The U.S. Preventive Services Task Force (USPSTF) makes recommendations on more than 70 topics covering screening and primary prevention for primary care clinicians.1 It has 16 volunteer members, mostly primary care physicians, who are experts in screening, prevention, and evidence-based medicine. Recommendations are graded based on the level of certainty regarding the evidence and the magnitude of net benefit (i.e., benefit minus harm): “A” indicates high certainty of a substantial net benefit; “B” indicates at least moderate certainty of a moderate net benefit; “C” indicates at least moderate certainty of a small net benefit; and “D” indicates no benefit or net harm, meaning the preventive service is not recommended. An “I” statement indicates a low level of certainty in the evidence, so a recommendation on net benefit or harm cannot be made.
In 2016, the USPSTF updated 14 previous recommendation statements and added a new recommendation on screening for autism spectrum disorder (ASD). Table 1 summarizes the services with a recommendation of A, B, C, or D.1 Several are notable for having significant changes vs. previous recommendations. For example, the USPSTF now recommends that physicians offer any one of seven options for colorectal cancer screening: annual fecal immunochemical testing (FIT); colonoscopy every 10 years; FIT plus fecal DNA (Cologuard) every one to three years; computed tomographic colonography every five years; the combination of flexible sigmoidoscopy and FIT (i.e., flexible sigmoidoscopy every 10 years plus FIT every year); flexible sigmoidoscopy alone every five years, or annual guaiac-based fecal occult blood testing. These tests all have a substantial benefit (221 to 270 life-years gained per 1,000 persons2) but with varying costs and potential harms. For instance, annual FIT gains 245 life-years and results in a mean of 1.7 colonoscopies per person over the patient's lifetime, whereas colonoscopy every 10 years gains 270 life-years but results in more than twice as many lifetime colonoscopies and higher costs. Comparing annual FIT with FIT plus fecal DNA every three years shows a similar number of life-years gained and a similar number of lifetime colonoscopies, but the latter is much more expensive on a per-test basis (roughly $500 for FIT-DNA vs. $10 for FIT).
Table 1. Updated 2016 USPSTF Recommendations with a Grade of A, B, C or D
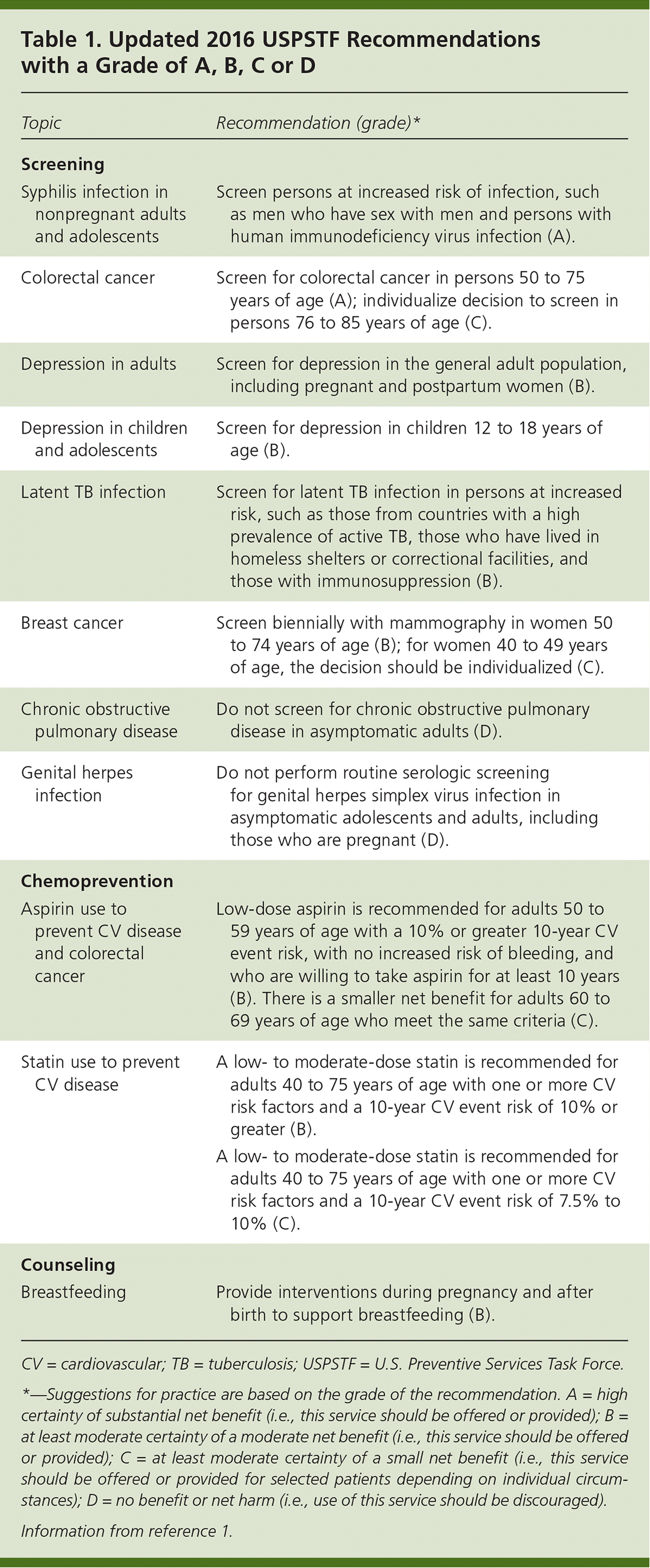
| Topic | Recommendation (grade)* |
|---|---|
| Screening | |
| Syphilis infection in nonpregnant adults and adolescents | Screen persons at increased risk of infection, such as men who have sex with men and persons with human immunodeficiency virus infection (A). |
| Colorectal cancer | Screen for colorectal cancer in persons 50 to 75 years of age (A); individualize decision to screen in persons 76 to 85 years of age (C). |
| Depression in adults | Screen for depression in the general adult population, including pregnant and postpartum women (B). |
| Depression in children and adolescents | Screen for depression in children 12 to 18 years of age (B). |
| Latent TB infection | Screen for latent TB infection in persons at increased risk, such as those from countries with a high prevalence of active TB, those who have lived in homeless shelters or correctional facilities, and those with immunosuppression (B). |
| Breast cancer | Screen biennially with mammography in women 50 to 74 years of age (B); for women 40 to 49 years of age, the decision should be individualized (C). |
| Chronic obstructive pulmonary disease | Do not screen for chronic obstructive pulmonary disease in asymptomatic adults (D). |
| Genital herpes infection | Do not perform routine serologic screening for genital herpes simplex virus infection in asymptomatic adolescents and adults, including those who are pregnant (D). |
| Chemoprevention | |
| Aspirin use to prevent CV disease and colorectal cancer | Low-dose aspirin is recommended for adults 50 to 59 years of age with a 10% or greater 10-year CV event risk, with no increased risk of bleeding, and who are willing to take aspirin for at least 10 years (B). There is a smaller net benefit for adults 60 to 69 years of age who meet the same criteria (C). |
| Statin use to prevent CV disease | A low- to moderate-dose statin is recommended for adults 40 to 75 years of age with one or more CV risk factors and a 10-year CV event risk of 10% or greater (B). |
| A low- to moderate-dose statin is recommended for adults 40 to 75 years of age with one or more CV risk factors and a 10-year CV event risk of 7.5% to 10% (C). | |
| Counseling | |
| Breastfeeding | Provide interventions during pregnancy and after birth to support breastfeeding (B). |
CV = cardiovascular; TB = tuberculosis; USPSTF = U.S. Preventive Services Task Force.
*—Suggestions for practice are based on the grade of the recommendation. A = high certainty of substantial net benefit (i.e., this service should be offered or provided); B = at least moderate certainty of a moderate net benefit (i.e., this service should be offered or provided); C = at least moderate certainty of a small net benefit (i.e., this service should be offered or provided for selected patients depending on individual circumstances); D = no benefit or net harm (i.e., use of this service should be discouraged).
Information from reference 1.
The 2016 recommendations for aspirin and statin use for primary prevention are also substantially different than before, now relying on a risk assessment using the Pooled Cohort Equations (http://tools.acc.org/ascvd-risk-estimator/). As recently discussed in American Family Physician,3 these equations may overestimate cardiovascular (CV) risk somewhat, so family physicians may choose to factor this into their decision making.
Aspirin use has two potential benefits: preventing predominantly nonfatal CV events and reducing the risk of colorectal cancer death. Patients who are at greater CV risk have more potential gain, and the reduction in colorectal cancer mortality occurs only after 10 or more years of aspirin use. Therefore, the USPSTF now recommends aspirin use only in adults 50 to 69 years of age who have a 10-year CV event risk of at least 10%, are willing to take aspirin for at least 10 years, and are not at increased risk of bleeding. The net benefit is judged to be moderate for persons 50 to 59 years of age but small for those 60 to 69 years of age.
Like the guidelines from the American College of Cardiology/American Heart Association (ACC/AHA),4 the USPSTF recommendations for statin use base the decision on the patient's 10-year CV event risk and do not identify specific low-density lipoprotein targets. They differ from the ACC/AHA guidelines in that they give a B rating for a low- or moderate-dose statin for patients with a 10-year CV event risk of 10% or greater, but a C rating for those with a 7.5% to 10% risk. The ACC/AHA guidelines are more aggressive and would result in more patients taking higher statin doses because they recommend a moderate- or high-dose statin for persons with a CV event risk of 7.5% or higher and shared decision making for persons with a CV event risk of 5% to 7.5%.
The recommendation to screen for depression in adults now explicitly includes pregnant and postpartum women. The Edinburgh Postnatal Depression Scale is the recommended screening tool and is available at http://pesnc.org/wp-content/uploads/EPDS.pdf.
Several 2016 recommendations were rated as I statements, including screening for skin cancer, screening for lipid disorders in children and adolescents, screening for impaired visual acuity in older adults, screening for depression in children younger than 12 years, aspirin use in adults younger than 50 years or older than 70 years, and statin use in adults older than 75 years with no history of CV disease.
A new I statement addresses screening for ASD. Although there have been several small clinical trials showing the benefit of treatment in children with ASD, all trials were conducted in children who were identified by parents or caregivers and who had relatively severe symptoms. The USPSTF recommendation covers screening in asymptomatic children whose parents and teachers have not identified any concerns. It is important to remember that an I statement is not a recommendation against providing the preventive service. Rather, clinicians should use their judgment in the context of their community and practice. If family physicians feel that children with behavioral problems suggestive of ASD are not consistently identified by families, they may wish to provide screening in their practices.
A more detailed description of all recommendations, rationale, and the methods of the USPSTF can be found at http://www.uspreventiveservicestaskforce.org. Clinicians can access the Electronic Preventive Services Selector, a free point-of-care tool for smartphones and website users (http://epss.ahrq.gov/PDA/index.jsp), as well as a free eBook with the full recommendation statements (http://www.ahrq.gov/professionals/clinicians-providers/guidelines-recommendations/guide/).
editor's note: Dr. Ebell is deputy editor for evidence-based medicine for AFP. He was previously a member of the USPSTF.

