Atypical moles are benign pigmented lesions. Although they are benign, they exhibit some of the clinical and histologic features of malignant melanoma. They are more common in fair-skinned individuals and in those with high sun exposure. Atypical moles are characterized by size of 6 mm or more at the greatest dimension, color variegation, border irregularity, and pebbled texture. They are associated with an increased risk of melanoma, warranting enhanced surveillance, especially in patients with more than 50 moles and a family history of melanoma. Because an individual lesion is unlikely to display malignant transformation, biopsy of all atypical moles is neither clinically beneficial nor cost-effective. The ABCDE (asymmetry, border irregularity, color unevenness, diameter of 6 mm or more, evolution) mnemonic is a valuable tool for clinicians and patients to identify lesions that could be melanoma. Also, according to the “ugly duckling” concept, benign moles tend to have a similar appearance, whereas an outlier with a different appearance is more likely to be undergoing malignant change. Atypical moles with changes suggestive of malignant melanoma should be biopsied, using an excisional method, if possible.
Atypical moles are pigmented lesions that, although benign, exhibit some of the clinical and histologic features of malignant melanoma. Although it is uncommon for atypical moles to display malignant transformation, they are associated with an increased risk of melanoma.
SORT: KEY RECOMMENDATIONS FOR PRACTICE

| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Because of the increased risk of melanoma, patients with atypical moles should be screened for melanoma, typically yearly, although the optimal methods and timing have not been determined. | C | 26 |
| Biopsy of all atypical moles is neither clinically valuable nor cost-effective. | C | 28 |
| Total excision of atypical moles with narrow margins is the preferable biopsy method when feasible. | C | 29 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
In the 1970s, a relationship between familial clusters of characteristic pigmented lesions and melanoma was reported. These moles may be sporadic or inherited. Although multiple names have been used to describe this type of mole and associated familial syndromes, including Clark nevus, dysplastic nevus, and familial atypical multiple-mole melanoma (FAMMM) syndrome, the National Institutes of Health recommends using the term atypical mole for those that occur sporadically and FAMMM for the most common familial cluster.1 However, the term dysplastic nevus continues to be used to describe an atypical mole.
FAMMM syndrome, the most well-known genetic mechanism, is characterized by the presence of more than 50 combined common and atypical moles plus a first- or second-degree relative with melanoma. It is transmitted in an autosomal dominant fashion.2
Epidemiology
The reported prevalence of atypical moles varies widely, in part depending on whether the diagnosis is clinical or histologic. They are more common in fair-skinned individuals and in those with high sun exposure. Both common and atypical moles decrease in number with age, starting in the 20s, although atypical moles continue to appear throughout life. Although the prevalence is higher in males, this is largely attributable to gender differences in ultraviolet radiation exposure. With those caveats in mind, a prevalence of 2% to 8% in fair-skinned persons is a valid estimate.3,4 In persons with skin of color, the prevalence of atypical moles is significantly lower (5% to 21% of the fair-skinned rate). They more commonly appear in non–sun-exposed areas and often go unnoticed by patients and clinicians, leading to delayed diagnosis and a poorer prognosis.5,6
Risk of Melanoma
A meta-analysis of case-control studies found that the relative risk of melanoma is 1.45 in patients with one atypical mole vs. none, and this increases to 6.36 in those with five atypical moles.7
Persons with FAMMM syndrome have a 10-year risk of melanoma of 10.7%, which is 17.3 times higher than in those without the syndrome. The lifetime risk in patients with FAMMM syndrome approaches 100%.8,9 Followed over five years, one-half of atypical moles remained unchanged, 15% developed increasing atypia, and 35% regressed or disappeared.8,10,11 Studies have reported a 0.5% to 46% rate of progression to melanoma, with this wide range attributable to differing diagnostic criteria.8,11 Still, the key message to convey to patients is that although most atypical moles do not become melanoma, patients with a high number of atypical moles have an increased lifetime risk of melanoma.3,12 Thus, it is important to be equally vigilant for changes in existing moles and new lesions.
Clinical Characteristics
Atypical moles present a diagnostic challenge because they often appear on a background of numerous common moles and, by definition, they overlap somewhat in appearance with melanomas. Figures 1 through 5 illustrate various presentations of atypical moles and related lesions. Characteristics of atypical moles are listed in Table 1.8,10,12,13
Figure 1.
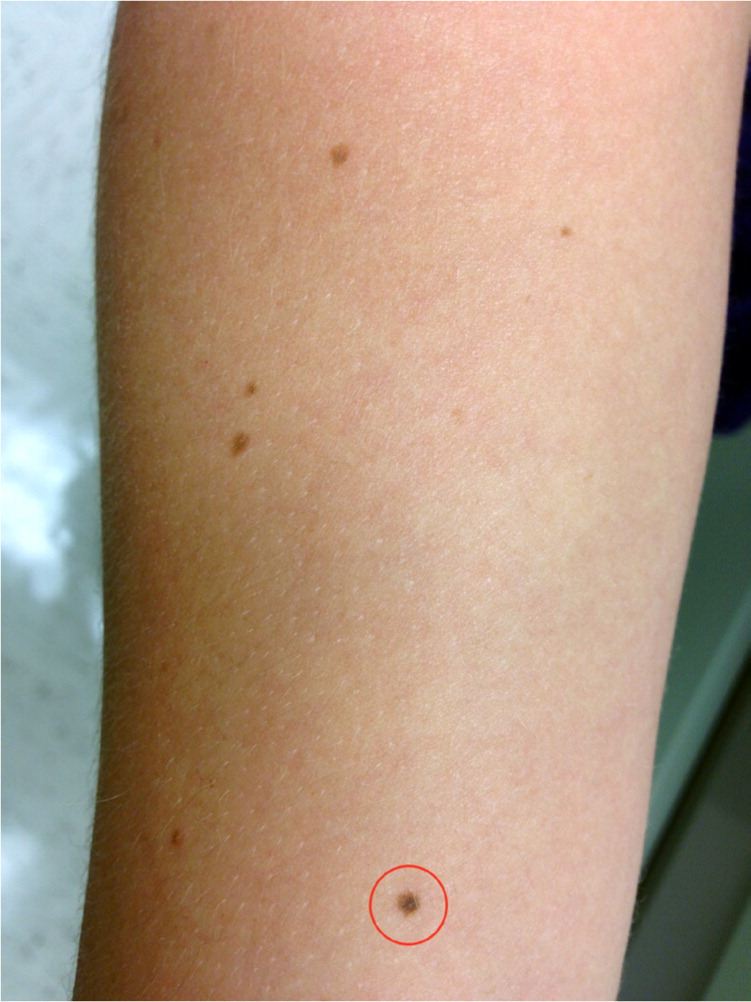
Common and atypical moles on the forearm of a young adult. Because of color variegation and border irregularity, the circled lesion is considered atypical despite a size of 4 mm.
Figure 2.
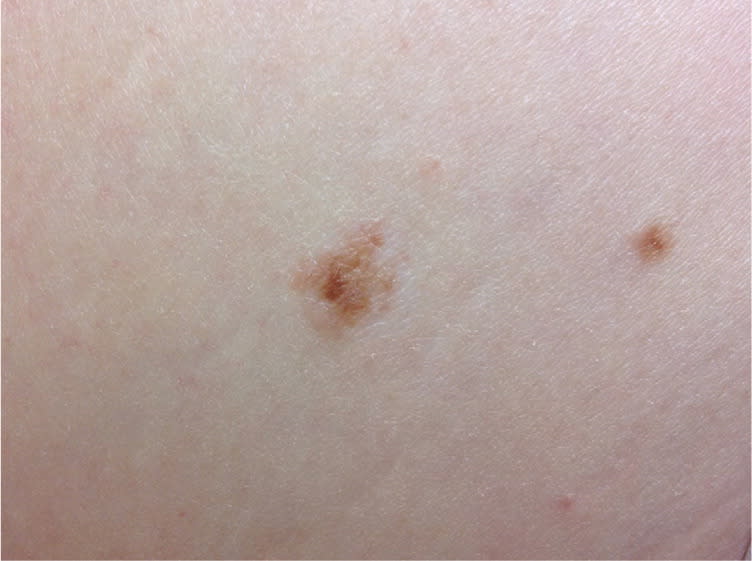
Atypical moles on the abdomen of an adult. The central lesion is 6 mm and displays color variegation and border irregularity. The smaller lesion also has some variable color.
Figure 3.
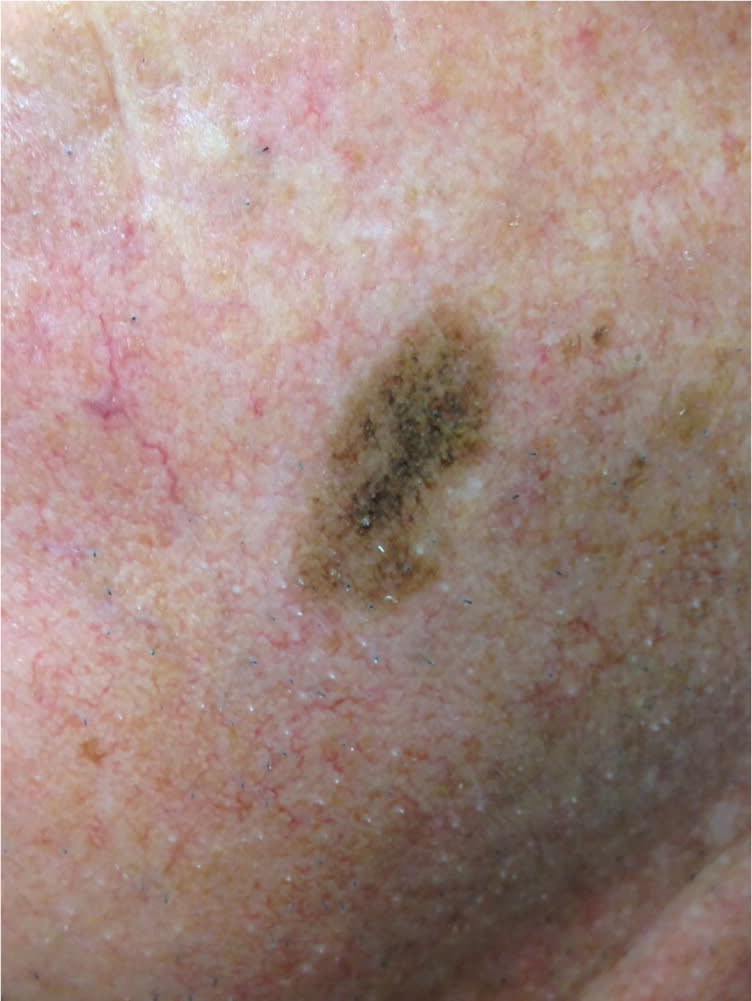
Atypical mole, 18 mm in size, on the face of a middle-aged adult. This is clinically indistinguishable from a lentigo maligna or lentigo maligna melanoma.
Figure 4.
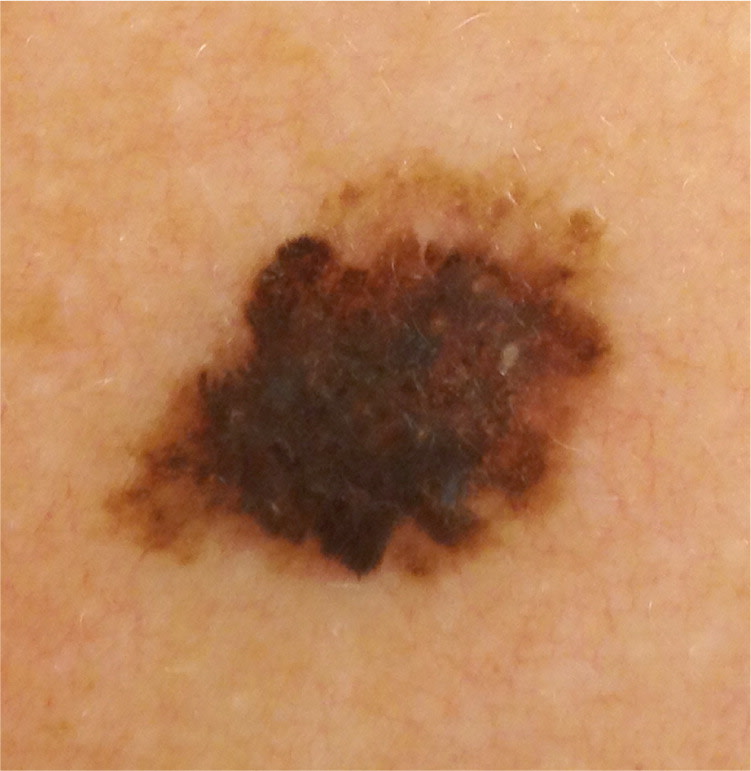
Atypical mole, 16 mm in size, on the upper back of an adult. The size, color variegation, border irregularity, and early nodularity prompt suspicion for melanoma.
Figure 5.
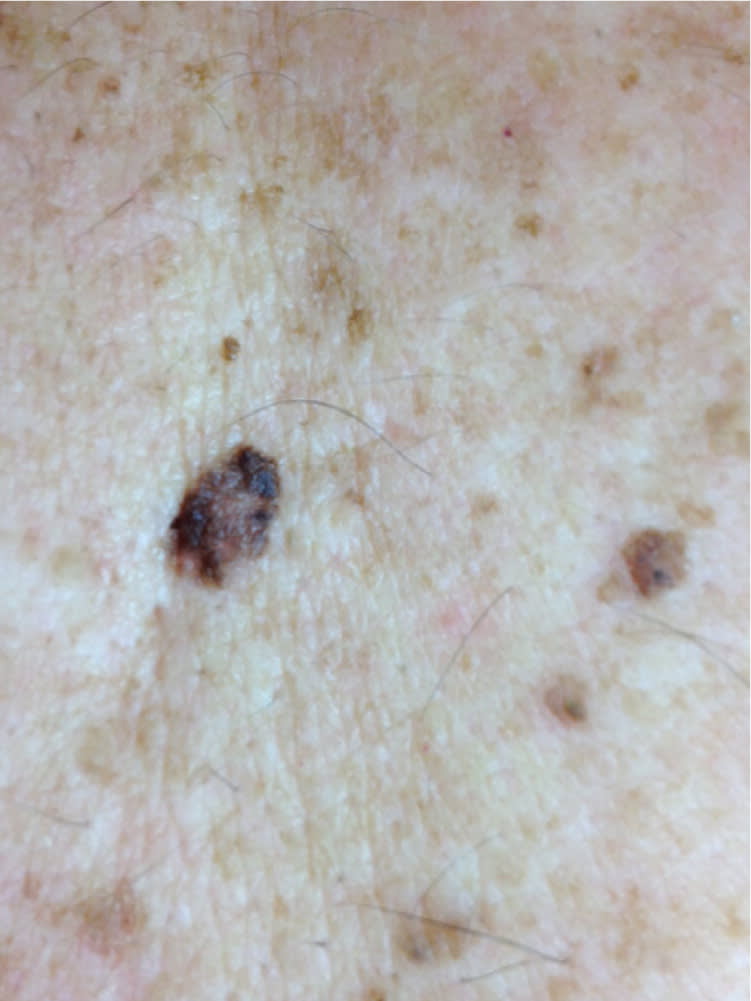
Melanoma, 8 mm in size, on the upper back of an adult. It is on a background of atypical moles, seborrheic keratoses, and freckles.
Table 1. Characteristics of Normal Moles, Atypical Moles, and Melanomas

| Lesion | Age at onset | Location | Number | Size | Color | Morphology |
|---|---|---|---|---|---|---|
| Normal moles | After 6 months, usually by 20 years of age | Anywhere | Few to hundreds | Usually < 6 mm | Evenly distributed, with only 1 or 2 shades of brown | Round, oval, symmetrical, with smooth and well-demarcated borders |
| Atypical moles | After 6 months, usually by 20 years of age | Anywhere, but most common on the trunk, especially the back | 1 to hundreds | Usually ≥ 6 mm, although they may be smaller | Variegated, with more than 2 shades of color, most commonly brown or tan, but possibly including pink or black | Round, oval, asymmetrical, with pebbled surface and irregular or poorly demarcated borders |
| Melanoma | Usually adulthood, may occur in children with giant congenital moles or atypical mole syndromes | Anywhere, including sun-protected areas; most common on the trunk in men and legs in women | 1 | Usually ≥ 6 mm, although they may be smaller | Variegated, with more than 2 shades of color, often very dark brown to black, that may have changed over time | Asymmetrical, with irregular or poorly demarcated borders |
Adapted with permission from Cyr PR. Atypical moles. Am Fam Physician. 2008;78(6):737, with additional information from references 10, 12, and 13.
Identifying atypical moles can be difficult because a mole exhibiting few or no findings may have dysplastic changes on microscopic examination, whereas a lesion with a worrisome appearance may be histologically benign. The agreement between the clinical diagnosis of an atypical mole and the pathologist's interpretation of dysplasia has been reported as low as 60%.12
Because the primary significance of atypical moles is their association with melanomas, the ABCDE (asymmetry, border irregularity, color unevenness, diameter of 6 mm or more, evolution) mnemonic13 is a valuable tool for clinicians and patients to identify suspicious lesions. Examination using this mnemonic is considered positive if one or more of the items are present. If the examination is completed by an experienced clinician, the sensitivity and specificity for the detection of melanoma are as high as 96% and 98%, respectively.13–15 However, the sensitivity and specificity of each item alone vary widely, creating the possibility of overdiagnosis or underdiagnosis.
Another weakness of the ABCDE mnemonic is that many common benign lesions, such as seborrheic keratoses, lentigines, and warts, will screen positive for possible melanoma. Although physicians may readily recognize these benign lesions, patients often cannot make the distinction. One approach to address this is to add the “ugly duckling” concept when observing persons with multiple moles. On a given patient, benign moles tend to have an overall similar appearance, whereas an outlier with a different appearance is more likely to be undergoing malignant change. Limited evidence suggests that observing for these outliers has 100% sensitivity for melanoma detection among dermatologists, with relatively high sensitivity (85%) and specificity (83%) among non-clinical office staff.16
Prevention
Exposure to ultraviolet radiation increases the risk of atypical moles and melanoma. Fair-skinned children who use sunscreen are less likely to develop moles,17 and adults who use sunscreen have a 50% lower incidence of melanoma over 10 years.18,19 Indoor tanning raises melanoma risk, with a dose-response effect related to early age of use and total exposure. The International Agency for Research on Cancer has named tanning beds as known carcinogens.20 The U.S. Preventive Services Task Force recommends counseling patients 10 to 24 years of age who have fair skin to minimize ultraviolet radiation exposure to reduce their risk of skin cancer; it found insufficient evidence regarding the benefits of behavioral counseling in older patients.21
Screening and Surveillance
The U.S. Preventive Services Task Force has found insufficient evidence to assess the balance of benefits and harms of routine screening for skin cancer by clinicians or patients, but acknowledges that screening in high-risk populations may have value.22 In the absence of prospective controlled studies comparing surveillance strategies, experts recommend individual risk stratification based on the patient's personal history of previous neoplasm, family history (especially FAMMM syndrome), number of moles, ABCDE findings, available screening tools, and examiner expertise. Of these factors, personal and family histories of melanoma are the most important.10,12–14 Therefore, it is reasonable to suggest an evaluation of family members of a patient who has a large number of moles indicative of a hereditary syndrome.
Although periodic self-examinations and physician examinations may increase detection of thin melanomas amenable to surgery, it may be that close surveillance detects more slow-growing lesions with an inherently favorable prognosis.10,13,14,22–25 Consequently, the optimal timing for follow-up examinations has not been determined. More than one-half of dermatologists recommend annual screening for patients with atypical moles, and 30% recommend screening every six months.26 Studies of more frequent examinations have not demonstrated improved diagnosis or prognosis.14,26 The yield of melanomas in screening examinations will obviously be higher in patients with familial syndromes that convey high cumulative lifetime risk. The evidence for improved survival is less compelling.8,10 It is often recommended to include ophthalmologic examination in these patients; however, in the absence of ocular symptoms, there is limited evidence that this practice is useful.14
Total body photographs, with copies given to the patient, may be considered for observational aid and reassurance, especially in patients with a large number and variety of moles. There is some evidence to suggest an increased melanoma yield based on photographic change in lesions that were not strongly suspected to be melanoma by appearance alone.13,14 Magnified evaluation of atypical moles with dermoscopy can be helpful in identifying lesions that merit biopsy, although outcomes are dependent on the skill of the operator.13,25,27 A recent review of dermoscopy published in American Family Physician is available at https://www.aafp.org/afp/2013/1001/p441.html.
Biopsy
Although atypical moles are associated with an increased risk of melanoma, most melanomas do not arise from existing atypical moles, and this should guide biopsy decisions. A strategy of photographic and physical follow-up, for example, results in a reasonable ratio of 10 biopsies per melanoma discovered. In contrast, a policy of biopsying every atypical mole would require nearly 2,000 biopsies per melanoma diagnosed, while failing to diagnose the 85% of melanomas that did not arise from an atypical mole and leading to an untenable cost-benefit ratio.13,14,28
The primary goal of biopsy is to rule out melanoma in patients who develop suspicious lesions during surveillance. A secondary goal is to accurately determine the depth of penetration should a melanoma be found because this is the histologic factor most predictive of metastasis and survival. Biopsy techniques include excisional, punch, deep shave (scoop, scallop, or saucerization), and superficial shave biopsies. Full epidermal and dermal excisional biopsy, including the entire lesion with at least 4 mm in depth and a narrow 2-mm margin, is the preferred method because it provides the pathologist with the entire specimen, eliminating sampling variability.29 Punch biopsy also provides a cylindrical specimen of full thickness, but it leaves open the possibility of not sampling the area of greatest depth or of missing the area of melanoma entirely.
Clinicians have traditionally been advised against performing shave biopsy of skin lesions when there is significant suspicion of melanoma, out of concern that this technique could make accurate determination of lesion thickness difficult and that scarring could obscure the development of a recurrent lesion. However, studies have shown that a deep shave biopsy performed by an experienced clinician using the saucerization technique can usually achieve a depth that allows a valid appraisal of penetration depth, and it may even be preferable to punch biopsy when melanoma makes up only a small portion of a larger lesion. Overall, saucerization and punch biopsies lead to accurate diagnosis and thickness determination 88% of the time compared with later definitive excision, and are accepted practice.29,30
When melanoma is ruled out in a biopsied mole that displays atypia extending to or very near the margin of the excision, it is common practice to reexcise the lesion. This is likely not necessary because clinical recurrence of lesions with mild to moderate atypia extending to the margin is extremely rare on prolonged follow-up. Lesions with moderate to severe atypia may benefit from reexcision.31–33
Data Sources: A PubMed search was completed in Clinical Queries using the key terms atypical moles, atypical mole syndrome, Clark nevus, dysplastic nevus, FAMMM, FAMMM syndrome, and melanoma screening. Also searched were Essential Evidence Plus, the National Guideline Clearinghouse, UpToDate, and the U.S. Preventive Services Task Force. Search dates: July 6, 2014, and January 29, 2015.

