Women who have abnormal Papanicolaou test results may undergo colposcopy to determine the biopsy site for histologic evaluation. Traditional grading systems do not accurately assess lesion severity because colposcopic impression alone is unreliable for diagnosis. The likelihood of finding cervical intraepithelial neoplasia grade 2 or higher increases when two or more cervical biopsies are performed. Excisional and ablative methods have similar treatment outcomes for the eradication of cervical intraepithelial neoplasia. However, diagnostic excisional methods, including loop electrosurgical excision procedure and cold knife conization, are associated with an increased risk of adverse obstetric outcomes, such as preterm labor and low birth weight. Methods of endometrial assessment have a high sensitivity for detecting endometrial carcinoma and benign causes of uterine bleeding without unnecessary procedures. Endometrial biopsy can reliably detect carcinoma involving a large portion of the endometrium, but is suboptimal for diagnosing focal lesions. A 3- to 4-mm cutoff for endometrial thickness on transvaginal ultrasonography yields the highest sensitivity to exclude endometrial carcinoma in postmenopausal women. Saline infusion sonohysteroscopy can differentiate globally thickened endometrium amenable to endometrial biopsy from focal abnormalities best assessed by hysteroscopy. Hysteroscopy with directed biopsy is the most sensitive and specific method of diagnosing endometrial carcinoma, other than hysterectomy.
Cervical cancer was diagnosed in 12,200 American women in 2010, resulting in 4,210 deaths.1 The incidence of cervical cancer has decreased considerably with the use of Papanicolaou (Pap) testing. Treatment of high-grade cervical intraepithelial neoplasia (CIN) may be necessary to prevent progression to invasive cervical cancer, despite concern about adverse obstetric outcomes. Endometrial cancer, the most common female genital cancer, was diagnosed in more than 43,000 women and resulted in 7,950 deaths in 2010.1 This article summarizes recent updates for colposcopy, treatments for CIN, and methods of endometrial assessment.
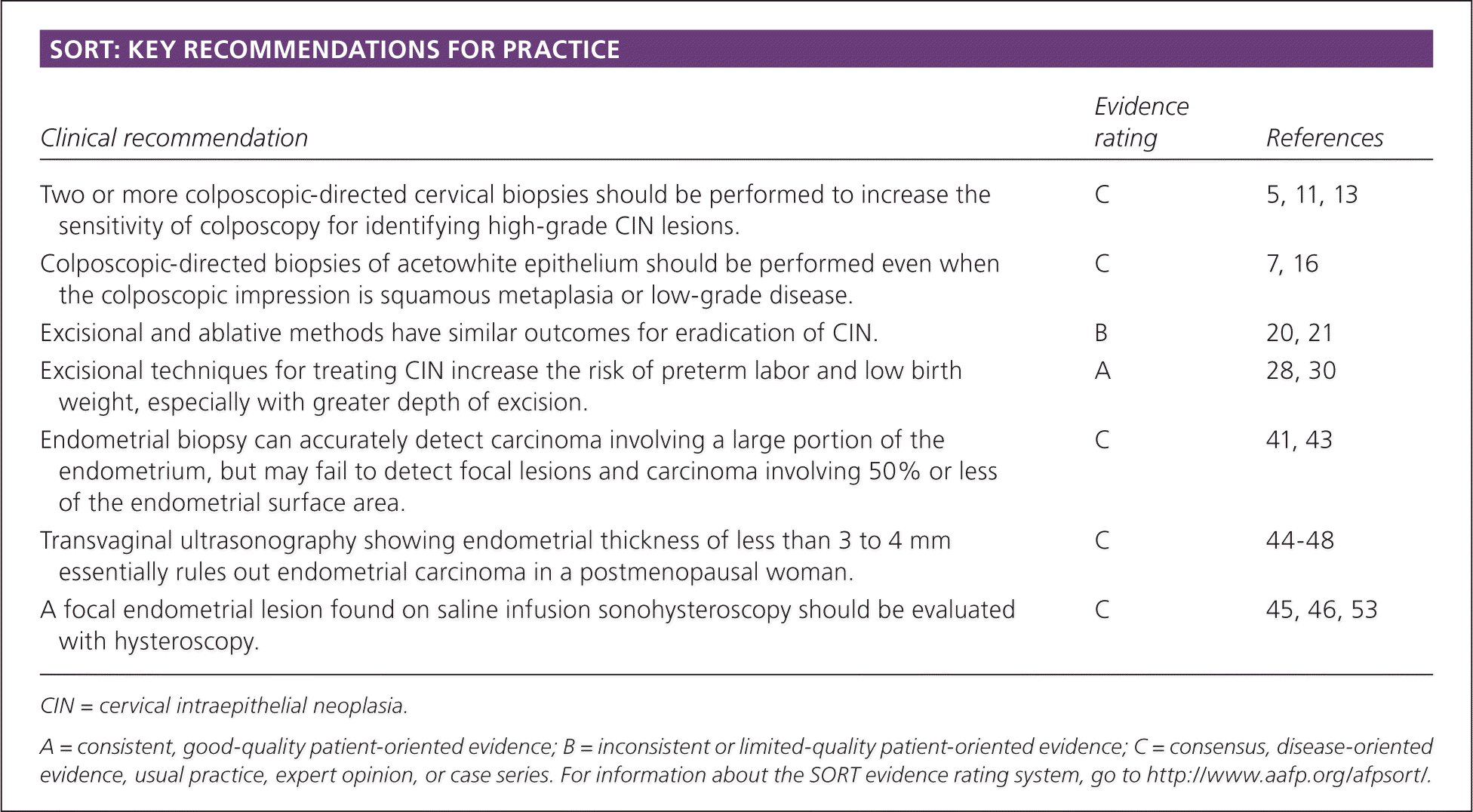
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Two or more colposcopic-directed cervical biopsies should be performed to increase the sensitivity of colposcopy for identifying high-grade CIN lesions. | C | 5, 11, 13 |
| Colposcopic-directed biopsies of acetowhite epithelium should be performed even when the colposcopic impression is squamous metaplasia or low-grade disease. | C | 7, 16 |
| Excisional and ablative methods have similar outcomes for eradication of CIN. | B | 20, 21 |
| Excisional techniques for treating CIN increase the risk of preterm labor and low birth weight, especially with greater depth of excision. | A | 28, 30 |
| Endometrial biopsy can accurately detect carcinoma involving a large portion of the endometrium, but may fail to detect focal lesions and carcinoma involving 50% or less of the endometrial surface area. | C | 41, 43 |
| Transvaginal ultrasonography showing endometrial thickness of less than 3 to 4 mm essentially rules out endometrial carcinoma in a postmenopausal woman. | C | 44–48 |
| A focal endometrial lesion found on saline infusion sonohysteroscopy should be evaluated with hysteroscopy. | C | 45, 46, 53 |
CIN = cervical intraepithelial neoplasia.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort/.
Colposcopy
COLPOSCOPIC IMPRESSION
Women who have abnormal Pap test results may undergo colposcopy to determine the biopsy site for histologic evaluation. Colposcopy allows for visualization of the lower genital tract using magnification and illumination after applying dilute acetic acid.2,3 The standard method of evaluating color, vessels, and margins of a colposcopic lesion (i.e., grading) to determine the most serious lesion for biopsy has been questioned.4 Colposcopic findings have not been shown to correlate strongly with the severity of cervical dysplasia5 (Figure 16). In one study, colposcopic impression of a high-grade lesion identified only 56% of histologic CIN grade 2 or higher (CIN 2+).7 The sensitivity of the first colposcopic-directed biopsy for detection of CIN 2+ is 52%, which confirms that many lesions may be difficult to detect and highlights the risk of a false-negative biopsy result.8 Human papillomavirus (HPV) type 16 is associated with the most prominent colposcopic abnormalities, whereas lesions with other oncogenic HPV types may be missed by colposcopy because they do not appear as distinctly acetowhite.9 High-grade lesions in thin (atrophic) epithelium may be underdiagnosed.10 Therefore, colposcopic impression cannot be a reliable indicator of where to perform cervical biopsies.11
Figure 1.
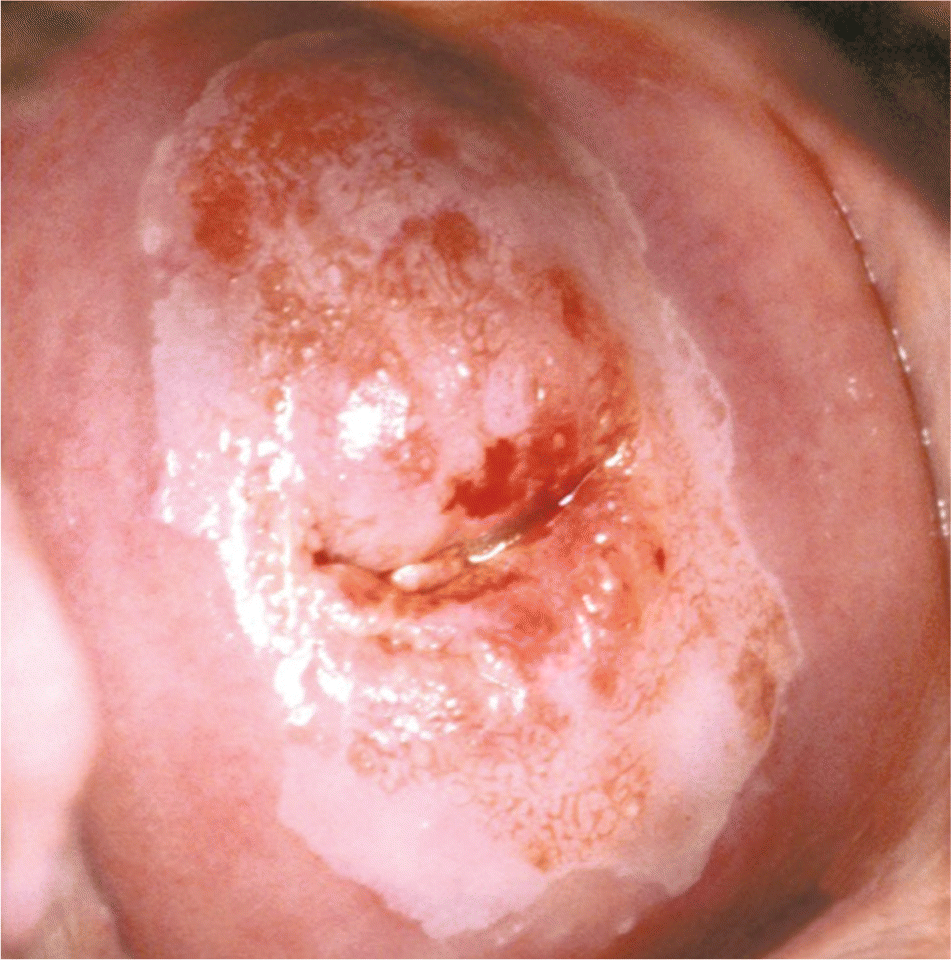
Colpophotograph of a woman with low-grade squamous intraepithelial lesion cytology. Colposcopic impression is low-grade disease with unsatisfactory colposcopy. Histology revealed cervical intraepithelial lesion grade 3.
Reprinted with permission from Apgar BS, Brotzman GL, Spitzer M. Colposcopy Principles and Practice: An Integrated Textbook and Atlas. 2nd ed. Philadelphia, Pa.: Saunders Elsevier; 2008:156.
Because histology determines disease severity and dictates management,12 methods have been sought to improve the sensitivity of colposcopy. Recent evidence demonstrates that colposcopists should perform multiple cervical biopsies and consistently sample acetowhite epithelium.8,13 Biopsies performed in areas with a colposcopic impression of high-grade findings increase the yield of histologic CIN 3+ lesions 10-fold (38%) compared with biopsies taken from areas that appear to be normal or low grade (3.8%).14 However, because colposcopic-directed biopsies of abnormal-appearing areas miss many CIN 2+ lesions, two or more cervical biopsies should be considered.15 This approach increases the sensitivity of colposcopy for identifying CIN 2+ lesions, regardless of the expertise of the colposcopist5,11,13 (Figure 26).
Figure 2.
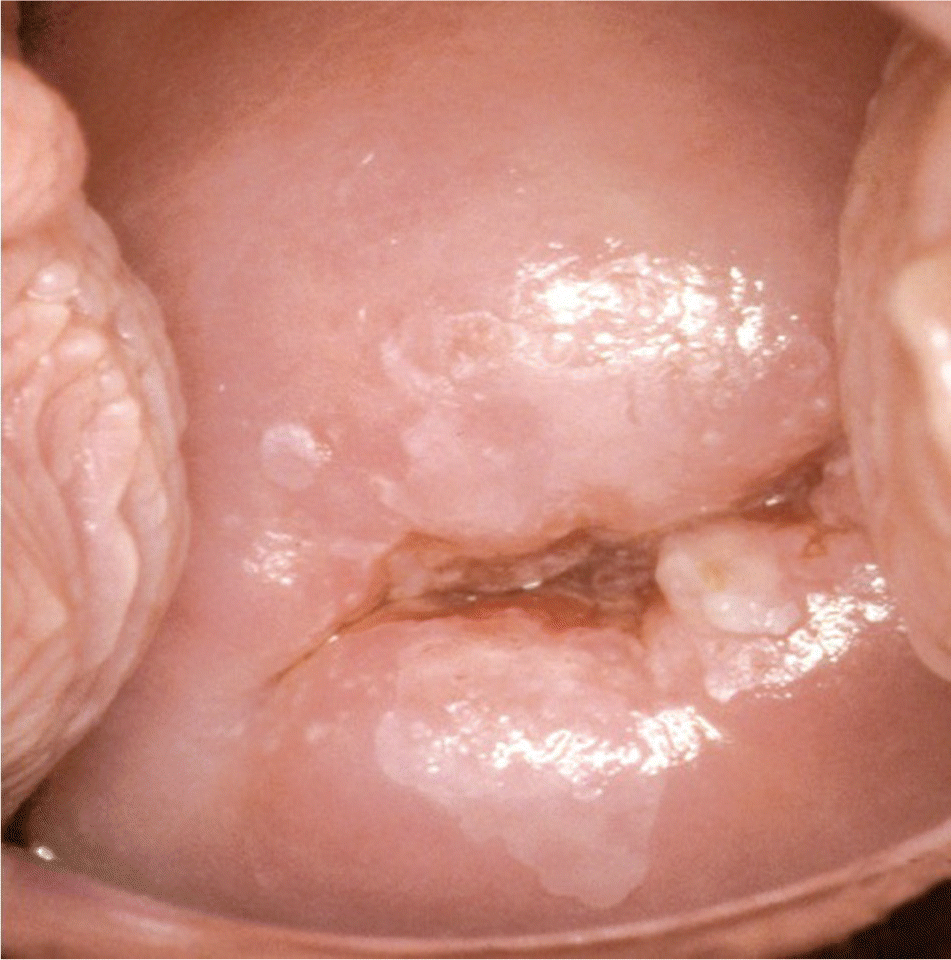
Colpophotograph of a woman with atypical squamous cells, cannot exclude high-grade squamous intraepithelial lesion (ASC-H). There is a significant amount of acetowhite epithelium. Multiple biopsies showed cervical intraepithelial lesion grade 1 and grade 3.
Reprinted with permission from Apgar BS, Brotzman GL, Spitzer M. Colposcopy Principles and Practice: An Integrated Textbook and Atlas. 2nd ed. Philadelphia, Pa.: Saunders Elsevier; 2008:224.
The role of random biopsies in increasing the sensitivity of colposcopy is being investigated.13,15 In one study, random four-quadrant biopsies at the squamocolumnar junction in areas without visible lesions, plus an endocervical curettage, diagnosed 37% of CIN 2+ lesions.15 The CIN lesions detected by random biopsy were significantly smaller, involved fewer quadrants, and were lower grade than those detected by colposcopic-directed biopsy.15 These lesions may have a higher rate of regression than those diagnosed by colposcopic-directed biopsy.14 Because of this, current guidelines do not recommend random cervical biopsies of normal-appearing epithelium.
Given that the accuracy of colposcopy is lower than anticipated, criteria for determining where to biopsy are needed. Identification of an acetowhite lesion on colposcopy has a sensitivity of 93% for predicting subsequent identification of CIN 2+ over two years.16 However, the specificity is 67% to 74% because most women with acetowhite lesions do not have high-grade disease.16 Sensitivity is higher when a biopsy is taken from an acetowhite area rather than from a lesion based on traditional grading characteristics. Using a grading system may encourage colposcopists to perform a biopsy only if the lesion appears high grade, and high-grade lesions that are small and subtle may be missed. Data from one study 16 demonstrate that colposcopic-directed biopsies of acetowhite lesions should be performed even when the colposcopic impression is squamous metaplasia or low-grade disease7,16 (Figure 16).
ENDOCERVICAL CURETTAGE
According to the American Society for Colposcopy and Cervical Pathology (ASCCP), endocervical curettage should be performed in specific situations, such as unsatisfactory colposcopy following low-grade intraepithelial lesion, colposcopic evaluation of high-grade squamous intraepithelial lesion (Figure 36), or initial evaluation of all subcategories of atypical glandular cell cytology.12 However, there is concern that endocervical curettage may encourage unnecessary excisional procedures.17,18 There is an argument that endocervical curettage is of limited benefit if excisional treatment is recommended. One study demonstrated that 11% of CIN 3+ was diagnosed only by a positive endocervical curettage result.14 However, another investigation found that endocervical curettage increased the detection of CIN 2+ in only 1% of 13,115 colposcopic-directed biopsies, with 99 endocervical curettages needed to detect one additional case of CIN 2+.19 Therefore, the value of the procedure remains controversial.
Figure 3.
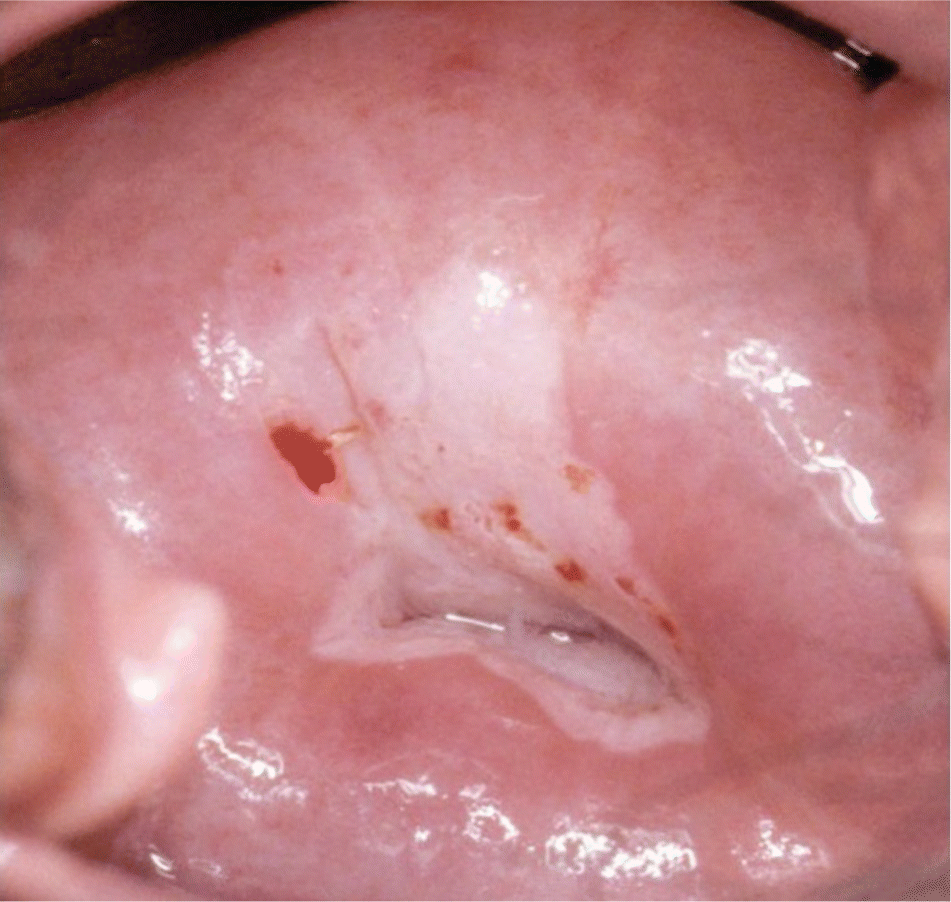
Colpophotograph of a woman with high-grade squamous intraepithelial lesion cytology. Colposcopic impression is high-grade disease and unsatisfactory colposcopy. Ectocervical histology showed cervical intraepithelial lesion (CIN) grade 1 and grade 3. Endocervical curettage revealed CIN 3. The patient is not a candidate for ablation, and diagnostic excision should be performed.
Reprinted with permission from Apgar BS, Brotzman GL, Spitzer M. Colposcopy Principles and Practice: An Integrated Textbook and Atlas. 2nd ed. Philadelphia, Pa.: Saunders Elsevier; 2008:251.
Treatment of Cervical Intraepithelial Neoplasia
EXCISIONAL AND ABLATIVE PROCEDURES
Following a colposcopic-directed biopsy result showing CIN 2/3 or persistent CIN 1, treatment options include ablation (i.e., cryotherapy or laser) or diagnostic excision (i.e., loop electrosurgical excision procedure [LEEP] or cold knife conization).12 Excisional and ablative methods have similar treatment outcomes for eradication of CIN. No significant differences in success rates were observed with any modality.3,20–22 Procedure-specific indications are reviewed in Table 1.3,12,20–31
Table 1. Treatments for Cervical Intraepithelial Neoplasia
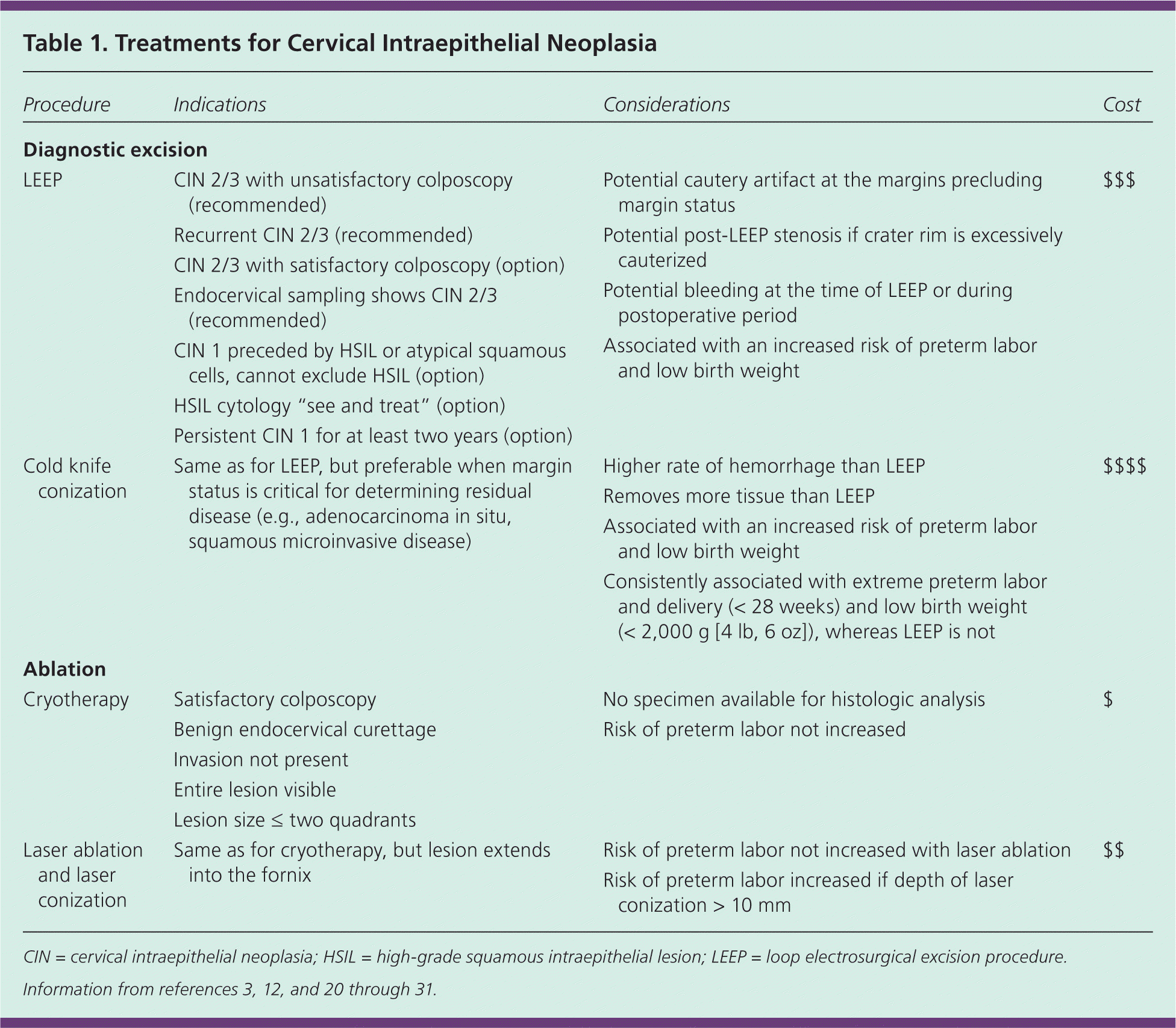
| Procedure | Indications | Considerations | Cost |
|---|---|---|---|
| Diagnostic excision | |||
| LEEP |
|
| $$$ |
| Cold knife conization |
|
| $$$$ |
| Ablation | |||
| Cryotherapy |
|
| $ |
| Laser ablation and laser conization |
|
| $$ |
CIN = cervical intraepithelial neoplasia; HSIL = high-grade squamous intraepithelial lesion; LEEP = loop electrosurgical excision procedure.
LEEP and cold knife conization allow histologic review of the excised tissue, whereas ablative techniques destroy the transformation zone, precluding histologic evaluation.3 LEEP excises the cervical transformation zone and a small amount of stroma.23 Compared with cold knife conization, LEEP can be performed in the office under local anesthesia and removes less tissue.20,21,24 Because LEEP can cause cautery artifact at the margins, cold knife conization is preferable when margin status is critical for determining residual disease and clinical management, as in adenocarcinoma in situ and suspected squamous microinvasion.25 Complications vary based on method of excision (Table 13,12,20–31 ).
Effect on Pregnancy Outcomes. Retrospective, case-control observations conclude that overly aggressive use of diagnostic excisional procedures may produce long-term adverse obstetric outcomes.28–30 Cold knife conization and LEEP are associated with a small increase in the risk of preterm labor and low birth weight.28,30 The risk of preterm delivery increases if the depth of LEEP or laser conization is more than 10 mm.28 The value of cervical length measurements during pregnancy for predicting preterm labor in women with a history of CIN treatments is unknown.30 Laser vaporization and cryotherapy did not affect outcomes, supporting the theory that ablation removes less tissue than excision.28–30 According to absolute risks, previous treatment with LEEP would result in two perinatal deaths per 1,000 pregnancies.29 The small absolute risk of preterm labor following cervical excisional procedures must be balanced with the risk of untreated CIN 3.30 None of the treatments affected fertility.28
Post-LEEP Margin Status. The goals of excisional treatment are complete removal of the lesion and the transformation zone, resulting in interpretable margins.31 There are conflicting reports as to whether margin status predicts the risk of recurrent disease.12 Treatment failure can occur in women with clear or involved margins.32 Women with clear margins show a pooled prevalence of high-grade residual disease of 3% vs. 18% in women with involved or uncertain margins (P < .0001).31
Most women with involved margins will not develop persistent or recurrent disease.12 Based on the lack of definitive data showing that margin status independently predicts residual disease, it is preferred that women with CIN 2/3 and positive endocervical margins undergo cytologic and endocervical sampling four to six months after treatment, rather than immediate retreatment.12 If a decision is made to retreat, repeat excision or hysterectomy is acceptable.12
HUMAN PAPILLOMAVIRUS DNA TESTING
HPV DNA testing appears to be more sensitive than colposcopy or cytology for detecting treatment failures for CIN 2/3.33–35 One systematic review showed a sensitivity of nearly 77% for cytology and nearly 91% for HPV DNA testing.36 In another systematic review of women who had successful treatment, 84% had negative HPV DNA testing following LEEP.33 Women who tested negative for HPV had no recurrent disease during a two-year follow-up period.37
According to the ASCCP and the American College of Obstetricians and Gynecologists, HPV DNA testing is an acceptable option for post-LEEP management of CIN 2/3.12,38 HPV DNA testing and cytology (co-testing) should be performed posttreatment at 12 and 24 months.12 If either test result is positive, colposcopy with endocervical sampling is recommended.12 Women who have negative co-testing results twice can repeat co-testing in three years and return to routine screening if results are negative.12 The risk of neoplastic invasion has been reported as late as two decades after treatment.3,39,40 ASCCP recommends a 20-year period of routine follow-up screening.12
Endometrial Assessment
ENDOMETRIAL BIOPSY
Endometrial biopsy was developed for in-office assessment of the endometrium as an alternative to dilation and curettage.41 A meta-analysis concluded that the highest sensitivity of available devices for detecting endometrial carcinoma was greater than 99% in postmenopausal women and 91% in premenopausal women.42 Studies of preoperative endometrial biopsy in women with known endometrial carcinoma showed lower sensitivities (68% to 92%).41 In a study of hysterectomy specimens, endometrial biopsy detected all cases in which the tumor involved greater than 50% of the endometrium, but missed tumors involving less surface area.43 Endometrial biopsy can reliably detect carcinoma involving a large portion of the endometrium, but is suboptimal for evaluation of focal lesions.41,43
TRANSVAGINAL ULTRASONOGRAPHY
Transvaginal ultrasonography can be used to triage women with suspected endometrial pathology. In post-menopausal women, an endometrial thickness of 3 mm or less has the greatest sensitivity to exclude endometrial carcinoma, although using a cutoff of 4 mm or less may be more cost-effective.44–46 A woman with post-menopausal bleeding has a pretest probability of 10% for endometrial carcinoma.44,45 An endometrial thickness of 4 mm or less reduces the posttest probability to 1.2%, and a thickness of 3 mm or less reduces the posttest probability to 0.7%.44,45 Studies in premenopausal women found no carcinoma with thicknesses ranging from 4 to 8 mm. Current guidelines for the evaluation of premenopausal women recommend a cutoff of 5 mm or less46–48 (Figure 4).
Figure 4.
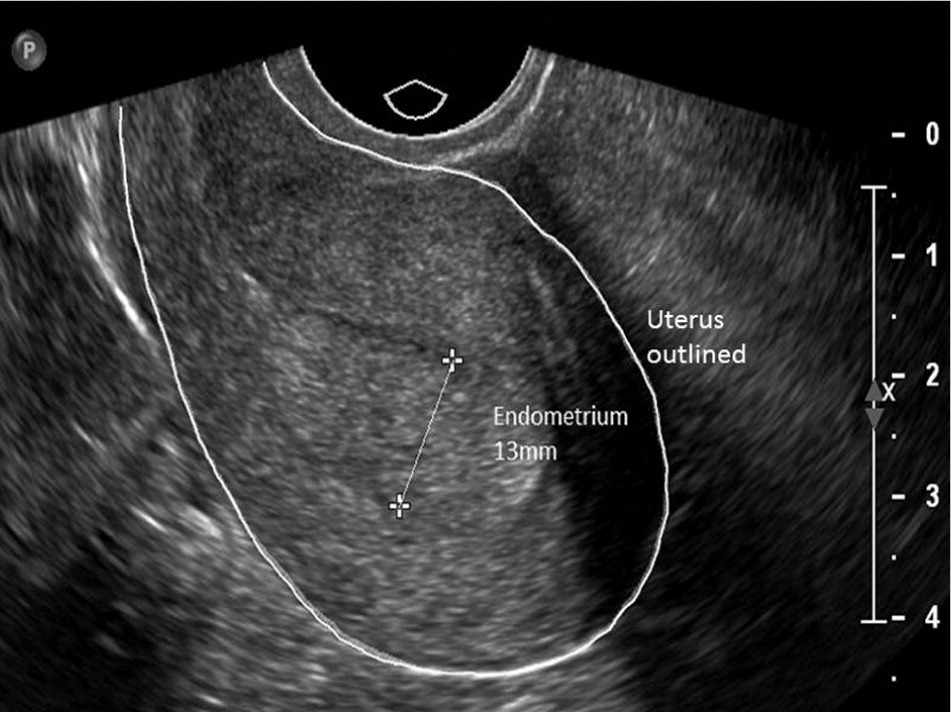
Transvaginal ultrasonography of a 22-year-old obese woman with atypical endometrial hyperplasia on endometrial biopsy. Imaging shows a thickened endometrium of 13 mm (calipers). Uterus is outlined in white.
Women whose endometrial thickness does not vary (e.g., postmenopausal women not taking hormone therapy, postmenopausal women taking continuous combined hormone therapy, premenopausal women taking oral contraceptives) can undergo transvaginal ultrasonography any time.46 For women with cyclic bleeding (e.g., premenopausal women not taking hormone therapy, postmenopausal women taking sequential hormone therapy), transvaginal ultrasonography should be performed at the end of menses, when the endometrium is thinnest.46
Incomplete or insufficient imaging can occur in women with previous uterine surgery (e.g., myomectomy, endometrial ablation), fibroids, obesity, or an axial uterus.49,50 About 10% of perimenopausal women need an additional evaluation because an adequate endometrial measurement cannot be obtained.47 Also, benign endometrial pathology can be missed on transvaginal ultrasonography even when the endometrial thickness is less than 3 mm.51
SALINE INFUSION SONOHYSTEROSCOPY
Saline infusion sonohysteroscopy is an office procedure that causes minimal discomfort without the risks of hysteroscopy.46 Infusing saline into the uterine cavity before ultrasonography gives definition to the endometrial structures. It can differentiate normal anatomic findings, globally thickened endometrium amenable to endometrial biopsy, and focal abnormalities best assessed by hysteroscopy.46,52 Saline infusion sonohysteroscopy cannot be performed if there is significant cervical stenosis, if the uterus cannot be distended with fluid, or if the endometrium otherwise cannot be visualized.46,47,52 Because of the high correlation of filling defects with carcinoma, additional evaluation should be performed if the uterus is unable to fill with fluid.52
DIAGNOSTIC HYSTEROSCOPY
Diagnostic hysteroscopy can be performed in the office, often without sedation, whereas operative hysteroscopy is usually performed in the operating room under anesthesia. Diagnostic hysteroscopy with directed biopsy has been found to be the most sensitive and specific method of diagnosing endometrial carcinoma, other than hysterectomy.53,54 A meta-analysis of diagnostic hysteroscopy demonstrated a low rate of serious adverse events (e.g., vasovagal collapse, creation of false tracts, uterine perforation).53 Diagnostic hysteroscopy and saline infusion sonohysteroscopy are accurate and safe, equally invasive, but less cost-effective than endometrial biopsy.46,53,55
DIAGNOSTIC APPROACH
Transvaginal ultrasonography is the initial preferred test,45 although endometrial biopsy is also an option.49 If endometrial biopsy histology is benign and symptoms persist, imaging should be considered because of the false-negative rate if focal lesions are present.42,43,46 The cutoff for endometrial thickness on transvaginal ultrasonography is less than 3 to 4 mm for postmenopausal women and less than 5 mm for premenopausal women.44–46,48 Saline infusion sonohysteroscopy should be considered if the endometrial thickness is greater than the threshold or an adequate measurement cannot be obtained.46,51,52 If saline infusion sonohysteroscopy demonstrates no focal abnormalities, carcinoma is likely excluded.46,52 However, if it shows a global process, a histologic diagnosis can usually be obtained with endometrial biopsy.46 If a focal endometrial lesion is present, hysteroscopy should be considered as the next diagnostic step.45,46,53 Table 2 summarizes endometrial assessment.41–47,49,51–54
Table 2. Methods of Endometrial Assessment
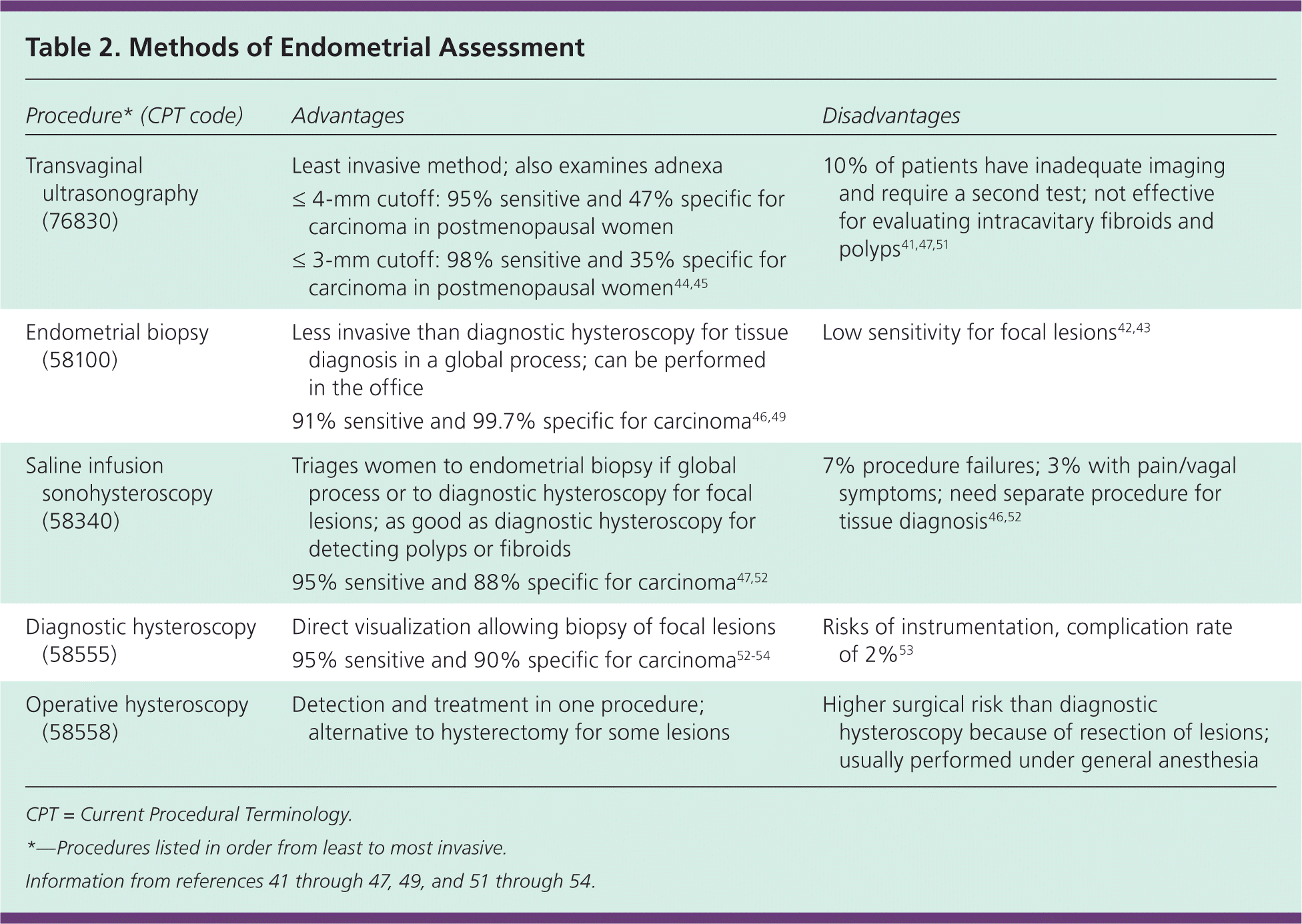
| Procedure* (CPT code) | Advantages | Disadvantages |
|---|---|---|
| Transvaginal ultrasonography (76830) | 10% of patients have inadequate imaging and require a second test; not effective for evaluating intracavitary fibroids and polyps41,47,51 | |
| Endometrial biopsy (58100) | Low sensitivity for focal lesions42,43 | |
| Saline infusion sonohysteroscopy (58340) | 7% procedure failures; 3% with pain/vagal symptoms; need separate procedure for tissue diagnosis46,52 | |
| Diagnostic hysteroscopy (58555) | Risks of instrumentation, complication rate of 2%53 | |
| Operative hysteroscopy (58558) |
| Higher surgical risk than diagnostic hysteroscopy because of resection of lesions; usually performed under general anesthesia |
CPT = Current Procedural Terminology.
*—Procedures listed in order from least to most invasive.
Information from references 41 through 47, 49, and 51 through 54.
Data Sources: A PubMed search was completed in Clinical Queries using the key terms cervical intraepithelial neoplasia, colposcopy, LEEP, endometrium, postmenopausal bleeding, and abnormal uterine bleeding. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Additional searches included National Guideline Clearinghouse, the Cochrane database, and the U.S. Preventive Services Task Force. Search date: October 1, 2011.

