Interventions following a transient ischemic attack are aimed at preventing a future episode or stroke. Hypertension, current smoking, obesity, physical inactivity, diabetes mellitus, and dyslipidemia are all well-known risk factors, and controlling these factors can have dramatic effects on transient ischemic attack and stroke risk. For patients presenting within 48 hours of resolution of transient ischemic attack symptoms, advantages of hospital admission include rapid diagnostic evaluation and early intervention to reduce the risk of stroke. For long-term prevention of future stroke, the American Heart Association/American Stroke Association recommends antiplatelet agents, statins, and carotid artery intervention for advanced stenosis. Aspirin, extended-release dipyridamole/aspirin, and clopidogrel are acceptable first-line antiplatelet agents. Statins have also been shown to reduce the risk of stroke following transient ischemic attack, with maximal benefit occurring with at least a 50 percent reduction in low-density lipoprotein cholesterol level or a target of less than 70 mg per dL (1.81 mmol per L). For those with transient ischemic attack and carotid artery stenosis, carotid endarterectomy is recommended if stenosis is 70 to 99 percent, and perioperative morbidity and mortality are estimated to be less than 6 percent.
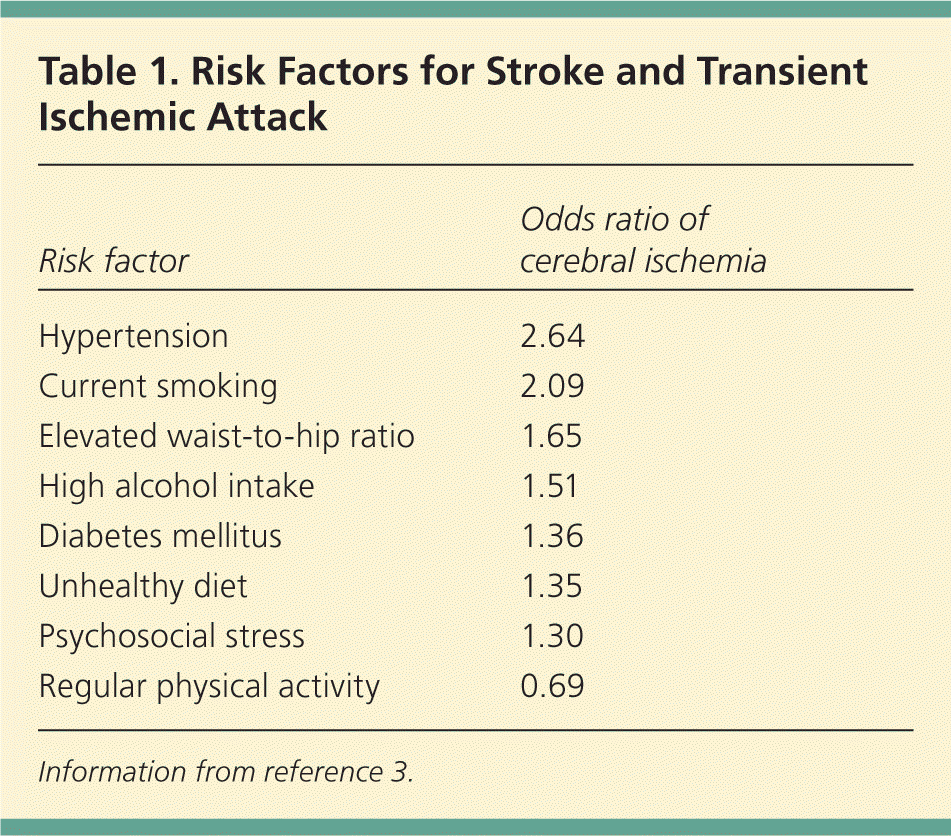
Following a transient ischemic attack (TIA), the focus of treatment should be prevention of future stroke. Risk factor modification with medical therapy is the cornerstone of stroke prevention.1,2 Five modifiable risk factors account for 82 percent of strokes: hypertension, current smoking, obesity, unhealthy diet, and physical inactivity.3 Table 1 lists odds ratios (ORs) for these and other common stroke and TIA risk factors.3 For stroke prevention following TIA, medical interventions such as antiplatelet therapy and carotid endarterectomy can lead to further decreases in recurrent events.1 However, one study showed that up to 25 percent of patients discontinued one or more of the prescribed drugs within three months of initiation, making patient education about TIA and future stroke risk paramount.4 Table 2 lists relative risk reductions (RRRs) with recommended preventive interventions.5–11 This article, part II of a two-part series, focuses on recommended interventions after a TIA. Part I discusses diagnosing the condition.12
SORT: KEY RECOMMENDATIONS FOR PRACTICE

| Clinical recommendation | Evidence rating | References | ||
|---|---|---|---|---|
| After a TIA, patients should be screened for the following risk factors, and these risk factors should be managed accordingly: | ||||
| Hypertension | A | 1, 6, 7 | ||
| Smoking | C | 1, 3, 16 | ||
| Physical inactivity | C | 1, 3, 5, 25–27 | ||
| Diabetes mellitus | B | 1, 28–32 | ||
| Dyslipidemia | A | 1, 3, 33, 34 | ||
| Antiplatelet agents are recommended for patients with a history of noncardioembolic TIA or stroke to prevent a subsequent stroke. | A | 1 | ||
| Acceptable first-line antiplatelet agents include the following: | ||||
| Aspirin alone | A | 1, 8, 40 | ||
| Extended-release dipyridamole/aspirin (Aggrenox) | B | 1, 8, 9, 42 | ||
| Clopidogrel (Plavix) | B | 1, 43, 44 | ||
| Statins are recommended for patients who have a history of TIA and a low-density lipoprotein cholesterol level of 100 mg per dL (2.59 mmol per L) or more. A reasonable low-density lipoprotein target is at least a 50 percent reduction or less than 70 mg per dL (1.81 mmol per L). | B | 1, 10, 11, 45 | ||
| For patients who have had a recent TIA or ischemic stroke and have ipsilateral stenosis, carotid endarterectomy is recommended if stenosis is 70 to 99 percent and perioperative morbidity and mortality are estimated to be less than 6 percent. If stenosis is 50 to 69 percent and perioperative morbidity and mortality are estimated to be less than 6 percent, carotid endarterectomy is recommended depending on patient-specific factors (e.g., age, comorbidities, surgical risk). | B | 1, 46, 47 | ||
TIA = transient ischemic attack.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Table 1. Risk Factors for Stroke and Transient Ischemic Attack
| Risk factor | Odds ratio of cerebral ischemia |
|---|---|
| Hypertension | 2.64 |
| Current smoking | 2.09 |
| Elevated waist-to-hip ratio | 1.65 |
| High alcohol intake | 1.51 |
| Diabetes mellitus | 1.36 |
| Unhealthy diet | 1.35 |
| Psychosocial stress | 1.30 |
| Regular physical activity | 0.69 |
Information from reference 3.
Table 2. Relative Risk Reduction with Recommended Interventions to Prevent Stroke
Risk Factor Modification
HYPERTENSION
Patients with hypertension have an increased risk of stroke, and blood pressure control reduces this risk.2 Blood pressure control after TIA is associated with a 30 to 40 percent RRR, with larger blood pressure decreases conferring a greater decrease in stroke risk.6,7 Key lifestyle modifications associated with blood pressure reduction include decreasing dietary sodium intake; losing weight; eating a diet rich in fruits, vegetables, and low-fat dairy products; exercising regularly; and consuming alcohol only in moderation.7
Variable blood pressure readings in a patient with hypertension may be an important predictor of cerebral ischemia, as well as the effects of antihypertensive agents. In a cohort of patients with TIA, increased visit-to-visit variability of systolic blood pressure and maximum systolic blood pressure reached were strong predictors of subsequent stroke (hazard ratios were 6.22 and 15.01, respectively).13 Calcium channel blockers have been shown to reduce this variability, thus lowering the risk of stroke.14,15
SMOKING
Current smoking has been shown to increase blood pressure, augment atherosclerosis, and increase the risk of stroke two- to fourfold compared with not smoking.3,16 There is a dose-response relationship between smoking and cerebral ischemia, with the heaviest smokers at the highest risk.1 The most effective method of cessation is a combination of behavioral therapy, nicotine replacement therapy, and social support.17 Nicotine replacement therapy alone increases the odds of quitting by 50 to 70 percent.18 Triple combination pharmacotherapy (nicotine patch, nicotine inhaler, and bupropion [Zyban]) for up to six months has been shown to be more effective than the nicotine patch alone for outpatient smokers with medical illnesses.19 All smokers should be advised to quit, and those with new medical diagnoses, such as TIA and stroke, are three times more likely to successfully change their lifestyle habits.20 Varenicline (Chantix) is another medical option for smoking cessation; however, the therapy is controversial because of its potential association with suicide and cardiovascular risk.21–23 High-quality studies addressing these risks are lacking, and results of a large ongoing study are not expected until 2017.
OBESITY, PHYSICAL INACTIVITY, AND DIET
Obesity (defined as a body mass index of 30 kg per m2 or more) is associated with a greater risk of death.24 More than one in four persons will become clinically obese, and increased waist-to-hip ratio increases the risk of stroke (OR = 1.65).3,25 Regular physical activity has been shown to reduce the risk of TIA and stroke (OR = 0.69).3 High-intensity activity leads to an RRR of 64 percent, compared with inactivity.5 Diets rich in fruits and vegetables, such as the Mediterranean diet, can help control body weight and have been shown to reduce the risk of stroke and myocardial infarction by at least 60 percent.26,27 The American Heart Association/American Stroke Association (AHA/ASA) recommends weight reduction, at least 30 minutes of moderate-intensity physical activity daily, and a diet low in sodium and high in fruits, vegetables, and low-fat dairy products, such as the DASH (Dietary Approaches to Stop Hypertension) diet.2
DIABETES MELLITUS
Diabetes is a well-established risk factor for cardiovascular disease and confers a hazard ratio of 2.27 for ischemic stroke.28 Patients with newly diagnosed diabetes have double the rate of stroke compared with the general population, making early intervention and risk factor modification imperative.29 In most patients who have had a TIA, the A1C target is less than 7 percent.1,30 A more intensive A1C reduction to targets of less than 6 percent has not been shown to decrease cardiovascular deaths or all-cause mortality.31 However, some studies have suggested a decrease in macrovascular events with lowering glucose levels to standard targets.30,32
DYSLIPIDEMIA
In the INTERSTROKE study, dyslipidemia was a significant risk factor for ischemic stroke (OR = 1.89).3 A large prospective cohort study showed a strong association between serum cholesterol levels and cerebral ischemia, with risk increasing proportionally to serum levels.33 In addition, a large meta-analysis studying the effect of statins on stroke reduction showed that the larger the reduction in low-density lipoprotein cholesterol (LDL-C) levels, the greater the reduction in stroke risk.34
Initial Management
The risk of stroke within 90 days after a TIA has been reported at 10 to 20 percent, with approximately one-half of these strokes occurring within the first 48 hours after initial presentation.35,36 Early initiation of treatment after a TIA, including medication and surgical intervention, can significantly reduce the risk of early stroke.37 This may suggest that hospitalization is beneficial in patients at high risk. Advantages of admission include the opportunity for complete diagnostic evaluation, confirmation of the diagnosis, and early treatment to reduce the risk of stroke.38 The potential administration of tissue plasminogen activator may be optimized if an early stroke occurs while the patient is hospitalized and should be considered in any patients with lesions on magnetic resonance imaging.1 Urgent access to tissue plasminogen activator and management of TIA can reduce subsequent stroke risk.
Of those presenting with TIA or minor stroke, 50 to 80 percent have elevated blood pressure on initial evaluation.39 Patients with systolic blood pressure greater than 140 mm Hg or diastolic blood pressure greater than 90 mm Hg are at higher risk of stroke after TIA (OR = 2.1, 1.9, and 1.6 at two, seven, and 90 days, respectively).39 Blood pressure reduction should be individualized based on comorbidities, age, and risk of hypotension.
ANTIPLATELET AGENTS
Antiplatelet agents are recommended for patients with a history of noncardioembolic TIA or stroke for the prevention of subsequent stroke. According to AHA/ASA guidelines, aspirin, extended-release dipyridamole/aspirin (Aggrenox), and clopidogrel (Plavix) are acceptable first-line agents.1 Aspirin is the most commonly used antiplatelet agent, and has been shown to be effective in the prevention of stroke for those with a history of TIA.8,40 The European Stroke Prevention Study 2 compared aspirin, dipyridamole (Persantine), and their combination for the prevention of stroke. Patients with a history of TIA or stroke who were given aspirin had an RRR of 18 percent for subsequent stroke and of 22 percent for subsequent TIA.8 The balance between effectiveness and safety of aspirin is optimized at 81 mg daily.41
Multiple studies, including the European Stroke Prevention Study 2, have shown that adding extended-release dipyridamole to aspirin leads to a further decrease in recurrent cerebral ischemic events, with an absolute risk reduction of 5.9 percent compared with placebo (number needed to treat = 17).8,9,42 The ESPRIT (European/Australasian Stroke Prevention in Reversible Ischaemia Trial) showed an additional 1 percent per year absolute risk reduction in stroke with dipyridamole/aspirin, compared with aspirin alone.42 A meta-analysis showed that dipyridamole/aspirin led to a 23 percent RRR when compared with aspirin, and a 37 percent RRR when compared with placebo.9
The PRoFESS (Prevention Regimen for Effectively Avoiding Second Strokes) noninferiority trial compared clopidogrel with dipyridamole/aspirin in patients with a history of ischemic stroke.43 The incidence of recurrent events was essentially the same in both groups (9 percent in the dipyridamole/aspirin group and 8.8 percent in the clopidogrel group).43 In the MATCH (Management of Atherothrombosis with Clopidogrel in High-Risk Patients) trial, clopidogrel plus aspirin was compared with clopidogrel alone for prevention of stroke following TIA or previous stroke. The combination led to higher bleeding risk without a statistically significant reduction in vascular events.44
Given the equivalence of antiplatelet agents in the prevention of stroke following TIA, choice of agent should be individualized based on patient risk and clinical profiles, cost consideration, and adverse effects.1 Using clopidogrel with aspirin is not recommended for stroke prevention because of bleeding risk, unless there is another indication for the regimen, such as cardiac stents or recent myocardial infarction.1
STATINS
The AHA/ASA recommends that patients with known coronary heart disease and patients with hypertension who are at high risk of cardiovascular disease take a statin for stroke prevention, regardless of initial LDL-C level.2 Statins are recommended for patients with a previous TIA or stroke without known coronary heart disease and an LDL-C level of greater than 100 mg per dL (2.59 mmol per L), and it is reasonable to aim for at least a 50 percent reduction in LDL-C level, or a target of less than 70 mg per dL (1.81 mmol per L) .1
For patients who have a history of TIA or stroke, statin therapy lowers the five-year absolute risk of stroke by 2.2 percent (16 percent RRR; number needed to treat = 45).10 A 50 percent or greater reduction in LDL-C level confers a 33 percent RRR for ischemic stroke and a 37 percent RRR for major coronary events, compared with no change or an elevation in LDL-C levels.11 For patients who have a history of TIA or stroke and no known coronary heart disease, statin therapy decreases the five-year absolute risk of developing clinically evident coronary heart disease from 8.6 to 5.1 percent (absolute risk reduction = 3.5 percent; number needed to treat = 29).45
CAROTID ARTERY INTERVENTION
Carotid artery atherosclerosis or stenosis is a well-established etiology of embolic stroke. Patients who have had a TIA or stroke are considered to have symptomatic disease. Recent studies have helped to clarify the debate between carotid endarterectomy and carotid artery stenting as the treatment of choice. The EVA-3S (Endarterectomy versus Angioplasty in Patients with Symptomatic Severe Carotid Stenosis) trial demonstrated that the incidence of stroke or death at 30 days in symptomatic patients was 3.9 percent following carotid endarterectomy and was 9.6 percent following carotid artery stenting.46 At six months, the risk of stroke or death was 6.1 percent for carotid endarterectomy and was 11.7 percent for carotid artery stenting.46 In the CREST (Carotid Revascularization Endarterectomy versus Stenting Trial), the four-year risk of stroke or death was 4.7 percent following carotid endarterectomy and was 6.4 percent following carotid artery stenting; however, the rates of myocardial infarction were higher after carotid endarterectomy.47
For patients who have had a TIA or ischemic stroke within the past six months and have ipsilateral stenosis, carotid endarterectomy is recommended if stenosis is 70 to 99 percent and perioperative morbidity and mortality are estimated to be less than 6 percent.1 If stenosis is 50 to 69 percent and morbidity and mortality are estimated to be less than 6 percent, carotid endarterectomy is recommended depending on patient-specific factors (e.g., age, comorbidities, surgical risk).1 No intervention is indicated if stenosis is less than 50 percent. Carotid artery stenting may be considered as an alternative under certain circumstances (e.g., difficulty accessing the area of stenosis surgically, low risk of complications with endovascular intervention).1
Data Sources: We searched Medline via Ovid and PubMed, Essential Evidence Plus, the National Guideline Clearinghouse, and the Cochrane database. Search terms included TIA, transient ischemic attack, TIA mimics, ABCD2, and cerebral ischemia. Search dates: January 2011 to February 2012.


