There are approximately 300 million cases of malaria each year, resulting in 1 million deaths worldwide. Family physicians often encounter patients preparing to travel to malaria-endemic regions. Physicians should have basic knowledge of parasite transmission and malaria prevention. The risk of malaria acquisition is based largely on geographic location and travel season. Most cases occur in sub-Saharan Africa, the Indian subcontinent, and Southeast Asia between the months of May and December. Key elements in prevention include barrier protection and chemoprophylaxis. Travelers to malaria-endemic areas should be advised to use mosquito repellent at all times and bed netting at night. Prophylactic medication should be initiated before travel and continued after return. Travelers should be warned that malaria symptoms can present up to one year after a mosquito bite. Symptoms are vague, and may include fever, chills, arthralgias, and headaches. Travelers experiencing symptoms should seek prompt medical attention.
There are approximately 300 million cases of malaria each year, resulting in 1 million deaths worldwide.1 Reports from the Centers for Disease Control and Prevention (CDC) indicate that there are between 1,200 and 1,600 cases of malaria annually in the United States.2 In 2009, there was a 14 percent increase in reported cases of malaria (from 1,298 cases in 2008 to 1,484 cases in 2009).2 One factor contributing to disease resurgence is global climate change.3 Between 2011 and 2020, the global mean temperature is expected to rise by 0.4°C.3 This increase in temperature has been projected to lead to a 30 to 100 percent increase in mosquito abundance worldwide.3
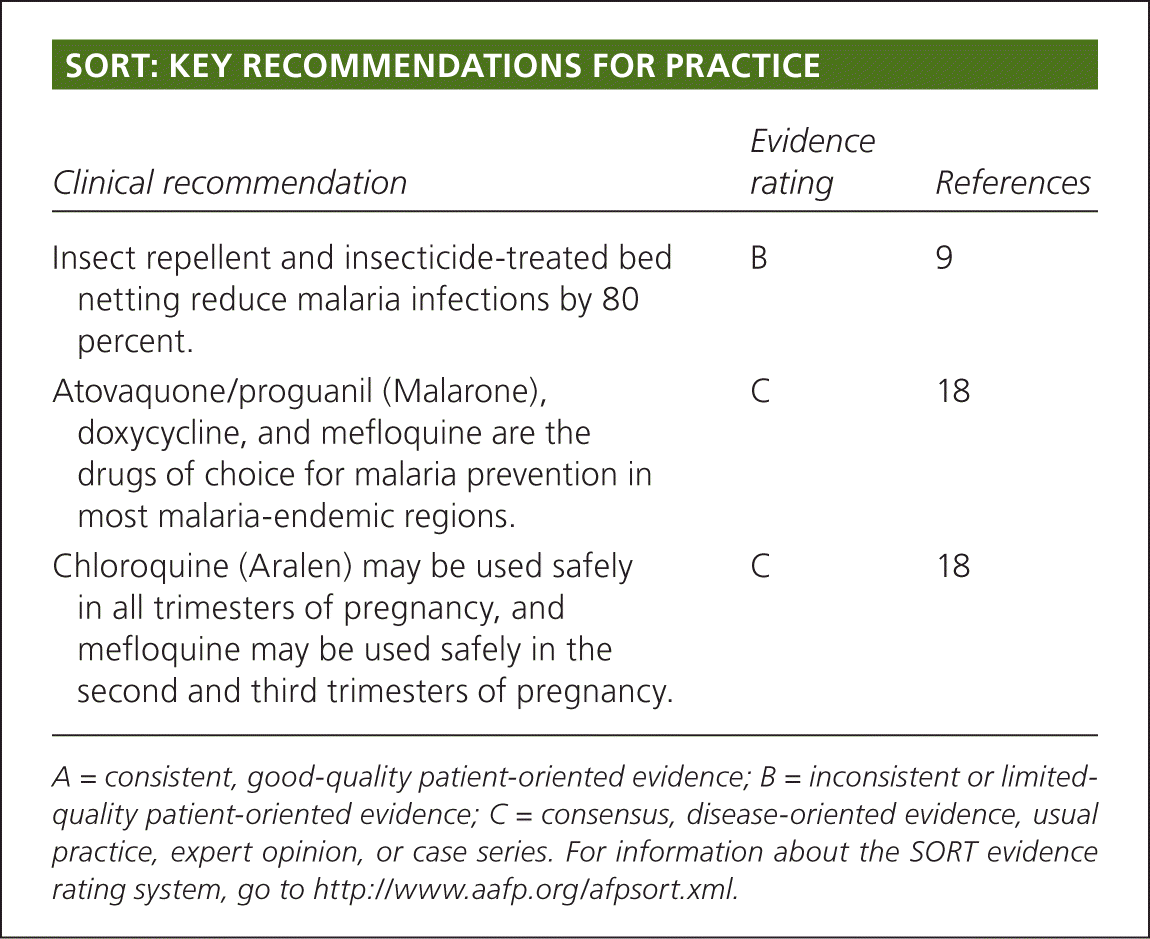
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Insect repellent and insecticide-treated bed netting reduce malaria infections by 80 percent. | B | 9 |
| Atovaquone/proguanil (Malarone), doxycycline, and mefloquine are the drugs of choice for malaria prevention in most malaria-endemic regions. | C | 18 |
| Chloroquine (Aralen) may be used safely in all trimesters of pregnancy, and mefloquine may be used safely in the second and third trimesters of pregnancy. | C | 18 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Most malaria infections in this country occur among persons who have traveled to areas with ongoing malaria transmission. In the United States, cases also can occur through exposure to infected blood products, congenital transmission, or local mosquito-borne transmission.2
Not only are mosquitoes proliferating with environmental change, but recent findings also suggest that malaria is becoming resistant to treatment. Family physicians can address these issues with a preventive approach that includes traveler education, risk assessment, barrier protection, and chemoprophylaxis.
Sources of Transmission
Five main species of parasites are responsible for transmission of malaria in humans: Plasmodium falciparum, Plasmodium vivax, Plasmodium ovale, Plasmodium knowlesi, and Plasmodium malariae.4 These protozoa are concentrated in different areas of the world, and each produces a different manifestation of infection. P. falciparum is the most life-threatening form of malaria.
These parasites are transmitted to humans by the bite of an infective female Anopheles mosquito. To produce eggs, the mosquito usually consumes a blood meal, thus needing humans and animals as hosts. The development of the protozoa in the mosquito takes 10 to 21 days, depending on the species of the parasite. After the parasites enter the host's liver, the replication stage begins. Subsequent replication occurs in erythrocytes and may last from one week to one year. Symptoms of malaria appear after the parasites leave the liver and start lysing red blood cells.
Risk Assessment
An individual risk assessment should be conducted for every traveler, taking into account the destination and season of travel.5 Physicians should provide travelers with resources that discuss risk factors for malaria transmission (Table 1).
Table 1. Malaria Resources

| Resource | Web site |
|---|---|
| Bill and Melinda Gates Foundation | http://www.gatesfoundation.org/topics/Pages/malaria.aspx |
| Centers for Disease Control and Prevention | http://www.cdc.gov/malaria/ |
| Roll Back Malaria | http://www.rollbackmalaria.org/ |
| UNICEF | http://www.unicef.org/health/index_malaria.html |
| World Health Organization | http://www.who.int/topics/malaria/en/ |
According to the World Health Organization, malaria was endemic in 106 countries in 2010.6 Most cases occur in sub-Saharan Africa, the Indian subcontinent, and Southeast Asia. A map of worldwide malaria endemicity is available on the CDC Web site at http://cdc.gov/malaria/map/. Malaria accounts for 5 percent of febrile illnesses in Ethiopia between the months of January and April, and up to 30 percent between the months of May and December.7
Precipitation is also a contributing factor for vector transmission because riverbeds and stagnant pools of water are breeding grounds for the Anopheles mosquito. Travelers should be advised that the highest risk of malaria is during and after the rainy season.8
Mosquito Bite Prevention
Mosquito sprays and bed netting are effective in preventing malaria transmission. A trial in the Bolivian Amazon showed that episodes of malaria were reduced by 80 percent among persons using insect repellent and insecticide-treated bed netting.9
The CDC recommends diethyltoluamide (DEET) and picaridin as repellents for malaria prevention.10 DEET concentrations between 4 and 30 percent are effective for malaria protection.11 Higher concentrations are not associated with increased levels of toxicity. The effectiveness of DEET plateaus at a concentration of 30 percent. A formulation of 4 percent offers a complete mean protection time of approximately 90 minutes, whereas a 23 percent formulation offers more than five hours of protection. Adverse effects of DEET include dermatitis, allergic reactions, and rare neurotoxicity. The American Academy of Pediatrics does not recommend DEET for infants younger than two months.12 The recommendations for DEET use in pregnant and lactating women are similar to those for nonpregnant adults.11
A 20 percent solution of picaridin is comparable to a 35 percent DEET solution.13 The highest concentration of picaridin sold in the United States is 15 percent, and the data are insufficient to support adequate protection against Anopheles mosquitoes at this concentration. Picaridin does not cause skin irritation and is safe to use in children and pregnant women.
In 2007, scientists in South America developed a mosquito repellent containing p-menthane-3,8-diol (PMD), a eucalyptus plant extract.14 The formula is less toxic, cheaper, and more effective against malaria than a 20 percent solution of DEET.14 In the United States, PMD is available as 65 percent and 10 percent concentrations.15 The U.S. Environmental Protection Agency recommends these products as repellents against mosquitoes, biting flies, and gnats.15 Adverse effects include skin and eye irritation.15
Barriers such as insecticide-treated netting and clothing are as important as repellents in the prevention of malaria. A study in sub-Saharan Africa concluded that bed netting reduces the incidence of malaria by at least 50 percent.16 Use of clothing treated with permethrin (a synthetic mosquito repellent) is effective in preventing mosquito bites.17
Chemoprophylaxis
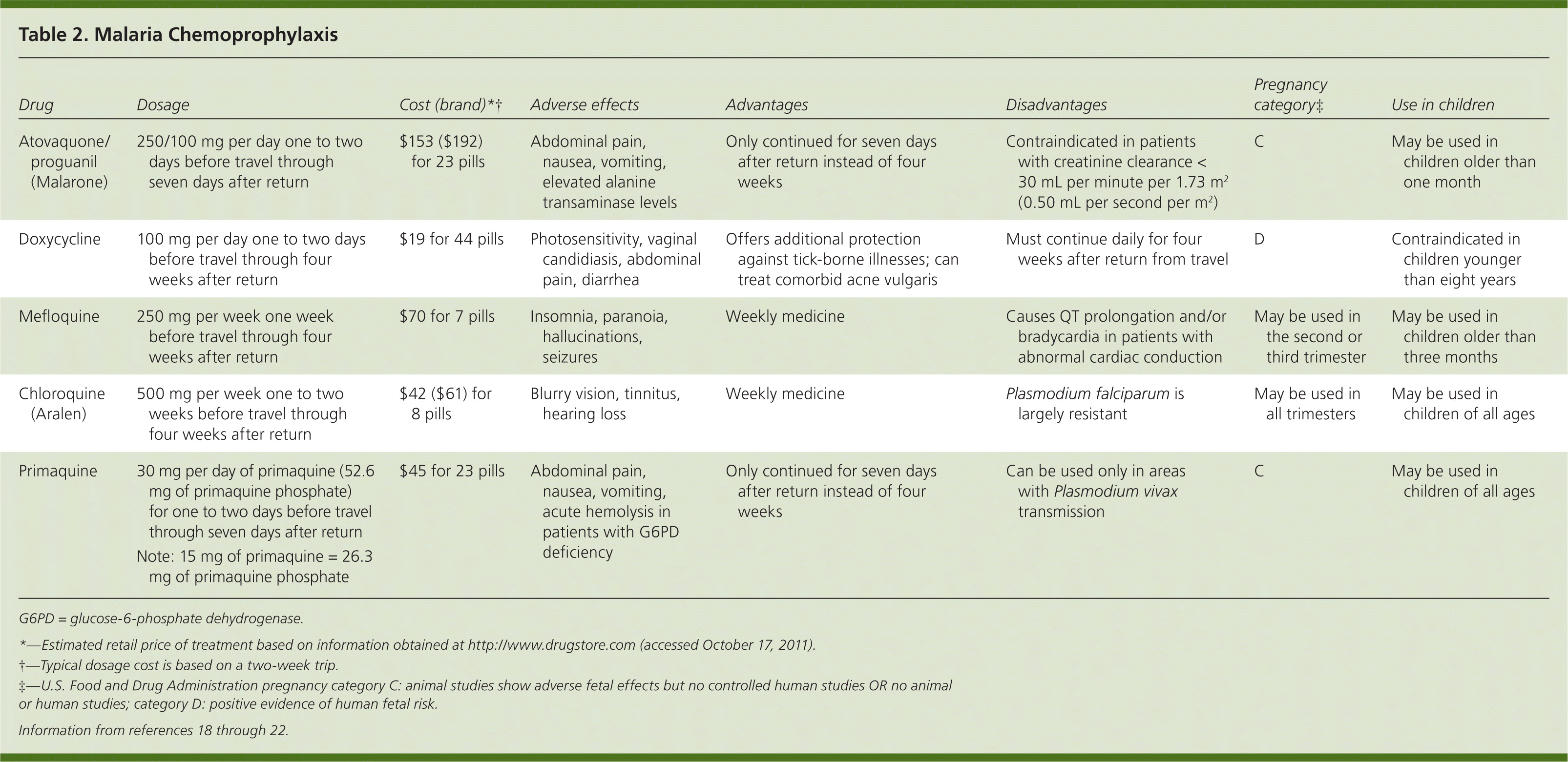
All recommended chemoprophylactic regimens involve taking medication before travel, during travel, and for a period of time after leaving the malaria-endemic region (Table 2).18–22 Beginning the regimen before travel is necessary to allow the antimalaria agent to enter the bloodstream before exposure to malaria-carrying parasites.18 Atovaquone/proguanil (Malarone), doxycycline, and mefloquine are the drugs of choice for malaria prevention in most malaria-endemic regions.18
Table 2. Malaria Chemoprophylaxis
| Drug | Dosage | Cost (brand)*† | Adverse effects | Advantages | Disadvantages | Pregnancy category‡ | Use in children |
|---|---|---|---|---|---|---|---|
| Atovaquone/proguanil (Malarone) | 250/100 mg per day one to two days before travel through seven days after return | $153 ($192) for 23 pills | Abdominal pain, nausea, vomiting, elevated alanine transaminase levels | Only continued for seven days after return instead of four weeks | Contraindicated in patients with creatinine clearance < 30 mL per minute per 1.732 (0.50 mL per second per m2) | C | May be used in children older than one month |
| Doxycycline | 100 mg per day one to two days before travel through four weeks after return | $19 for 44 pills | Photosensitivity, vaginal candidiasis, abdominal pain, diarrhea | Offers additional protection against tick-borne illnesses; can treat comorbid acne vulgaris | Must continue daily for four weeks after return from travel | D | Contraindicated in children younger than eight years |
| Mefloquine | 250 mg per week one week before travel through four weeks after return | $70 for 7 pills | Insomnia, paranoia, hallucinations, seizures | Weekly medicine | Causes QT prolongation and/or bradycardia in patients with abnormal cardiac conduction | May be used in the second or third trimester | May be used in children older than three months |
| Chloroquine (Aralen) | 500 mg per week one to two weeks before travel through four weeks after return | $42 ($61) for 8 pills | Blurry vision, tinnitus, hearing loss | Weekly medicine | Plasmodium falciparum is largely resistant | May be used in all trimesters | May be used in children of all ages |
| Primaquine [ | 30 mg per day of primaquine (52.6 mg of primaquine phosphate) for one to two days before travel through seven days after return | $45 for 23 pills | Abdominal pain, nausea, vomiting, acute hemolysis in patients with G6PD deficiency | Only continued for seven days after return instead of four weeks | Can be used only in areas with Plasmodium vivax transmission | C | May be used in children of all ages |
| Note: 15 mg of primaquine = 26.3 mg of primaquine phosphate |
G6PD = glucose-6-phosphate dehydrogenase.
*—Estimated retail price of treatment based on information obtained at http://www.drugstore.com (accessed October 17, 2011).
†—Typical dosage cost is based on a two-week trip.
‡—U.S. Food and Drug Administration pregnancy category C: animal studies show adverse fetal effects but no controlled human studies OR no animal or human studies; category D: positive evidence of human fetal risk.
ATOVAQUONE/PROGUANIL
Atovaquone/proguanil is a good choice for last-minute travelers because it can be started one to two days before travel, as opposed to one to two weeks with some of the other drugs.18 Common adverse effects include abdominal pain, nausea, vomiting, and elevated alanine transaminase levels. It is contraindicated in patients with a creatinine clearance of less than 30 mL per minute per 1.73 m2 (0.50 mL per second per m2).18 Atovaquone/proguanil is a U.S. Food and Drug Administration (FDA) pregnancy category C medication.
DOXYCYCLINE
Doxycycline is taken daily and provides additional protection against many infections, including tick-borne illnesses.18 Travelers should be aware that photosensitivity may increase in persons with prolonged sun exposure. Other adverse effects include vaginal candidiasis, abdominal pain, and diarrhea. Doxycycline is FDA pregnancy category D, and should be used only if maternal benefits outweigh fetal risks. It is contraindicated in children younger than eight years.
MEFLOQUINE
Mefloquine is taken weekly. It is considered safe to use during the second and third trimesters of pregnancy.18 Resistance to mefloquine is found in areas of China, Myanmar, Laos, Vietnam, and Cambodia.23 Five percent of patients taking mefloquine will experience neuropsychiatric effects (e.g., insomnia, paranoia, hallucinations, seizures) that lead to discontinuation of the drug.19,20
CHLOROQUINE
Chloroquine (Aralen) was the standard of care for malaria prevention for many years. However, as P. falciparum has become largely resistant to chloroquine, it is now recommended only for travelers going to the Middle East, Central America, Haiti, and the Dominican Republic.18 Chloroquine can be used in all trimesters of pregnancy and in children of all ages.18 Adverse effects may include blurry vision, tinnitus, and hearing loss.
PRIMAQUINE
Primaquine is used mainly in areas where P. vivax is the primary strain of malaria (e.g., parts of Central and South America). Patients must be tested for glucose-6-phosphate dehydrogenase deficiency before taking primaquine because it may cause hemolysis in affected persons.21 Other adverse effects include nausea, vomiting, and abdominal pain.21 Primaquine is an FDA pregnancy category C medication.
Five to 80 percent of patients treated for P. vivax malaria will relapse.22 As a preemptive measure, patients with P. vivax infection should be treated with a 14-day course of primaquine to prevent further disease.22 Primaquine therapy should be started on the same day as malaria treatment.22
Recognition of Illness
Travelers should be warned that adequate chemoprophylaxis does not guarantee full protection against malaria. Symptoms may appear from one week to one year after infection with the parasite. Relapsing illness may occur in patients who have completed a course of treatment.10 Travelers to malaria-endemic areas should seek medical attention for signs and symptoms of malaria, including fever, chills, headaches, and arthralgias.10
Presumptive Treatment
Travelers who decline malaria prophylaxis or who will be traveling to remote areas with limited access to health care may be prescribed a three-day supply of presumptive malaria treatment before travel.23 Travelers should be advised that self-treatment of a possible malaria infection is only a temporary measure, and that prompt medical evaluation is imperative.23 A three-day course of high-dose oral atovaquone/proguanil or artemether/lumefantrine (Coartem) may be prescribed.23 Travelers should take the medication if they experience high fevers, chills, or myalgias.23 Physicians who need assistance with the diagnosis or treatment of malaria should call the CDC Malaria Hotline (855-856-4713).
The Future of Malaria Prevention
A malaria vaccine is being developed for delivery through the World Health Organization's Expanded Programme on Immunization.24 It is being studied in African infants during the first 13 months of life, and has been reported to reduce transmission of malaria by 65 percent with few adverse effects.24 Along with barrier protection and chemoprophylaxis, vaccination may eventually play a key role in the eradication of malaria worldwide.24
Data Sources: We searched PubMed, Essential Evidence Plus, the Cochrane database, and UpToDate using variations of the key term malaria prevention. Search dates: July to September 2010, and July 2011.

