Clinical Question
Is prophylactic vaccination against human papillomavirus (HPV) safe and effective in preventing HPV infection and resultant cervical cancer precursors?
Evidence-Based Answer
HPV vaccination in women 15 to 26 years of age prevents the development of cervical intraepithelial neoplasia (CIN) 2 and CIN 3 in women regardless of previous HPV exposure (number needed to treat [NNT] = 39). No serious adverse effects are associated with administration of the HPV vaccine.1 (Strength of Recommendation: C, based on consistent disease-oriented evidence.)
Practice Pointers
Cervical cancer is the fourth most common cancer in women worldwide, with more than one-half of this type of cancer occurring in women younger than 45 years. Persistent HPV infection can progress to CIN, with a subsequent 12% to 30% probability of CIN 3 (i.e., the highest grade CIN) progressing to invasive cancer.2 Cytology screening programs have decreased the incidence and mortality related to cervical cancer, but the treatment of high-grade CIN (i.e., CIN 2 or greater) remains a source of patient morbidity. Therefore, the primary prevention of high-grade CIN lesions with prophylactic HPV vaccination has the potential to reduce morbidity and mortality related to HPV infection.
This Cochrane review involved 26 randomized controlled trials and 73,428 international women 15 to 45 years of age.1 Included trials evaluated vaccine effectiveness and safety for the monovalent, bivalent, or quadrivalent HPV vaccines; the nonavalent HPV vaccine was not included. The surrogate primary outcome of high-grade CIN and adenocarcinoma in situ (AIS) incidence following prophylactic vaccination vs. placebo was evaluated, because assessment for the development of invasive cervical cancer would require prohibitively large and long-term studies. Although all but one trial were funded by the respective vaccine manufacturers, most trials were assessed to be at low risk of bias.
In women 15 to 26 years of age, HPV vaccination provided protection against persistent high-risk HPV types 16/18 infection at six and 12 months, as well as the development of CIN or AIS. Although women were HPV negative at study enrollment, the vaccine seems to prevent dysplasia. Even when HPV status was unknown, the vaccine still prevents CIN 2 (NNT = 60) and CIN 3 (NNT = 113). A subset of trials determined that significant protection against CIN 2 and CIN 3 was still afforded by receipt of only one or two doses of the bivalent and quadrivalent vaccines.
Only two studies included women 24 to 45 years of age. HPV vaccination in this age group did not appear to provide protection against CIN 2, and there were no data on CIN 3 or AIS.
Short-term local adverse effects (pain, erythema, or swelling at the injection site) were more common in women who received the vaccine vs. the control, but no serious adverse effects were observed. There was no increased risk of miscarriage among women who became pregnant during the trials.
Criticisms of this review have suggested that it does not include enough data about the nonavalent vaccine nor enough unpublished data; that authors of this review may not have adequately assessed risk of bias; and that the original reviews may have underestimated the risks and/or severity of adverse effects of HPV vaccine.3 In response, the Cochrane editors commissioned an independent review, which concluded that this analysis contained enough data to report the stated outcomes; that bias was adequately assessed; and that the risks and adverse effects were appropriately estimated.4
The Centers for Disease Control and Prevention and the Advisory Committee on Immunization Practices recommend prophylactic HPV vaccination for all girls 11 to 12 years of age, with catch-up vaccination for girls 13 to 26 years of age for the primary prevention of cervical cancer.5 Recently, the U.S. Food and Drug Administration expanded the approved age range to include women 27 to 45 years of age.6 Ideally, adolescent girls should be vaccinated before their first sexual contact to allow for the highest vaccine effectiveness, but vaccination will still protect against CIN or AIS regardless of previous HPV exposure.
SUMMARY TABLE: REPRESENTATIVE TREATMENT OUTCOMES WITH PLACEBO VS. HPV VACCINE
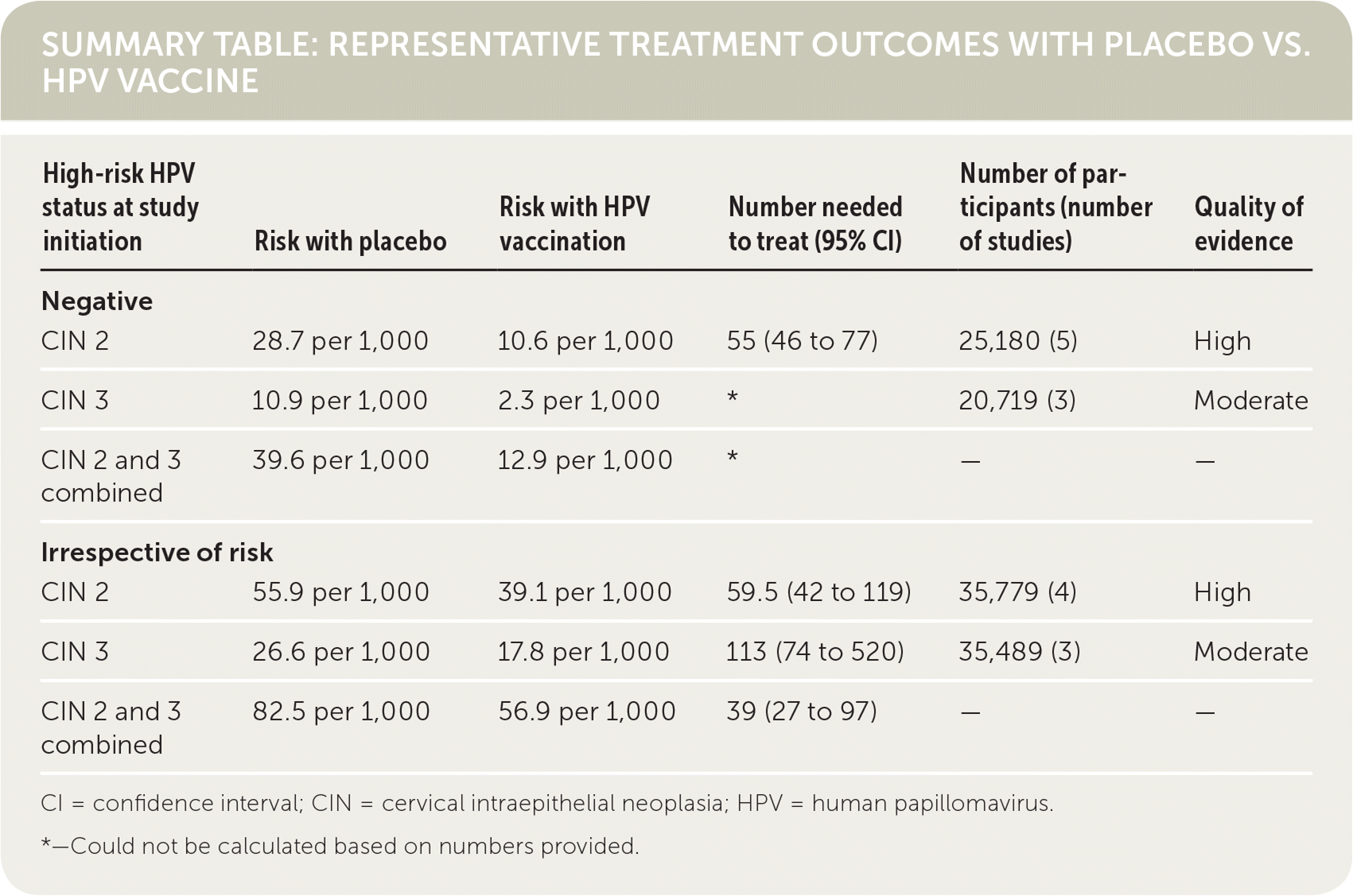
| High-risk HPV status at study initiation | Risk with placebo | Risk with HPV vaccination | Number needed to treat (95% CI) | Number of participants (number of studies) | Quality of evidence |
|---|---|---|---|---|---|
| Negative | |||||
| CIN 2 | 28.7 per 1,000 | 10.6 per 1,000 | 55 (46 to 77) | 25,180 (5) | High |
| CIN 3 | 10.9 per 1,000 | 2.3 per 1,000 | * | 20,719 (3) | Moderate |
| CIN 2 and 3 combined | 39.6 per 1,000 | 12.9 per 1,000 | * | — | — |
| Irrespective of risk | |||||
| CIN 2 | 55.9 per 1,000 | 39.1 per 1,000 | 59.5 (42 to 119) | 35,779 (4) | High |
| CIN 3 | 26.6 per 1,000 | 17.8 per 1,000 | 113 (74 to 520) | 35,489 (3) | Moderate |
| CIN 2 and 3 combined | 82.5 per 1,000 | 56.9 per 1,000 | 39 (27 to 97) | — | — |
CI = confidence interval; CIN = cervical intraepithelial neoplasia; HPV = human papillomavirus.
*—Could not be calculated based on numbers provided.
The practice recommendations in this activity are available at http://www.cochrane.org/CD009069.
Editor's Note: The numbers needed to treat reported in this Cochrane for Clinicians were calculated by the authors based on raw data provided in the original Cochrane review.

