The treatment of gastroenteritis in children focuses on preventing dehydration. A child with minimal or no dehydration should be encouraged to continue his or her usual diet plus drink adequate fluids. Many studies have shown that a child's regular diet reduces the duration of diarrhea. Oral rehydration therapy with a rehydration solution can be used to treat diarrhea in children with mild to moderate dehydration. Ondansetron can decrease vomiting or help avoid the need for intravenous fluid, but it increases episodes of diarrhea. Probiotics can be used to shorten the course of diarrhea. Good handwashing reduces the incidence of acute gastroenteritis, but not rotavirus. The introduction of two rotavirus vaccines in the United States in 2006 significantly reduced the incidence of rotavirus gastroenteritis. The oral, live vaccines have strong safety records, despite a minimal incidence of intussusception.
Prevention of dehydration is the cornerstone of gastroenteritis treatment in children. A child with minimal or no dehydration should be encouraged to continue his or her usual diet plus drink adequate fluids. Many studies have shown that a child's regular diet reduces the duration of diarrhea.1,2 The BRAT diet (bananas, rice, applesauce, and toast) is too restrictive, unless the foods are part of the child's regular diet, and is not recommended by the American Academy of Pediatrics.3 This article, Part II of a two-part series, discusses therapies for gastroenteritis in children. Part I focuses on the evaluation and diagnosis.4
SORT: KEY RECOMMENDATIONS FOR PRACTICE
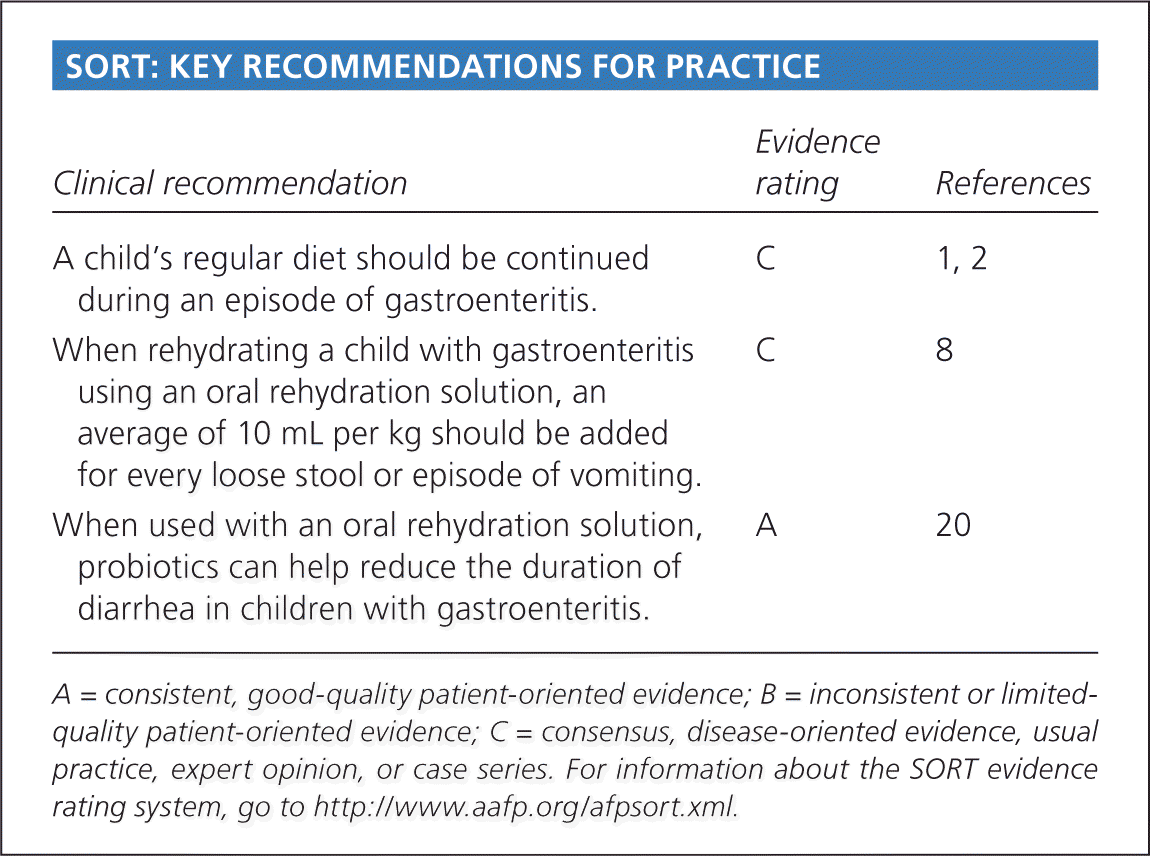
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| A child's regular diet should be continued during an episode of gastroenteritis. | C | 1, 2 |
| When rehydrating a child with gastroenteritis using an oral rehydration solution, an average of 10 mL per kg should be added for every loose stool or episode of vomiting. | C | 8 |
| When used with an oral rehydration solution, probiotics can help reduce the duration of diarrhea in children with gastroenteritis. | A | 20 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Early oral rehydration therapy using an oral rehydration solution (ORS), before the child becomes more severely dehydrated, is important and can be done at home.5 The best way to accomplish early treatment is to train the physician's office staff to explain how to use an ORS when caregivers call for help at the beginning of the child's illness. Clear liquids, such as water, sodas, chicken broth, and apple juice, should not replace an ORS because they are hyperosmolar and do not adequately replace potassium, bicarbonate, and sodium. These fluids, especially water and apple juice, can cause hyponatremia. An adult ORS also should not be used. The sodium-to-glucose ratio of the ORS should be equimolar at 1:1.
Using an over-the-counter ORS (e.g., Pedialyte, Infalyte, Rehydrate, Resol, Naturalyte) is recommended so that the child's caregiver can administer it easily. They are available in liter containers, “juice boxes,” and popsicles. An ORS is composed of sodium, dextrose, and bicarbonate in a ratio that does not overwhelm the hyperactive bowels with a hyperosmolar solution, but that is strong enough to replace the electrolyte loss.6,7 Caregivers should be instructed on the appropriate amount of ORS to use, because the label directions generally would not provide adequate replacement fluids in dehydrated children.8 Table 1 includes principles of gastroenteritis treatment in children that are endorsed by the Centers for Disease Control and Prevention, the World Health Organization, and the American Academy of Pediatrics.1,9
Table 1. Principles of Treating Children with Gastroenteritis and Dehydration

| Rehydration should be administered orally with an over-the-counter oral rehydration solution |
| Children should receive rapid oral rehydration (within three to four hours of symptom onset) |
| In infants who are breastfed, breastfeeding should continue |
| In infants who are formula-fed, diluting the formula is not recommended, and special formulas usually are not needed |
| As soon as the dehydration is corrected, a regular diet should resume |
| Ongoing diarrhea losses should be replaced with additional doses of an oral rehydration solution |
| Medications and unnecessary laboratory tests should be avoided |
Mild to Moderate Dehydration
In mild to moderate dehydration, the goal of treatment is to restore the fluid deficit and maintain hydration. The usual ORS composition is 50 mEq per L of sodium, 25 g per L of dextrose, and 30 mEq per L of bicarbonate. Mild to moderate dehydration can be managed by replacing the fluid loss as 50 mL per kg. This can be accomplished at home by competent caregivers using a syringe to administer approximately 1 mL of ORS per kg of body weight every five minutes over three to four hours.3 If vomiting occurs, the ORS should be restarted after 10 minutes to one hour. An average of 10 mL per kg should be added for every loose stool or episode of vomiting.8 A syringe or calibrated measuring device is highly recommended for more accurate measurement.10
Moderate to Severe Dehydration
Moderate to severe dehydration usually requires hospitalization, although oral rehydration therapy can be attempted in the emergency department using a syringe or a nasogastric tube if the infant or child refuses to drink. Criteria for hospital admission include caregivers who are unable to adequately administer an ORS at home, intractable vomiting, poor ORS intake by mouth or nasogastric tube, profuse diarrhea, unusual irritability or drowsiness, or no symptom improvement after 24 hours of ORS administration at home.5 The child's regular diet should be continued during oral rehydration therapy, if possible, but may have to wait until after intravenous fluids are administered when the child is hospitalized.
An accelerated method of oral rehydration therapy for infants and children with severe diarrhea and/or vomiting entails administering 30 mL per hour of an ORS for infants, 60 mL per hour for toddlers, and 90 mL per hour for older children.7 An average of 10 mL per kg should be added for every loose stool or episode of vomiting.8 Ondansetron (Zofran), a 5-hydroxytryptamine-3 serotonin antagonist, can be used in the emergency department if vomiting is hindering oral rehydration therapy.11 A meta-analysis showed that ondansetron (0.15 or 0.3 mg per kg intravenously, or 2 to 8 mg orally [single dose] depending on body weight or 1.6 to 4 mg per kg orally [six divided doses] depending on age) significantly decreased vomiting in children with acute gastroenteritis soon after administration of the drug (number needed to treat = 5). [corrected] The risk of requiring rehydration with intravenous fluids was significantly reduced (relative risk = 0.4; 95% confidence interval, 0.3 to 0.7). Patients taking ondansetron also had a significantly reduced risk of hospital admission (7.5 versus 14.6 percent; relative risk = 0.52; 95% confidence interval, 0.27 to 0.95). Ondansetron was well tolerated, except for increased episodes of diarrhea for up to 48 hours after use.12 Another meta-analysis showed that ondansetron decreased persistent vomiting, need for intravenous fluids, and hospital admissions in children with gastroenteritis. Other antiemetics should not be used because of potential adverse effects.11
Administration of intravenous fluids requires at least four to six hours in the emergency department or an overnight stay in the hospital. Rehydration with intravenous fluids replaces the sodium and water deficit, as well as the ongoing fluid deficit. Depending on the severity of dehydration, a child might need two intravenous lines or an intraosseous line. A rapid fluid bolus is given at a rate of 20 mL per kg of body weight. A bolus of 10 mL per kg should be used for a frail child. The intravenous fluid of choice is normal saline 0.9%, although a lactated Ringer solution also may be used. The choice of intravenous fluid depends on the level of serum sodium. Urinary output and serum electrolyte, blood urea nitrogen, creatinine, and serum glucose levels should be checked often. Maintenance fluids should be given at a daily rate of 100 mL per kg for the first 10 kg, 50 mL per kg for the next 10 kg, and 20 mL per kg for the next 10 kg.13,14 As soon as adequate rehydration has been achieved, oral rehydration therapy can begin along with a regular diet, and the patient should be weaned from intravenous fluids. Patients at high risk of aspiration due to obtundation from electrolyte imbalances can be given an ORS via nasogastric tube.15 Complications of rehydration with intravenous fluids include hyponatremia, hypernatremia, and hypoglycemia; serum electrolyte levels should be monitored closely.16
Probiotics
Probiotics are beneficial in modulating the immune response against foreign antigens in children with gastroenteritis.17 Probiotics do not colonize the gastrointestinal tract and are eliminated within one to two hours after ingestion. Probiotics degrade and modify dietary antigens and balance the anti-inflammatory response of cytokines. There is no known interaction between probiotics and medications.18 Probiotics are widely used in countries outside the United States because they are available over the counter, can be given orally at home, and are commonly recommended by physicians to limit the duration of diarrhea when it occurs.19 Their use is much less common in the United States.
A Cochrane review of 63 studies concluded that probiotics reduce the duration of diarrhea by approximately one day when used in conjunction with an ORS, but the review did not specify if one type of probiotic is superior.20 A meta-analysis of Lactobacillus therapy for acute gastroenteritis in inpatients showed that the therapy reduced the duration of diarrhea by 0.7 days and reduced the frequency of diarrhea by 1.6 stools on day 2 of therapy.21 A meta-analysis of the Lactobacillus GG strain showed that the therapy significantly reduced the duration of diarrhea by 1.1 days and of rotavirus-related diarrhea by 2.1 days.19
Studies of Saccharomyces boulardii in children who presented to the emergency department with acute gastroenteritis revealed significantly decreased stool frequency after 48 hours, a nearly one-day decrease in duration of diarrhea, and a one-day decrease in duration of hospitalization. There was no significant decrease in the number of children requiring intravenous fluids.22 In a randomized controlled trial of outpatients, S. boulardii therapy decreased duration of diarrhea by 1.5 days, accelerated recovery to less than seven days, and decreased the risk of prolonged diarrhea (greater than seven days). S. boulardii therapy worked best if given within 48 hours of diarrhea onset.23
In a randomized controlled trial of five different probiotics given for five days in an outpatient setting, the median duration of diarrhea and the number of stools were significantly reduced after the first 24 hours in children who took Lactobacillus GG or a mix of four bacterial strains (i.e., Lactobacillus delbrueckii var bulgaricus, Streptococcus thermophilus, Lactobacillus acidophilus, and Bifidobacterium bifidum). There was no effect on the number of children admitted to the hospital, on fever or its duration, or on vomiting or its duration. Of note, those who took S. boulardii showed no improvement.24
Antidiarrheals
In general, antidiarrheal medications should not be used in children with acute gastroenteritis because they delay the elimination of infectious agents from the intestines. Loperamide (Imodium) inhibits intestinal motility and can affect electrolyte and water movement through the bowel. Although loperamide is commonly used for gastroenteritis in older children, there are limited data to support it. Loperamide is not recommended in children younger than two years, and older children are at risk of constipation, central nervous system depression–like sedation, and nausea.5,25
Racecadotril (Acetorphan) is an antisecretory drug commonly used in Europe that inhibits intestinal enkephalinase without slowing intestinal transit or promoting overgrowth of bacteria.5 The drug reduces stool output and duration of diarrhea in acute gastroenteritis, including that caused by rotavirus. Although it is safe and effective, it is not available in the United States.26,27
Prevention
HANDWASHING
A meta-analysis of 30 studies revealed that improved hand hygiene reduced the incidence of gastrointestinal illness by 31 percent (95% confidence interval, 19 to 42). The use of regular soap was most beneficial, and antibacterial soap provided little additional benefit.28 Another study found that good hand hygiene reduced the incidence of gastroenteritis in general, but had little effect on the transmission of rotavirus.29,30
ROTAVIRUS VACCINE
The rotavirus vaccine is an oral, live vaccine. The Centers for Disease Control and Prevention's Advisory Committee on Immunization Practices recommends routine vaccination at two, four, and six months of age.30 There are specific guidelines for premature infants and infants who have missed the initial doses. Contraindications to the vaccine in infants are hypersensitivity to the vaccine, gastrointestinal tract congenital malformation, and severe combined immunodeficiency. The live virus is shed in the stool of 25 percent of infants who receive the vaccine and could be transmitted to an unvaccinated contact.31
The first rotavirus vaccine (Rotashield) was removed from the market because it was associated with an increased risk of intussusception, with an incidence of one in 10,000 infants.32 The current two vaccines are Rotarix (monovalent human vaccine) and Rotateq (pentavalent bovine-human reassortant vaccine). These vaccines have not been associated with an increased risk of intussusception at 30- and 42-day intervals, respectively.32 A recent study of Rotarix in Mexico and Brazil revealed a small risk of intussusception of one in 51,000 to 68,000 infants. However, in Mexico, vaccination with Rotarix would prevent 11,551 hospitalizations and 663 childhood deaths from rotavirus while causing two additional deaths and 41 additional hospitalizations from intussusception.32 A recent preliminary study of Rotateq in Australia also showed a similarly small increased risk of intussusception.33
Both rotavirus vaccines have strong safety records based on extensive studies, including randomized clinical trials. They prevent rotavirus gastroenteritis and reduce the severity of the disease.30–33 Among all infants, additional physician visits for gastroenteritis were reduced during the 2007 to 2008 rotavirus season by more than 90 percent in the South, Northeast, and Midwest regions of the United States, and by 53 to 63 percent in the West region after the rotavirus vaccines were introduced in 2006.34 Rotavirus-coded hospitalizations in the United States decreased by 83 percent in the 2007 to 2008 season and by 66 percent in the 2008 to 2009 season.35 In 2010, the finding of porcine circovirus (PCV) type 1 DNA in the Rotarix vaccine caused the U.S. Food and Drug Administration to temporarily halt its use. However, subsequent evaluation found no evidence that PCV1 and PCV2 pose a safety risk to humans.36
Data Sources: Essential Evidence Plus, PubMed, the Cochrane database, the Agency for Healthcare Research and Quality evidence reports, and the National Guideline Clearinghouse were searched using the key terms of pediatric gastroenteritis, pediatric dehydration, rotavirus vaccines, oral rehydration therapy, oral rehydration solutions, and probiotics. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Search dates: February 2011; PubMed was searched again on June 20, 2011.

