Clinical Question
Are there any weight-loss medications that are effective and safe for use in patients with type 2 diabetes mellitus?
Evidence-Based Answer
Fluoxetine (Prozac) and orlistat (Xenical) produce modest short-term weight loss, but their long-term benefits are unclear and their safety is uncertain. (Strength of Recommendation [SOR]: B, based on a meta-analysis of randomized controlled trials.) Topiramate (Topamax; immediate- and controlled-release formulations) can produce weight loss, but potential psychiatric and neurologic adverse effects limit its usefulness (SOR: B, based on randomized controlled trials.) Sibutramine (Meridia) produces weight loss but has been withdrawn from the U.S. market because of potential cardiovascular adverse effects.
Evidence Summary
A summary of weight-loss medications is presented in Table 1.1–4 Participants in all studies were obese middle-aged patients with diabetes under moderately poor control; all were taking medications for diabetes. Patients with diabetes can safely achieve modest short-term weight loss using fluoxetine or orlistat.
Table 1. Effectiveness and Safety of Weight-Loss Medications in Obese Middle-aged Patients with Type 2 Diabetes Mellitus
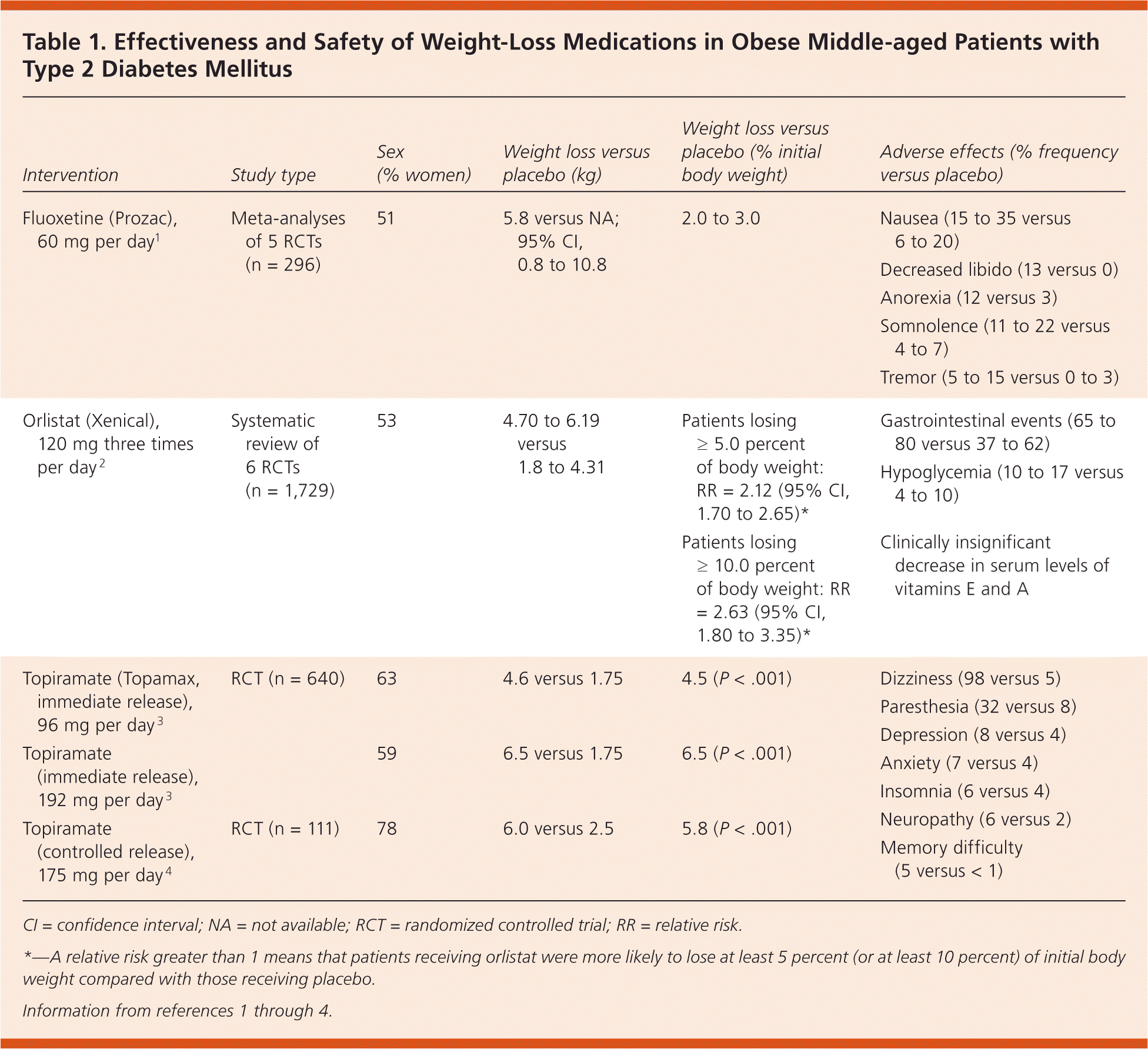
| Intervention | Study type | Sex (% women) | Weight loss versus placebo (kg) | Weight loss versus placebo (% initial body weight) | Adverse effects (% frequency versus placebo) |
|---|---|---|---|---|---|
| Fluoxetine (Prozac), 60 mg per day1 | Meta-analyses of 5 RCTs (n = 296) | 51 | 5.8 versus NA; 95% CI, 0.8 to 10.8 | 2.0 to 3.0 |
|
| Orlistat (Xenical), 120 mg three times per day2 | Systematic review of 6 RCTs (n = 1,729) | 53 | 4.70 to 6.19 versus 1.8 to 4.31 | Patients losing ≥ 5.0 percent of body weight: RR = 2.12 (95% CI, 1.70 to 2.65)* |
|
| Patients losing ≥ 10.0 percent of body weight: RR = 2.63 (95% CI, 1.80 to 3.35)* | |||||
| Topiramate (Topamax, immediate release), 96 mg per day3 | RCT (n = 640) | 63 | 4.6 versus 1.75 | 4.5 (P < .001) |
|
| Topiramate (immediate release), 192 mg per day3 | 59 | 6.5 versus 1.75 | 6.5 (P < .001) |
| |
| Topiramate (controlled release), 175 mg per day4 | RCT (n = 111) | 78 | 6.0 versus 2.5 | 5.8 (P < .001) |
|
CI = confidence interval; NA = not available; RCT = randomized controlled trial; RR = relative risk.
*—A relative risk greater than 1 means that patients receiving orlistat were more likely to lose at least 5 percent (or at least 10 percent) of initial body weight compared with those receiving placebo.
FLUOXETINE
Fluoxetine, a centrally acting appetite suppressant, facilitated gradual weight loss after eight to 16 weeks of treatment, with maximum effect after 52 weeks.1 Five studies involving 296 patients were identified. Only one study (n = 19) was continued for 52 weeks, but all five demonstrated weight loss compared with baseline. Weight loss was 3.4 kg at eight to 16 weeks (95% confidence interval [CI], –1.7 to –5.2 kg); 5.1 kg at 24 to 30 weeks (95% CI, –3.3 to –6.9 kg); and 5.8 kg at 52 weeks (95% CI, –0.8 to –10.8 kg). Investigators did not report data for weight loss in placebo groups, and they did not provide data on weight loss maintenance. The attrition rate during the study was 20 percent in the intervention group compared with 12 percent in the control group.
ORLISTAT
A greater proportion of patients with diabetes achieved a 5 or 10 percent loss from their initial body weight with orlistat compared with placebo (relative risk = 2.50 for 5 or 10 percent loss; 95% CI, 2.02 to 2.97).2 Participants receiving orlistat were more likely than those receiving placebo to have transient mild-to-moderate gastrointestinal adverse effects (relative risk = 1.46; 95% CI, 1.37 to 1.55).
TOPIRAMATE
Immediate-release topiramate produced significant weight loss in patients receiving metformin (Glucophage) for glycemic control. This was associated with absolute decreases in A1C levels of 0.1 percent in patients receiving placebo, 0.4 percent in those receiving 96 mg of topiramate per day, and 0.6 percent in those receiving 192 mg of topiramate per day (P < .001).3 Nine percent of participants in the lower-dosage group and 18 percent in the higher-dosage group withdrew because of serious adverse effects, compared with 7 percent of those in the placebo group. In addition to the adverse effects noted in Table 1,1–4 mild effects that occurred more often among persons receiving any dosage of topiramate included constipation (7 versus 2 percent in the placebo group), dry mouth (6 versus 1 percent), fatigue (12 versus 5 percent), and altered taste (7 versus 1 percent). The statistical significance of these effects was not reported. The study was terminated early to develop a controlled-release form of topiramate with fewer side effects. However, a subsequent study using controlled-release topiramate (175 mg per day) found that this formulation produced similar effects.4 Two persons receiving controlled-release topiramate had central nervous system effects, and four had psychiatric effects that persisted at the end of the study.
SIBUTRAMINE
Sibutramine produced greater weight loss than placebo over 12 to 26 weeks, but no studies have evaluated its long-term effectiveness.5 Potential adverse effects include increased diastolic blood pressure (effect size = 0.22; 95% CI, 0.07 to 0.38; P = .005) and increased heart rate (effect size = 0.53; 95% CI, 0.39 to 0.67).
Recommendations from Others
The American Diabetes Association recommends weight loss for patients who have or are at risk of developing diabetes, noting that even modest weight loss reduces insulin resistance.6 It does not recommend a specific weight-loss agent, and states that low-carbohydrate or low-fat calorie-restricted diets may produce weight loss for up to one year, after which behavior modification and physical activity are most helpful.

