Am Fam Physician. 2017;95(2):66-69
Related U.S. Preventive Services Task Force recommendation statement: Statin Use for the Primary Prevention of Cardiovascular Disease in Adults: Recommendation Statement
Author disclosure: No relevant financial affiliations.
This issue of American Family Physician includes a review of patient-centered evidence supporting the use of lipid-lowering therapy for the prevention of atherosclerotic cardiovascular disease (ASCVD).1 An important basis of the review is the 2013 American College of Cardiology (ACC) and American Heart Association (AHA) cholesterol guideline.2 This guideline's unique approach was to shift away from the practice of treating lipid targets to specific goals (i.e., for low-density lipoprotein cholesterol [LDL-C] and non–high-density lipoprotein cholesterol [non–HDL-C]). Instead, the guideline recommended moderate- to high-intensity statin therapy for four groups of patients most likely to achieve maximal net benefit based on their increased risk.
These statin benefit groups have been identified based on rigorous analysis of data from randomized controlled trials (RCTs) for primary (groups 2, 3, and 4) and secondary (group 1) prevention of ASCVD. The four groups consist of: (1) adults with clinical ASCVD; (2) adults 21 years or older with primary LDL-C elevations of 190 mg per dL (4.92 mmol per L) or greater; (3) adults 40 to 75 years of age without ASCVD but with diabetes mellitus and LDL-C of 70 to 189 mg per dL (1.81 to 4.90 mmol per L); and (4) adults 40 to 75 years of age without ASCVD or diabetes but with LDL-C of 70 to 189 mg per dL and an estimated 10-year ASCVD risk of 7.5% or greater (using the Pooled Cohort Equations).
The 2013 ACC/AHA guideline was purposely developed using only RCTs and meta-analysis of these trials.2 The guideline became statin-centric because most high-quality RCTs were statin trials. Although no trials were designed to treat to specific LDL-C goals, their secondary analyses, as well as epidemiologic data, show a linear relationship between LDL-C levels and ASCVD events.3 A recent stratified propensity score analysis of a large cohort of adults taking statins showed that statin intensity was not a significant predictor of major adverse cardiovascular events. However, achieving a reduction in LDL-C levels was a predictor.4 These results support the view that LDL-C levels are important, and LDL-C titration strategies merit further investigation in RCTs.
Statins are powerful, well-studied LDL-C–lowering agents.3 However, emerging RCT data suggest that lowering LDL-C levels with nonstatins reduces ASCVD risk in proportion with LDL-C reduction, as demonstrated with ezetimibe (Zetia) in the IMPROVE-IT trial.5 Like statins, ezetimibe has pleiotropic effects, leading to a decrease in inflammatory markers.6 The LDL-C–lowering effect of ezetimibe (a reduction of approximately 20%) is dwarfed by that of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors, which are injectable monoclonal antibodies that can lower LDL-C levels by 50% to 80%.7–11 Post hoc analysis of PCSK9-inhibitor trials demonstrated an additional 48% to 53% reduction in cardiovascular events.12,13 Major trials of PCSK9 inhibitors for ASCVD outcomes are underway. This emerging evidence has prompted the development of the 2016 ACC Expert Consensus Decision Pathway (ECDP) on the use of nonstatin therapy.14
Two PCSK9 inhibitors (evolocumab [Repatha] and ali-rocumab [Praluent]) are approved by the U.S. Food and Drug Administration (FDA) for use in addition to diet and maximally tolerated statin therapy in adults with heterozygous familial hypercholesterolemia or clinical ASCVD who require additional reductions in LDL-C.15,16 Evolocumab is also approved to treat patients with homozygous familial hypercholesterolemia.15 The wording in the indication raises the question of what constitutes additional lowering of LDL-C when no goals for LDL-C were provided in the 2013 ACC/AHA guideline.2 Such goals must be defined, because indiscriminate use of expensive monoclonal antibodies (i.e., the current cost of therapy is approximately $14,000 per year) for primary and secondary prevention of ASCVD will have a deep impact on health care costs.17,18
The 2016 ACC ECDP has reintroduced optional lipid goals, thus helping with the decision to initiate nonstatins in patients who are already taking statin therapy or who are intolerant of appropriate statin doses. An overview of these recommendations is provided in Table 1 based on the algorithms presented in the ECDP report.2,14
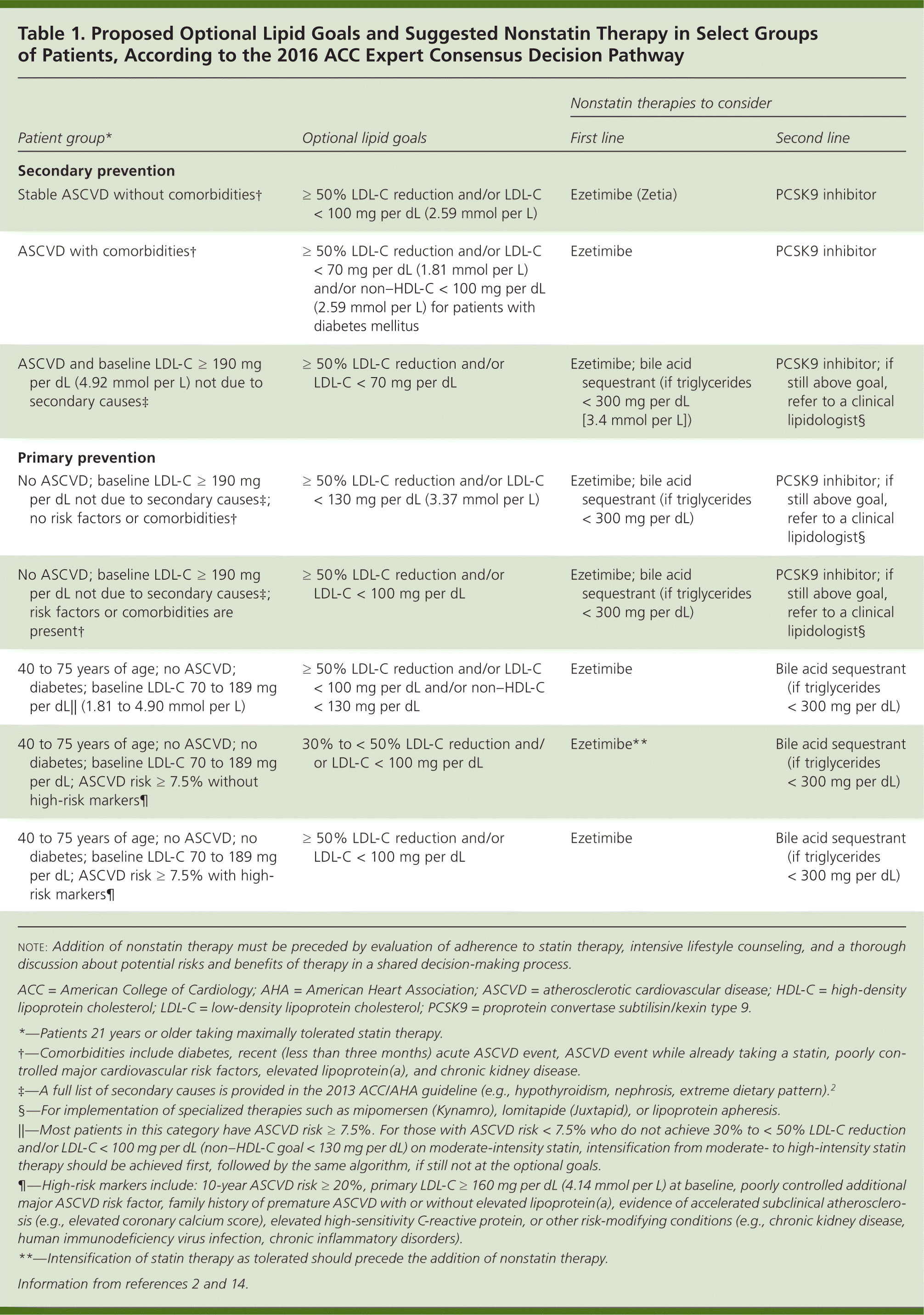
| Nonstatin therapies to consider | |||
|---|---|---|---|
| Patient group* | Optional lipid goals | First line | Second line |
| Secondary prevention | |||
| Stable ASCVD without comorbidities† | ≥ 50% LDL-C reduction and/or LDL-C < 100 mg per dL (2.59 mmol per L) | Ezetimibe (Zetia) | PCSK9 inhibitor |
| ASCVD with comorbidities† | ≥ 50% LDL-C reduction and/or LDL-C < 70 mg per dL (1.81 mmol per L) and/or non–HDL-C < 100 mg per dL (2.59 mmol per L) for patients with diabetes mellitus | Ezetimibe | PCSK9 inhibitor |
| ASCVD and baseline LDL-C ≥ 190 mg per dL (4.92 mmol per L) not due to secondary causes‡ | ≥ 50% LDL-C reduction and/or LDL-C < 70 mg per dL | Ezetimibe; bile acid sequestrant (if triglycerides < 300 mg per dL [3.4 mmol per L]) | PCSK9 inhibitor; if still above goal, refer to a clinical lipidologist§ |
| Primary prevention | |||
| No ASCVD; baseline LDL-C ≥ 190 mg per dL not due to secondary causes‡; no risk factors or comorbidities† | ≥ 50% LDL-C reduction and/or LDL-C < 130 mg per dL (3.37 mmol per L) | Ezetimibe; bile acid sequestrant (if triglycerides < 300 mg per dL) | PCSK9 inhibitor; if still above goal, refer to a clinical lipidologist§ |
| No ASCVD; baseline LDL-C ≥ 190 mg per dL not due to secondary causes‡; risk factors or comorbidities are present† | ≥ 50% LDL-C reduction and/or LDL-C < 100 mg per dL | Ezetimibe; bile acid sequestrant (if triglycerides < 300 mg per dL) | PCSK9 inhibitor; if still above goal, refer to a clinical lipidologist§ |
| 40 to 75 years of age; no ASCVD; diabetes; baseline LDL-C 70 to 189 mg per dL|| (1.81 to 4.90 mmol per L) | ≥ 50% LDL-C reduction and/or LDL-C < 100 mg per dL and/or non–HDL-C < 130 mg per dL | Ezetimibe | Bile acid sequestrant (if triglycerides < 300 mg per dL) |
| 40 to 75 years of age; no ASCVD; no diabetes; baseline LDL-C 70 to 189 mg per dL; ASCVD risk ≥ 7.5% without high-risk markers¶ | 30% to < 50% LDL-C reduction and/or LDL-C < 100 mg per dL | Ezetimibe** | Bile acid sequestrant (if triglycerides < 300 mg per dL) |
| 40 to 75 years of age; no ASCVD; no diabetes; baseline LDL-C 70 to 189 mg per dL; ASCVD risk ≥ 7.5% with high-risk markers¶ | ≥ 50% LDL-C reduction and/or LDL-C < 100 mg per dL | Ezetimibe | Bile acid sequestrant (if triglycerides < 300 mg per dL) |
Although the ECDP supports the use of ezetimibe, bile acid sequestrants, and PCSK9 inhibitors in select groups of patients, the document argues against the use of niacin for ASCVD prevention. Recent RCTs studying the effects of niacin in combination with statins did not show a reduction in ASCVD risk, although it did show a potential increase in adverse events.19,20 This led to the withdrawal of FDA approval of niacin for cardiovascular risk reduction. Still, niacin remains a “natural” alternative for some patients, because it has demonstrated cardiovascular benefit as a stand-alone therapy.21 Fibrates experienced a similar fate following the ACCORD trial,22 yet fibrates can also be beneficial in specific patient populations, as shown by subgroup analysis.23
The landscape of ASCVD prevention is changing rapidly. Recently, the U.S. Preventive Services Task Force (USPSTF) released its own guideline for statin use in primary prevention. Initiation of low- to moderate-intensity statin therapy is recommended in adults 40 to 75 years of age without a history of cardiovascular disease (CVD) who have one or more CVD risk factors and a calculated 10-year CVD risk of 10% or greater (grade B recommendation), as well as selectively in those with CVD risk of 7.5% to 10% (grade C recommendation). At the same time, the USPSTF found no evidence to recommend for or against statin therapy for primary prevention in patients older than 75 years (I statement).24
Statins will long remain the cornerstone of treatment because of their proven benefits and cost-effectiveness, especially in the four statin benefit groups.2 However, emerging RCT evidence shows that the degree of LDL-C lowering may be more important than the drug used to achieve it.4,6,11–13 The 2016 ACC ECDP offers a practical approach to escalate lipid-lowering therapy in addition to statins in the four benefit groups while also suggesting LDL-C and non–HDL-C goals in terms of absolute numbers or percentages of reduction from baseline.14 This approach will likely be recommended by new ACC/AHA guidelines to facilitate the selection of patients who would benefit from stepwise addition of nonstatin therapy. This is especially important considering the price of PCSK9 inhibitors, because their indiscriminate use may greatly increase health care costs.17,18