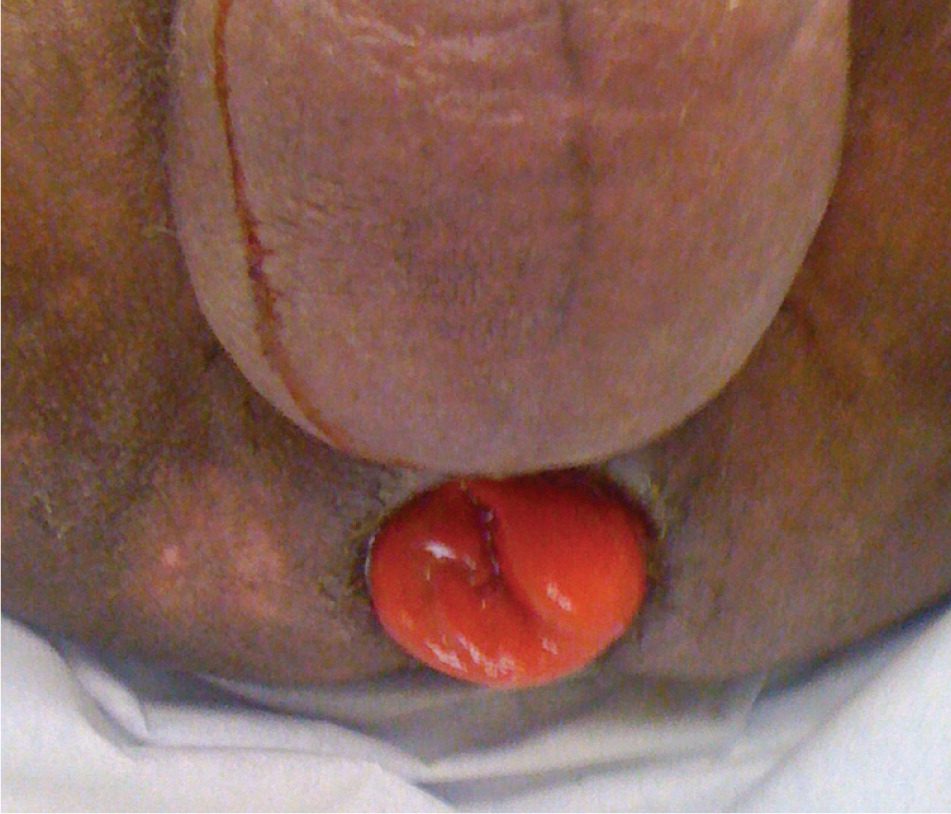
Rectal prolapse is caused by weakened pelvic support. Patients may have concomitant rectocele, cystocele, or other pelvic organ prolapse.
Condylomas (anogenital warts) are easily diagnosed by visual inspection (Figure 4). The extent of involvement should be documented with vaginal speculum examination or anoscopy. Biopsy is needed only if the patient is immunocompromised or if there are atypical features, such as ulceration, pigmentation, fixation, rapid growth, or failure to respond to three rounds of treatment.22 Other sexually transmitted diseases may be present and should be evaluated. Women should have a Papanicolaou smear. Multiple treatments are available, but evidence is insufficient to demonstrate superiority of one method over another. All have considerable adverse effects, failure rates, and recurrence rates.23 Condyloma size and location will guide treatment modality: surgical excision, physical or chemical ablation, or immunomodulation. Patients with very large lesions should be referred to a surgeon. Office treatment includes application of topical trichloroacetic acid 80% to 90%, or podophyllin, cryotherapy, or laser treatment. Home treatment includes podophyllotoxin or imiquimod (Aldara). Trichloroacetic acid is the only topical treatment approved for use in pregnant women.
Figure 4.
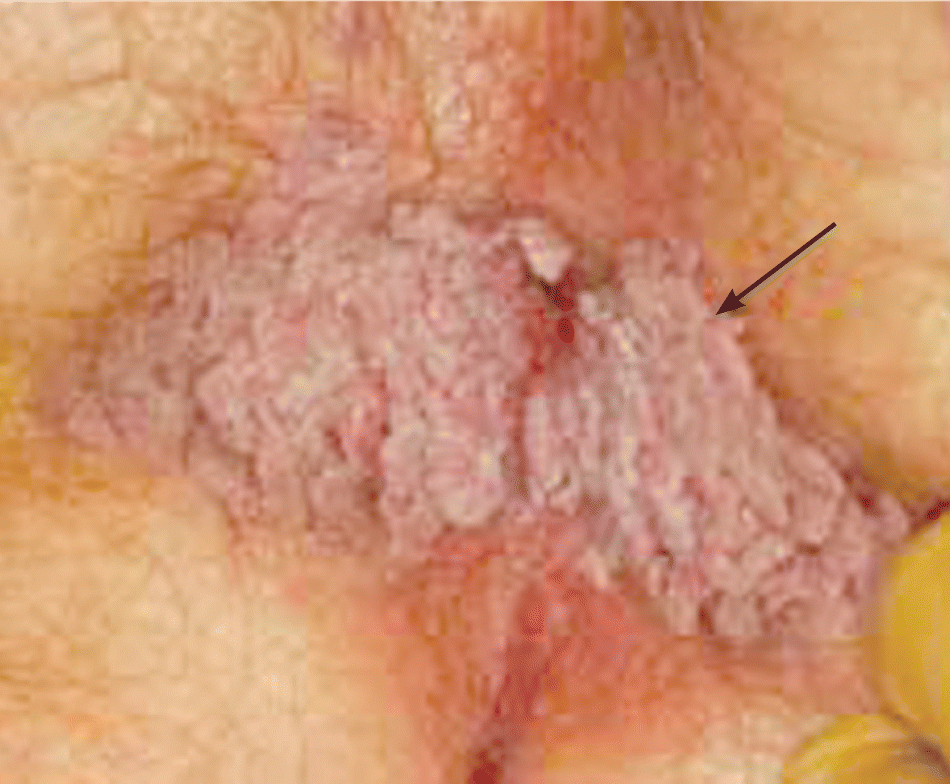
Extensive perianal condyloma acuminata (arrow). This condition is generally caused by infection with human papillomavirus 6 or 11.
Reprinted with permission from the National Procedures Institute, Midland, Mich. All rights reserved, 2000.
Anorectal Fistulas
Fistulas should be suspected in any patient with discharge, pain, swelling, or bleeding. One-half of anorectal abscesses form a fistula.21,24 On physical examination, there may be spontaneous or digitally-expressed discharge, an open sinus, granulation tissue, or a palpable cord.25 If the patient cannot tolerate a digital examination, anesthesia is needed. Fistulas are complex if they have an anterior tract, multiple tracts, cross 50 percent of the external anal sphincter, or are recurrent.25 Crohn disease, obstetric trauma, or local irradiation can increase the risk of developing anorectal fistulas. Patients with complex fistulas have a high risk of incontinence after treatment. A reasonable approach for low-risk patients with simple fistula is a trial of conservative therapy, such as sitz baths, high-fiber diet, and analgesics. Patients with nonhealing fistulas, complex risk factors, or complex fistulas should be referred to a surgeon.21 The tract must be precisely defined for any surgical therapy. Examination under anesthesia combined with magnetic resonance imaging or ultrasonography approaches 100 percent accuracy in classification.40
Anorectal Bleeding
Anorectal bleeding can be caused by hemorrhoids, fissures, polyps, diverticular disease, inflammatory bowel disease, or colorectal cancer.26,27,41 All anorectal bleeding must be evaluated and diagnosed. Colonoscopy is indicated in patients with unexplained bleeding, bleeding that persists despite treatment, associated systemic signs or symptoms (e.g., weight loss, iron deficiency anemia), age older than 40 years, and a family history of colorectal cancer.41
Hemorrhoids are the most common cause of anorectal bleeding, but can also present as pruritus or a mass on examination.26,27 Internal hemorrhoids occur above the dentate line and external hemorrhoids occur below the dentate line. Internal hemorrhoids are classified as grade I to IV based on degree of prolapse (Table 3). Increased fluid and fiber intake should be the initial treatment for symptomatic hemorrhoids. A Cochrane review showed fiber supplementation was associated with a 53 percent reduction in the risk of persistent symptoms or bleeding.28 Painful external hemorrhoids can be treated with topical steroids and analgesics. Persistent grade I, grade II, and grade III hemorrhoids can be treated by a subspecialist with rubber band ligation, sclerotherapy, or infrared coagulation. Rubber band ligation is the most effective treatment.29 Hemorrhoidectomy decreases the rate of recurrence of grade III hemorrhoids compared with rubber band ligation, but increases the risk of complications and pain, whereas grade IV hemorrhoids are best treated with hemorrhoidectomy.30 Stapled hemorrhoidopexy is a less painful alternative but is associated with a higher rate of recurrence.31
Table 3. Classification of Internal Hemorrhoids

| Grade | Description |
|---|---|
| I | Bulge with defecation |
| II | Prolapse with defection, recede spontaneously |
| III | Require digital replacement after prolapse |
| IV | Cannot be replaced |
Fecal Impaction and Incontinence
Fecal impaction is a partial or complete blockage of the colon by hard, dry stool. It presents as constipation or overflow incontinence. Approximately 1 percent of hospitalized older patients and 13 percent of patients with spinal cord injuries experience fecal impaction.32 Etiologies include dietary (low-fiber intake), metabolic (hypothyroidism), drug effects (opiate use), and neurologic (spinal cord injuries). Fecal impaction is diagnosed by a history of constipation or overflow incontinence, abdominal and digital rectal examination, and abdominal radiography. Red flags include fever, bloody diarrhea, or elevated white blood cell count, which could signify infection or diverticulitis. Treatment includes oral medications, enemas, suppositories, and manual disimpaction. Oral polyethylene glycol (Miralax) plus an electrolyte solution has an 89 percent response rate.33
Fecal incontinence is the involuntary loss of bowel function, and is a significant source of embarrassment and social isolation. Approximately 2 percent of the general public and 21 percent of community-dwelling older adults experience fecal incontinence.34 Fecal incontinence is clinically classified as overflow (secondary to fecal impaction), reservoir (secondary to diminished colonic or rectal capacity), or rectosphincteric (structural or neurologic damage to the anal sphincter). Physicians should ask about symptoms of rectal fullness or urgency. Patients with fecal incontinence may also have urinary incontinence, which should be concomitantly evaluated. A medication history will rule out laxative abuse or other medications associated with diarrhea. Medical and surgical history will identify anal sphincter injury or dysfunction (e.g., previous cerebral vascular accident, intervertebral disk herniation, rectal surgeries, anal sphincter injury during vaginal delivery). Physical examination should include a Mini-Mental State Examination if there is concern for dementia, a thorough neurologic examination, direct anal inspection, and a digital rectal examination to evaluate for impaction or decreased rectal tone. Further diagnostic studies could include anal manometry to evaluate rectal tone, gastrointestinal endoscopy to evaluate for obstructing masses, or ultrasonography and magnetic resonance imaging to evaluate for sphincter defects.34
Treatment of fecal incontinence should target the underlying etiology. Begin by removing physical barriers to the bathroom and instituting scheduled defecation schedules for patients with dementia. Fecal impaction should be treated, if present. Biofeedback and antidiarrheal agents such as loperamide (Imodium) can be helpful.34–36 Patients with incontinence that is refractory to conservative management or with evidence of sphincter injury or dysfunction (e.g., severed sphincter muscles, rectal prolapse, intussusception) should be referred to a surgeon. Surgical treatment options for fecal incontinence include an overlapping sphincter repair, total pelvic floor reconstruction or, less commonly, artificial bowel sphincters. Evidence for the effectiveness of surgical treatment is insufficient; the optimal treatment is likely a combination of nonsurgical and surgical treatments.37
Data Sources: We performed an initial review using Essential Evidence Plus, the Cochrane Database of Systemic Reviews, PEPID, and the National Guideline Clearinghouse. Topics included pruritus ani; anorectal pain (including anal fissures, rectal abscesses, thrombosed external hemorrhoids, proctitis, perineal sepsis, and proctalgia fugax); anorectal masses (including rectal condylomas, rectal polyps, rectal prolapse, and hemorrhoids); rectal bleeding (including hemorrhoids, diverticular disease, colorectal cancer, and inflammatory bowel disease); constipation; fecal impaction; and fecal incontinence. We performed a literature search in PubMed using the above topics as key words for our search. Search dates: May 2010 to September 2010.

