Clinical Question
In which acutely ill children should urinary tract infection (UTI) be suspected, prompting a urine culture?
Evidence Summary
A recent observational study found that 2.2% of acutely ill children younger than five years met criteria for a UTI when their urine was cultured. However, in the same study, overall clinician impression that a UTI was present (fairly to very certain) identified only 28 out of 60 children with a microbiologically confirmed UTI.1 Because obtaining a urine specimen from all acutely ill children would be impractical, time-consuming, and potentially invasive, and would increase costs, a number of clinical decision rules have been developed and validated to identify children at increased risk of UTI, in whom a urine culture should be obtained.
A prospective study of 15,781 children younger than five years with febrile illness developed a multivariate model to identify children with UTI.2 However, this model is complex, with 26 variables, and is not practical for outpatient primary care practice. It also included patients from emergency departments, where there is likely a different spectrum of disease than in routine primary care practice. Another study developed a simpler model that included only five variables, with one point for each variable (age younger than 12 months, temperature greater than 102.2°F [39°C], white race, fever for two or more days, and no other potential source of fever).3 Those with a score of 2 or higher had a 6.4% likelihood of UTI, compared with 0.8% in those with a score less than 2. In a prospective case-control validation study of children in the emergency department, a score of 3 or higher was a better cutoff.4
Most recently, the UK Diagnosis of Urinary Tract Infection in Young Children (DUTY) study enrolled children five years and younger presenting with acute illness characterized by at least one systemic symptom (fever, vomiting, lethargy/malaise, irritability, poor feeding, or failure to thrive) and one symptom suggestive of UTI (abdominal pain, jaundice if younger than three months, hematuria, offensive urine odor, cloudy urine, loin pain, urinary frequency, apparent pain on passing urine, or changes in continence).1 All patients underwent a standardized assessment of signs and symptoms, and 2,740 were able to provide a clean-catch urine sample for culture as a reference standard. The researchers developed two clinical decision rules (Table 1).1 One is based on signs and symptoms alone to determine whether a urine sample should be obtained, and one incorporates dipstick urine test results and determines whether an antibiotic is warranted. Because there were only 60 children with UTIs out of the 2,740 who provided a clean-catch urine sample, split sample validation was not possible; bootstrap validation was used to verify the predictive accuracy of the model. The signs and symptoms model had an area under the receiver operating characteristic curve (AUROCC) of 0.88, and the expanded model that adds three variables from dipstick urine testing (hematuria, nitrite, and leukocyte esterase) had an AUROCC of 0.93. Both of these models are significantly more accurate than overall clinical impression that a UTI is present, which had an AUROCC of 0.77.
TABLE 1. The DUTY Clinical Decision Rules
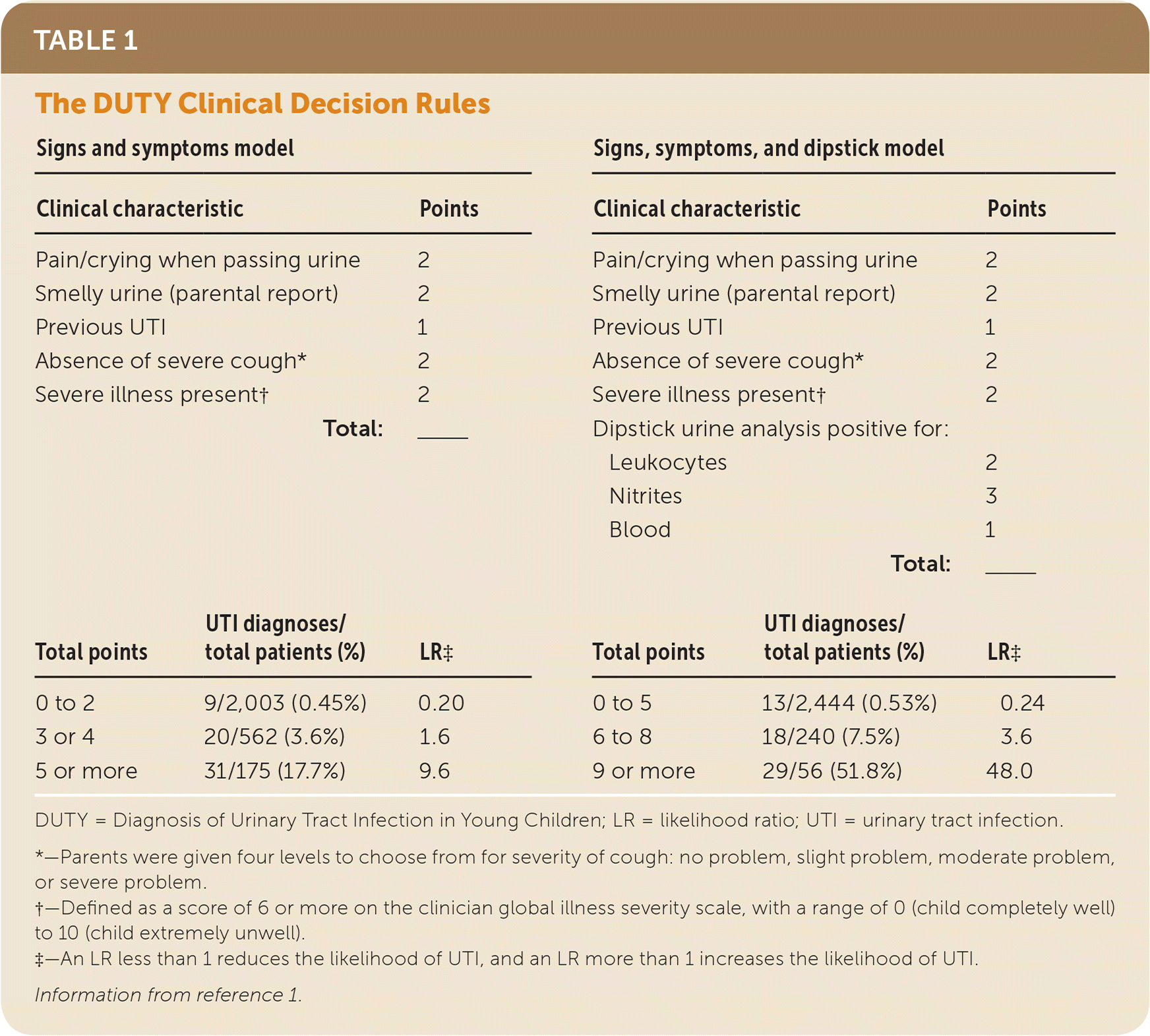
| Signs and symptoms model | Signs, symptoms, and dipstick model | |||||
| Clinical characteristic | Points | Clinical characteristic | Points | |||
| Pain/crying when passing urine | 2 | Pain/crying when passing urine | 2 | |||
| Smelly urine (parental report) | 2 | Smelly urine (parental report) | 2 | |||
| Previous UTI | 1 | Previous UTI | 1 | |||
| Absence of severe cough* | 2 | Absence of severe cough* | 2 | |||
| Severe illness present† | 2 | Severe illness present† | 2 | |||
| Total: | ____ | Dipstick urine analysis positive for: | ||||
| Leukocytes | 2 | |||||
| Nitrites | 3 | |||||
| Blood | 1 | |||||
| Total: | ____ | |||||
| Total points | UTI diagnoses/ total patients (%) | LR‡ | Total points | UTI diagnoses/ total patients (%) | LR‡ | |
| 0 to 2 | 9/2,003 (0.45%) | 0.20 | 0 to 5 | 13/2,444 (0.53%) | 0.24 | |
| 3 or 4 | 20/562 (3.6%) | 1.6 | 6 to 8 | 18/240 (7.5%) | 3.6 | |
| 5 or more | 31/175 (17.7%) | 9.6 | 9 or more | 29/56 (51.8%) | 48.0 | |
DUTY = Diagnosis of Urinary Tract Infection in Young Children; LR = likelihood ratio; UTI = urinary tract infection.
*—Parents were given four levels to choose from for severity of cough: no problem, slight problem, moderate problem, or severe problem.
†—Defined as a score of 6 or more on the clinician global illness severity scale, with a range of 0 (child completely well) to 10 (child extremely unwell).
‡—An LR less than 1 reduces the likelihood of UTI, and an LR more than 1 increases the likelihood of UTI.
Information from reference 1.
The authors of the DUTY trial recommend that clinicians first use the signs and symptoms model, and obtain a urine sample if a child has three or more signs and symptoms (5 points). Alternatively, clinicians could place children in three risk groups based on signs and symptoms. Those in the intermediate-risk group could be monitored more closely or a urine specimen could be obtained based on clinician judgment or the presence of other signs and symptoms for UTI not in the model. As with any clinical decision rule, the intent is to support clinician judgment and not replace it, and clinician judgment should prevail after considering all of the information.
Applying the Evidence
A three-year-old girl presents with smelly urine and apparent discomfort while urinating, according to her parents. She has no history of UTI and is not coughing. She has had decreased appetite but no vomiting or fever. She has somewhat less energy than usual. On examination, there are no other localizing signs of acute infection of the ears, throat, or lungs.
Using the DUTY sign and symptom decision rule, you calculate a score of 6 points, which corresponds to an 18% likelihood of UTI on urine culture. You obtain a urine specimen and dipstick urine test, which is positive for leukocytes and nitrites. Using the DUTY sign, symptom, and dipstick clinical decision rule, you calculate a score of 11 points, which corresponds to a 52% likelihood of UTI on urine culture. You decide to treat her with an oral antibiotic.

