Key Clinical Issue
What is the best test for celiac disease in terms of sensitivity, specificity, and safety?
Evidence-Based Answer
Current evidence on the accuracy of tests used to diagnose celiac disease supports the excellent sensitivity and specificity of anti-tissue transglutaminase immunoglobulin A (tTG IgA) tests. The available evidence supports the current recommendation of the American College of Gastroenterology (ACG) to use tTG IgA as the first-line test for patients two years and older with suspected celiac disease. (Strength of Recommendation [SOR]: C, based on disease-oriented evidence.) Testing for deamidated gliadin peptide (DGP) IgA or IgG may be more accurate in children younger than two years. (SOR: C, based on disease-oriented evidence.) Human lymphocyte antigen (HLA) typing that reveals the absence of HLA-DQ2 and HLA-DQ8 essentially rules out celiac disease. (SOR: C, based on disease-oriented evidence.) Video capsule endoscopy and endoscopy with duodenal biopsy are invasive and associated with a small risk of adverse events. (SOR: B, based on inconsistent or limited-quality patient-oriented evidence.)
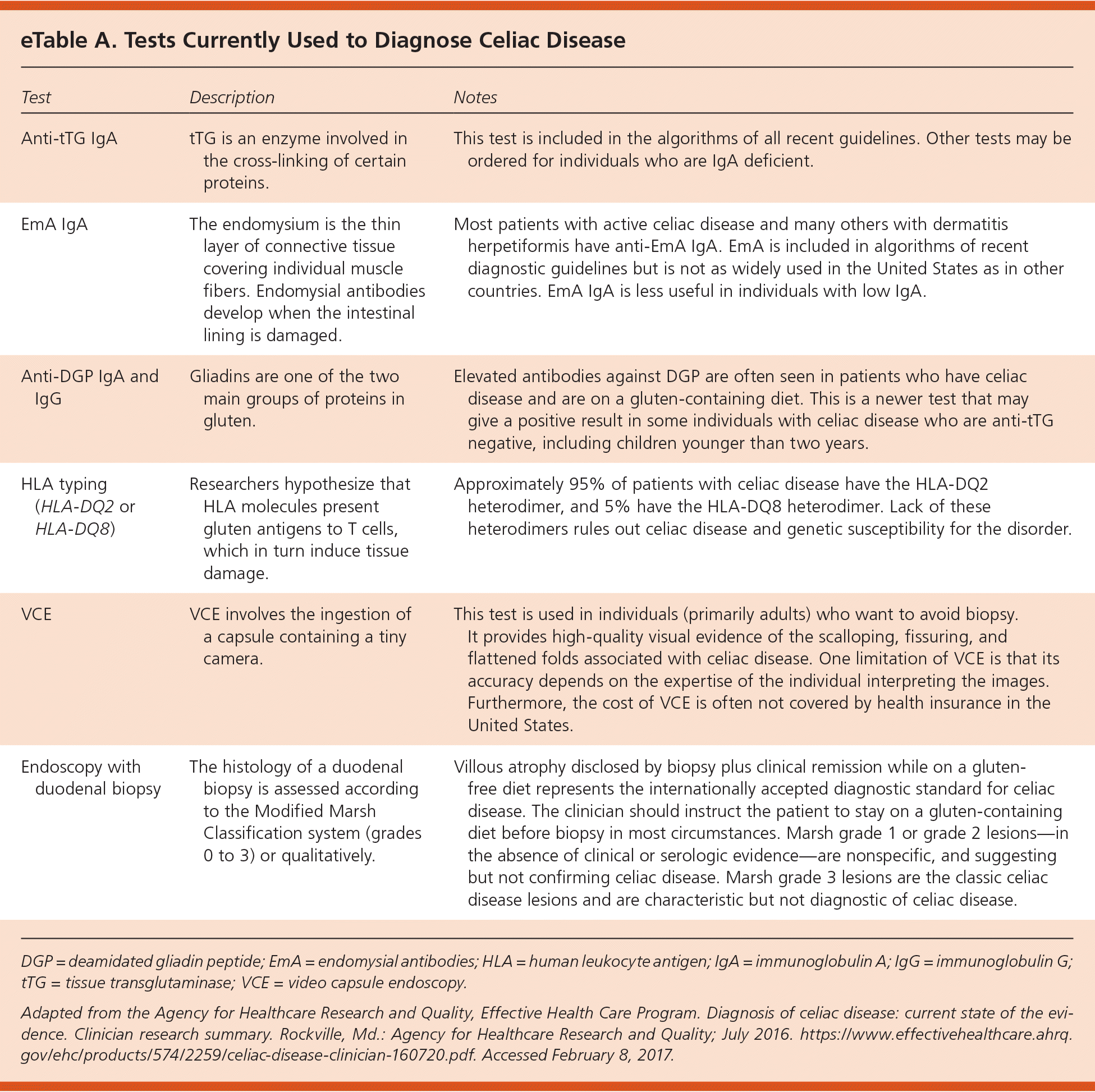
eTable A describes tests currently used to diagnose celiac disease.
eTable A. Tests Currently Used to Diagnose Celiac Disease
| Test | Description | Notes |
|---|---|---|
| Anti-tTG IgA | tTG is an enzyme involved in the cross-linking of certain proteins. | This test is included in the algorithms of all recent guidelines. Other tests may be ordered for individuals who are IgA deficient. |
| EmA IgA | The endomysium is the thin layer of connective tissue covering individual muscle fibers. Endomysial antibodies develop when the intestinal lining is damaged. | Most patients with active celiac disease and many others with dermatitis herpetiformis have anti-EmA IgA. EmA is included in algorithms of recent diagnostic guidelines but is not as widely used in the United States as in other countries. EmA IgA is less useful in individuals with low IgA. |
| Anti-DGP IgA and IgG | Gliadins are one of the two main groups of proteins in gluten. | Elevated antibodies against DGP are often seen in patients who have celiac disease and are on a gluten-containing diet. This is a newer test that may give a positive result in some individuals with celiac disease who are anti-tTG negative, including children younger than two years. |
| HLA typing (HLA-DQ2 or HLA-DQ8) | Researchers hypothesize that HLA molecules present gluten antigens to T cells, which in turn induce tissue damage. | Approximately 95% of patients with celiac disease have the HLA-DQ2 heterodimer, and 5% have the HLA-DQ8 heterodimer. Lack of these heterodimers rules out celiac disease and genetic susceptibility for the disorder. |
| VCE | VCE involves the ingestion of a capsule containing a tiny camera. | This test is used in individuals (primarily adults) who want to avoid biopsy. It provides high-quality visual evidence of the scalloping, fissuring, and flattened folds associated with celiac disease. One limitation of VCE is that its accuracy depends on the expertise of the individual interpreting the images. Furthermore, the cost of VCE is often not covered by health insurance in the United States. |
| Endoscopy with duodenal biopsy | The histology of a duodenal biopsy is assessed according to the Modified Marsh Classification system (grades 0 to 3) or qualitatively. | Villous atrophy disclosed by biopsy plus clinical remission while on a gluten-free diet represents the internationally accepted diagnostic standard for celiac disease. The clinician should instruct the patient to stay on a gluten-containing diet before biopsy in most circumstances. Marsh grade 1 or grade 2 lesions—in the absence of clinical or serologic evidence—are nonspecific, and suggesting but not confirming celiac disease. Marsh grade 3 lesions are the classic celiac disease lesions and are characteristic but not diagnostic of celiac disease. |
DGP = deamidated gliadin peptide; EmA = endomysial antibodies; HLA = human leukocyte antigen; IgA = immunoglobulin A; IgG = immunoglobulin G; tTG = tissue transglutaminase; VCE = video capsule endoscopy.
Adapted from the Agency for Healthcare Research and Quality, Effective Health Care Program. Diagnosis of celiac disease: current state of the evidence. Clinician research summary. Rockville, Md.: Agency for Healthcare Research and Quality; July 2016. https://www.effectivehealthcare.ahrq.gov/ehc/products/574/2259/celiac-disease-clinician-160720.pdf. Accessed February 8, 2017.
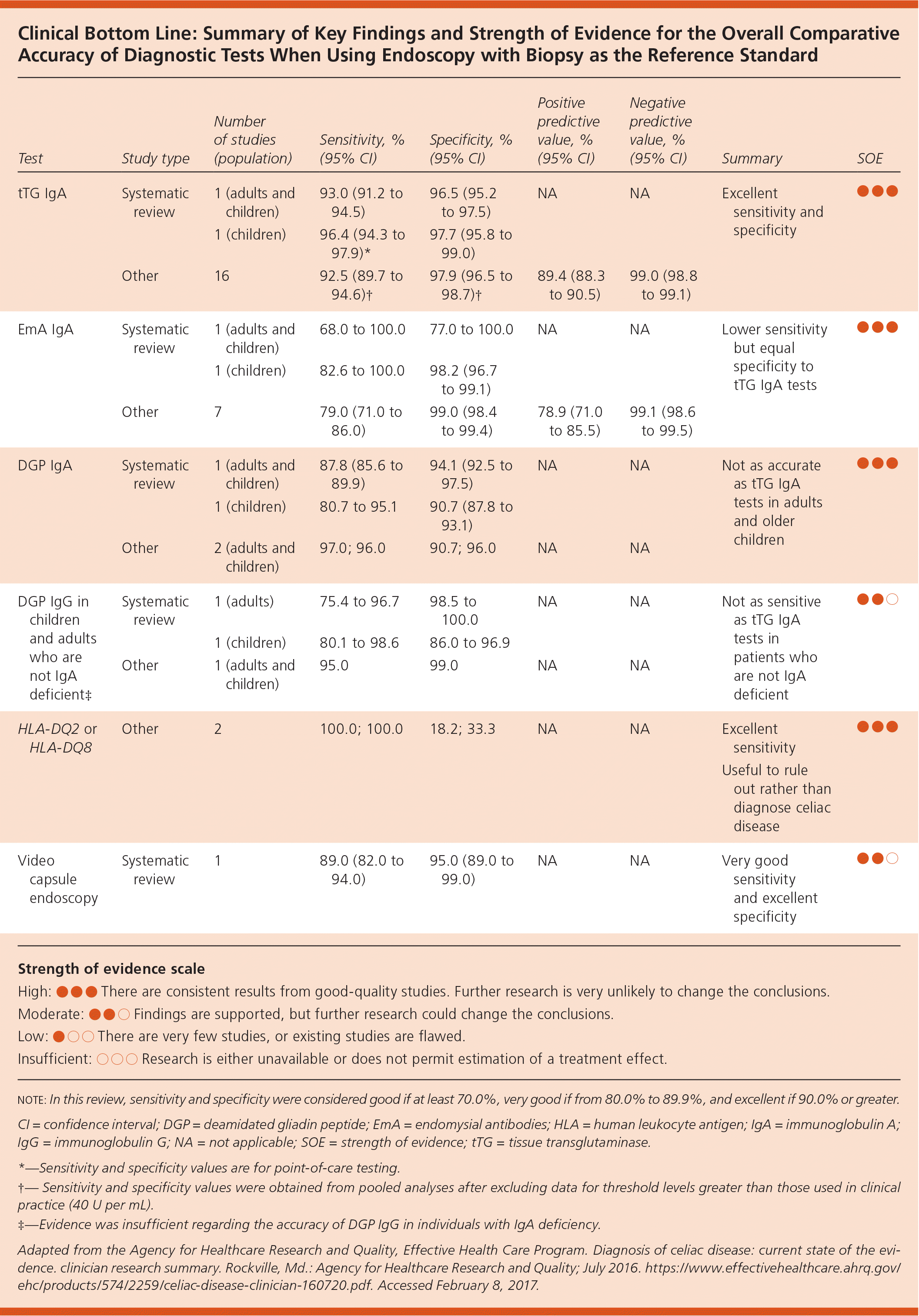
Clinical Bottom Line: Summary of Key Findings and Strength of Evidence for the Overall Comparative Accuracy of Diagnostic Tests When Using Endoscopy with Biopsy as the Reference Standard
| Test | Study type | Number of studies (population) | Sensitivity, % (95% CI) | Specificity, % (95% CI) | Positive predictive value, % (95% CI) | Negative predictive value, % (95% CI) | Summary | SOE |
|---|---|---|---|---|---|---|---|---|
| tTG IgA | Systematic review | 1 (adults and children) | 93.0 (91.2 to 94.5) | 96.5 (95.2 to 97.5) | NA | NA | Excellent sensitivity and specificity | ●●● |
| 1 (children) | 96.4 (94.3 to 97.9)* | 97.7 (95.8 to 99.0) | ||||||
| Other | 16 | 92.5 (89.7 to 94.6)† | 97.9 (96.5 to 98.7)† | 89.4 (88.3 to 90.5) | 99.0 (98.8 to 99.1) | |||
| EmA IgA | Systematic review | 1 (adults and children) | 68.0 to 100.0 | 77.0 to 100.0 | NA | NA | Lower sensitivity but equal specificity to tTG IgA tests | ●●● |
| 1 (children) | 82.6 to 100.0 | 98.2 (96.7 to 99.1) | ||||||
| Other | 7 | 79.0 (71.0 to 86.0) | 99.0 (98.4 to 99.4) | 78.9 (71.0 to 85.5) | 99.1 (98.6 to 99.5) | |||
| DGP IgA | Systematic review | 1 (adults and children) | 87.8 (85.6 to 89.9) | 94.1 (92.5 to 97.5) | NA | NA | Not as accurate as tTG IgA tests in adults and older children | ●●● |
| 1 (children) | 80.7 to 95.1 | 90.7 (87.8 to 93.1) | ||||||
| Other | 2 (adults and children) | 97.0; 96.0 | 90.7; 96.0 | NA | NA | |||
| DGP IgG in children and adults who are not IgA deficient‡ | Systematic review | 1 (adults) | 75.4 to 96.7 | 98.5 to 100.0 | NA | NA | Not as sensitive as tTG IgA tests in patients who are not IgA deficient | ●●○ |
| 1 (children) | 80.1 to 98.6 | 86.0 to 96.9 | ||||||
| Other | 1 (adults and children) | 95.0 | 99.0 | NA | NA | |||
| HLA-DQ2 or HLA-DQ8 | Other | 2 | 100.0; 100.0 | 18.2; 33.3 | NA | NA | Excellent sensitivity Useful to rule out rather than diagnose celiac disease | ●●● |
| Video capsule endoscopy | Systematic review | 1 | 89.0 (82.0 to 94.0) | 95.0 (89.0 to 99.0) | NA | NA | Very good sensitivity and excellent specificity | ●●○ |
Strength of evidence scale
High: ●●● There are consistent results from good-quality studies. Further research is very unlikely to change the conclusions.
Moderate: ●●○ Findings are supported, but further research could change the conclusions.
Low: ●○○ There are very few studies, or existing studies are flawed.
Insufficient: ○○○ Research is either unavailable or does not permit estimation of a treatment effect.
note: In this review, sensitivity and specificity were considered good if at least 70.0%, very good if from 80.0% to 89.9%, and excellent if 90.0% or greater.
CI = confidence interval; DGP = deamidated gliadin peptide; EmA = endomysial antibodies; HLA = human leukocyte antigen; IgA = immunoglobulin A; IgG = immunoglobulin G; NA = not applicable; SOE = strength of evidence; tTG = tissue transglutaminase.
*—Sensitivity and specificity values are for point-of-care testing.
†—Sensitivity and specificity values were obtained from pooled analyses after excluding data for threshold levels greater than those used in clinical practice (40 U per mL).
‡—Evidence was insufficient regarding the accuracy of DGP IgG in individuals with IgA deficiency.
Adapted from the Agency for Healthcare Research and Quality, Effective Health Care Program. Diagnosis of celiac disease: current state of the evidence. clinician research summary. Rockville, Md.: Agency for Healthcare Research and Quality; July 2016. https://www.effectivehealthcare.ahrq.gov/ehc/products/574/2259/celiac-disease-clinician-160720.pdf. Accessed February 8, 2017.
Practice Pointers
Celiac disease is an immune-mediated inflammatory condition that presents in genetically predisposed individuals after consumption of gluten-containing foods. Celiac disease affects about 1% of the population in the United States and Western Europe, most of whom are undiagnosed.1 The prevalence appears to be increasing, although the reason remains unclear. The presentation of celiac disease varies widely, making clinical suspicion an important key to recognition.2 The Agency for Healthcare Research and Quality review included 60 individual studies and 13 systematic reviews of the accuracy of multiple testing modalities. Only studies in which all patients underwent biopsy were included.3
Although endoscopy with duodenal biopsy is the definitive test for the diagnosis of celiac disease, there is evidence that accuracy is greater in academic centers than in community settings. Endoscopy is associated with a small risk of infection, and bleeding occurs in 1.6 per 1,000 patients. Video capsule endoscopy is an alternative to duodenal biopsy, and has very good sensitivity (89.0%) and excellent specificity (95%). However, it is associated with a capsule retention rate of 0.9% to 4.6%.3
The ACG recommends starting with tTG IgA testing in patients two years and older.4 tTG IgA testing has excellent sensitivity (more than 90%) and specificity (more than 95%), giving it a positive predictive value of 89.4% (95% confidence interval [CI], 88.3 to 90.5) and a negative predictive value of 99.0% (95% CI, 98.8 to 99.1). IgA deficiency is more common in patients with celiac disease and should be excluded if tTG IgA testing is used.2
For children younger than two years, the ACG recommends using DGP IgA and IgG tests initially.4 Limited evidence shows that DGP IgA and IgG are slightly less sensitive and specific in adults compared with tTG, but are more accurate in young children.
Endomysial IgA antibody testing has a lower sensitivity than tTG IgA (positive predictive value = 79%; 95% CI, 71.0 to 85.5) and equivalent specificity. It is included in several diagnostic algorithms but is not considered a first-line test in any patient population.
HLA typing can play a role in the diagnostic evaluation of patients with suspected celiac disease. Among patients with celiac disease, 95% have the HLA-DQ2 heterodimer, and the other 5% have the HLA-DQ8 heterodimer. This means the absence of both of these HLA heterodimers essentially rules out the disease.
Celiac disease is more common in certain populations, and screening has been recommended in patients with type 1 diabetes mellitus.5 The Agency for Healthcare Research and Quality review did not find enough evidence to recommend one testing strategy over another in any specific population, including persons with type 1 diabetes. Several diagnostic algorithms were evaluated (one suggested algorithm is available at https://www.aafp.org/afp/2014/0115/p99.html#afp20140115p99-f1). There was insufficient evidence to compare the relative accuracy of the various algorithms.
editor's note: AFP SOR ratings are different from the AHRQ Strength of Evidence (SOE) ratings.

