Abnormal uterine bleeding is a common symptom in women. The acronym PALM-COEIN facilitates classification, with PALM referring to structural etiologies (polyp, adenomyosis, leiomyoma, malignancy and hyperplasia), and COEIN referring to nonstructural etiologies (coagulopathy, ovulatory dysfunction, endometrial, iatrogenic, not otherwise classified). Evaluation involves a detailed history and pelvic examination, as well as laboratory testing that includes a pregnancy test and complete blood count. Endometrial sampling should be performed in patients 45 years and older, and in younger patients with a significant history of unopposed estrogen exposure. Transvaginal ultrasonography is the preferred imaging modality and is indicated if a structural etiology is suspected or if symptoms persist despite appropriate initial treatment. Medical and surgical treatment options are available. Emergency interventions for severe bleeding that causes hemodynamic instability include uterine tamponade, intravenous estrogen, dilation and curettage, and uterine artery embolization. To avoid surgical risks and preserve fertility, medical management is the preferred initial approach for hemodynamically stable patients. Patients with severe bleeding can be treated initially with oral estrogen, high-dose estrogen-progestin oral contraceptives, oral progestins, or intravenous tranexamic acid. The most effective long-term medical treatment for heavy menstrual bleeding is the levonorgestrel-releasing intrauterine system. Other long-term medical treatment options include estrogen-progestin oral contraceptives, oral progestins, oral tranexamic acid, nonsteroidal anti-inflammatory drugs, and depot medroxyprogesterone. Hysterectomy is the definitive treatment. A lower-risk surgical option is endometrial ablation, which performs as well as the levonorgestrel-releasing intrauterine system. Select patients with chronic uterine bleeding can be treated with myomectomy, polypectomy, or uterine artery embolization.
Abnormal uterine bleeding is a common condition, with a prevalence of 10% to 30% among women of reproductive age.1 It negatively affects quality of life and is associated with financial loss, decreased productivity, poor health, and increased use of health care resources.2–4 In 2011 the International Federation of Gynecology and Obstetrics convened a working group that produced standardized definitions and classifications for menstrual disorders, which the American College of Obstetricians and Gynecologists subsequently endorsed.5,6 The updated terminology pertains only to nonpregnant women of reproductive age, which is the scope of this review.
WHAT IS NEW ON THIS TOPIC
The acronym PALM-COEIN facilitates the classification of abnormal uterine bleeding, with PALM referring to structural etiologies (polyp, adenomyosis, leiomyoma, malignancy and hyperplasia), and COEIN referring to nonstructural etiologies (coagulopathy, ovulatory dysfunction, endometrial, iatrogenic, not otherwise classified).
Among medical therapies, the 20-mcg-per-day formulation of the levonorgestrel-releasing intrauterine system (Mirena) is most effective for decreasing heavy menstrual bleeding (71% to 95% reduction in blood loss) and performs similarly to hysterectomy when quality-adjusted life years are considered.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
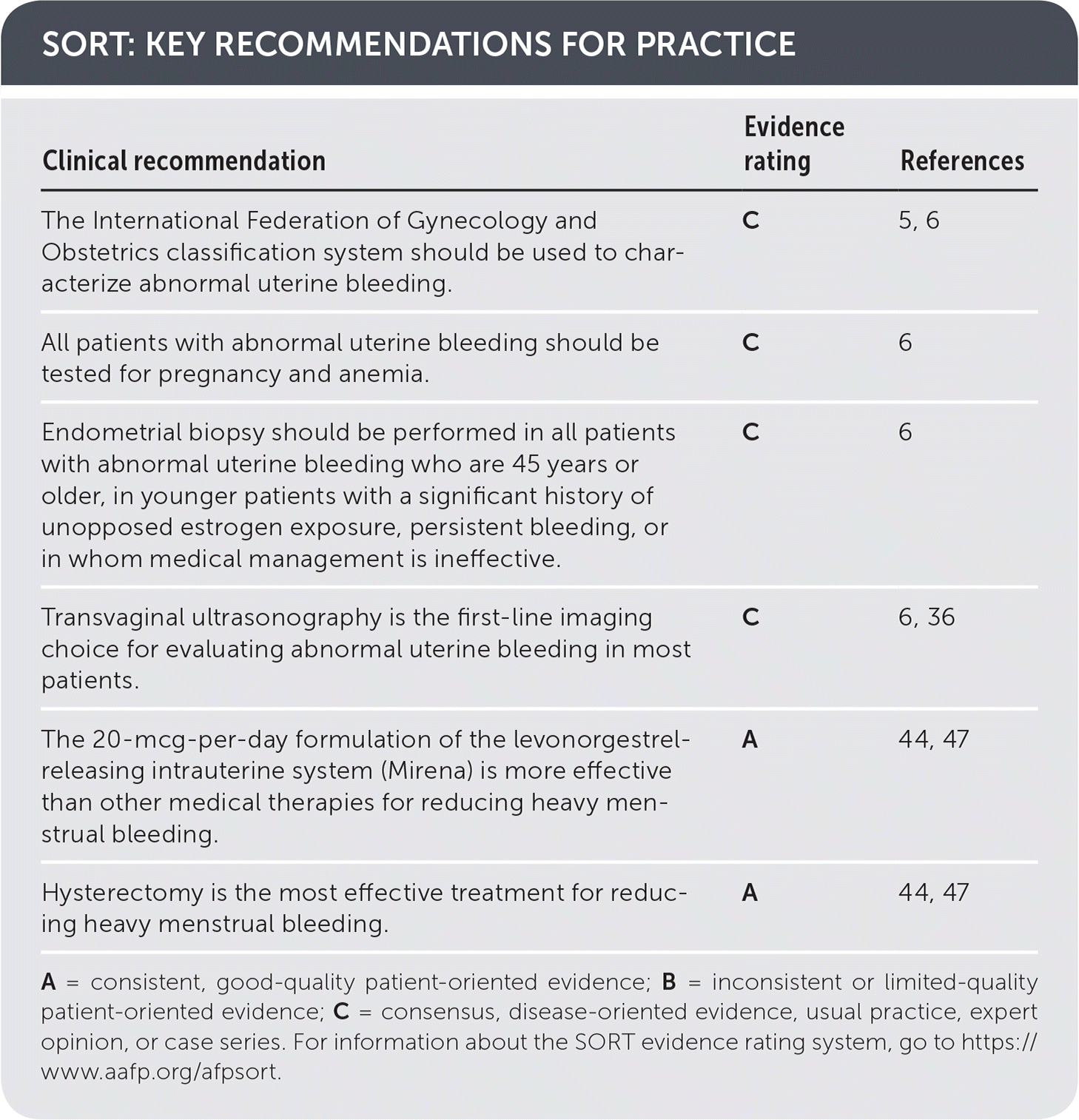
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| The International Federation of Gynecology and Obstetrics classification system should be used to characterize abnormal uterine bleeding. | C | 5, 6 |
| All patients with abnormal uterine bleeding should be tested for pregnancy and anemia. | C | 6 |
| Endometrial biopsy should be performed in all patients with abnormal uterine bleeding who are 45 years or older, in younger patients with a significant history of unopposed estrogen exposure, persistent bleeding, or in whom medical management is ineffective. | C | 6 |
| Transvaginal ultrasonography is the first-line imaging choice for evaluating abnormal uterine bleeding in most patients. | C | 6, 36 |
| The 20-mcg-per-day formulation of the levonorgestrel-releasing intrauterine system (Mirena) is more effective than other medical therapies for reducing heavy menstrual bleeding. | A | 44, 47 |
| Hysterectomy is the most effective treatment for reducing heavy menstrual bleeding. | A | 44, 47 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Definitions
Abnormal uterine bleeding is a symptom, not a diagnosis; the term is used to describe bleeding that falls outside population-based 5th to 95th percentiles for menstrual regularity, frequency, duration, and volume (Table 1).7 Abnormal bleeding is considered chronic when it has occurred for most of the previous six months, or acute when an episode of heavy bleeding warrants immediate intervention.5 Intermenstrual bleeding is bleeding that occurs between otherwise normal menstrual periods.7 Use of imprecise terms such as menorrhagia, metrorrhagia, and dysfunctional uterine bleeding is now discouraged.
TABLE 1 Definitions of Normal and Abnormal Menstrual Bleeding
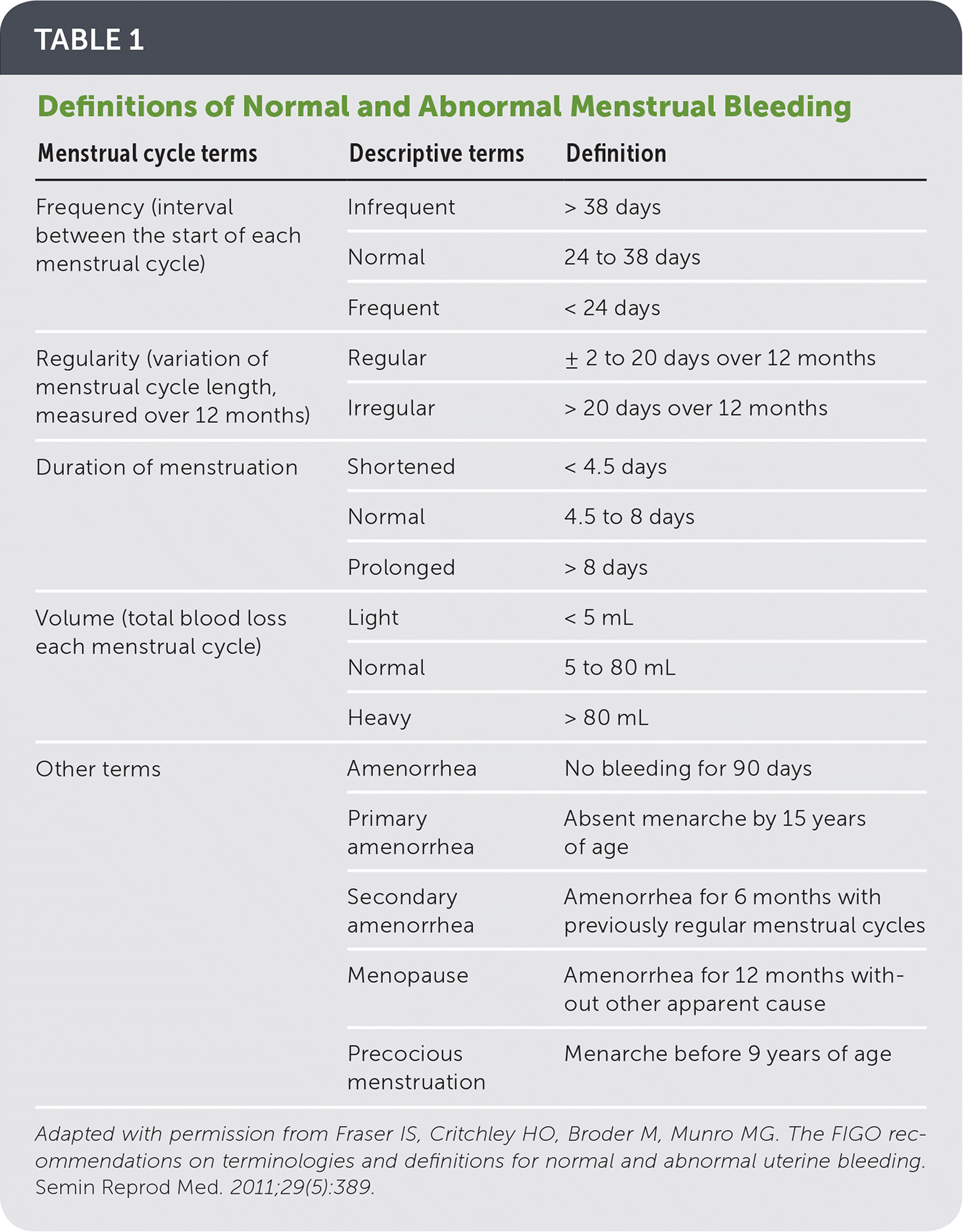
| Menstrual cycle terms | Descriptive terms | Definition |
|---|---|---|
| Frequency (interval between the start of each menstrual cycle) | Infrequent | > 38 days |
| Normal | 24 to 38 days | |
| Frequent | < 24 days | |
| Regularity (variation of menstrual cycle length, measured over 12 months) | Regular | ± 2 to 20 days over 12 months |
| Irregular | > 20 days over 12 months | |
| Duration of menstruation | Shortened | < 4.5 days |
| Normal | 4.5 to 8 days | |
| Prolonged | > 8 days | |
| Volume (total blood loss each menstrual cycle) | Light | < 5 mL |
| Normal | 5 to 80 mL | |
| Heavy | > 80 mL | |
| Other terms | Amenorrhea | No bleeding for 90 days |
| Primary amenorrhea | Absent menarche by 15 years of age | |
| Secondary amenorrhea | Amenorrhea for 6 months with previously regular menstrual cycles | |
| Menopause | Amenorrhea for 12 months without other apparent cause | |
| Precocious menstruation | Menarche before 9 years of age |
Adapted with permission from Fraser IS, Critchley HO, Broder M, Munro MG. The FIGO recommendations on terminologies and definitions for normal and abnormal uterine bleeding. Semin Reprod Med. 2011;29(5):389.
Differential Diagnosis
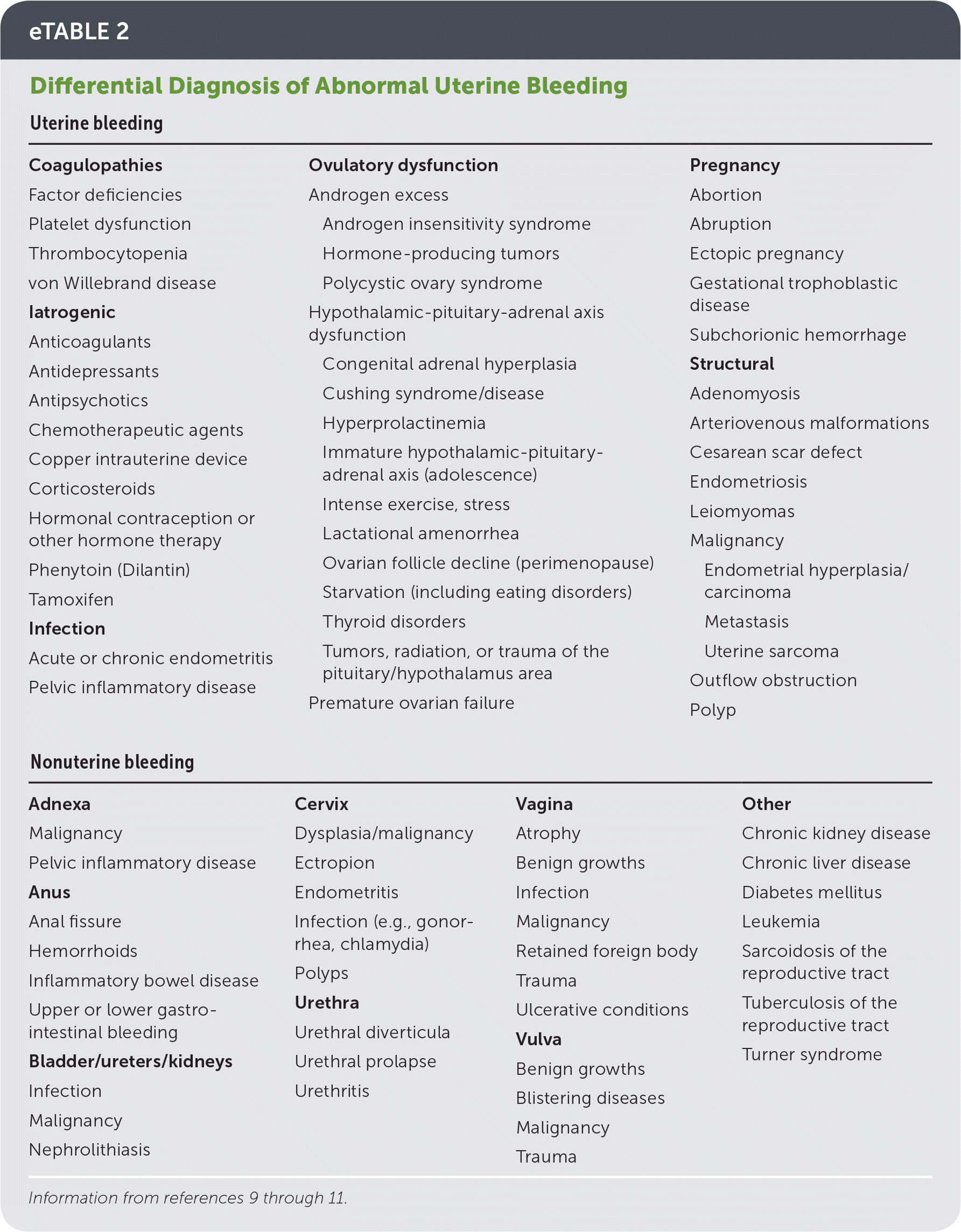
Although the uterus is often the source, any part of the female reproductive tract can result in vaginal bleeding. Women may also mistake bleeding from nongynecologic sites (e.g., bladder, urethra, perineum, anus) as vaginal bleeding. The prevalence of conditions that cause abnormal bleeding varies according to age. For example, anovulation is more common in adolescents and perimenopausal women, whereas the prevalence of structural lesions and malignancy increases with age.8 The differential diagnosis of abnormal uterine bleeding is presented in Table 2.9–11
TABLE 2 Differential Diagnosis of Abnormal Uterine Bleeding
| Uterine bleeding | |||
|---|---|---|---|
| Coagulopathies | |||
| Factor deficiencies | |||
| Platelet dysfunction | |||
| Thrombocytopenia | |||
| von Willebrand disease | |||
| Iatrogenic | |||
| Anticoagulants | |||
| Antidepressants | |||
| Antipsychotics | |||
| Chemotherapeutic agents | |||
| Copper intrauterine device | |||
| Corticosteroids | |||
| Hormonal contraception or other hormone therapy | |||
| Phenytoin (Dilantin) | |||
| Tamoxifen | |||
| Infection | |||
| Acute or chronic endometritis | |||
| Pelvic inflammatory disease | |||
| Ovulatory dysfunction | |||
| Androgen excess | |||
| Androgen insensitivity syndrome | |||
| Hormone-producing tumors | |||
| Polycystic ovary syndrome | |||
| Hypothalamic-pituitary-adrenal axis dysfunction | |||
| Congenital adrenal hyperplasia | |||
| Cushing syndrome/disease | |||
| Hyperprolactinemia | |||
| Immature hypothalamic-pituitary-adrenal axis (adolescence) | |||
| Intense exercise, stress | |||
| Lactational amenorrhea | |||
| Ovarian follicle decline (perimenopause) | |||
| Starvation (including eating disorders) | |||
| Thyroid disorders | |||
| Tumors, radiation, or trauma of the pituitary/hypothalamus area | |||
| Premature ovarian failure | |||
| Pregnancy | |||
| Abortion | |||
| Abruption | |||
| Ectopic pregnancy | |||
| Gestational trophoblastic disease | |||
| Subchorionic hemorrhage | |||
| Structural | |||
| Adenomyosis | |||
| Arteriovenous malformations | |||
| Cesarean scar defect | |||
| Endometriosis | |||
| Leiomyomas | |||
| Malignancy | |||
| Endometrial hyperplasia/carcinoma | |||
| Metastasis | |||
| Uterine sarcoma | |||
| Outflow obstruction | |||
| Polyp | |||
| Nonuterine bleeding | |||
| Adnexa | |||
| Malignancy | |||
| Pelvic inflammatory disease | |||
| Anus | |||
| Anal fissure | |||
| Hemorrhoids | |||
| Inflammatory bowel disease | |||
| Upper or lower gastrointestinal bleeding | |||
| Bladder/ureters/kidneys | |||
| Infection | |||
| Malignancy | |||
| Nephrolithiasis | |||
| Cervix | |||
| Dysplasia/malignancy | |||
| Ectropion | |||
| Endometritis | |||
| Infection (e.g., gonorrhea, chlamydia) | |||
| Polyps | |||
| Urethra | |||
| Urethral diverticula | |||
| Urethral prolapse | |||
| Urethritis | |||
| Vagina | |||
| Atrophy | |||
| Benign growths | |||
| Infection | |||
| Retained foreign body | |||
| Trauma | |||
| Ulcerative conditions | |||
| Vulva | |||
| Benign growths | |||
| Blistering diseases | |||
| Malignancy | |||
| Trauma | |||
| Other | |||
| Chronic kidney disease | |||
| Chronic liver disease | |||
| Diabetes mellitus | |||
| Leukemia | |||
| Sarcoidosis of the reproductive tract | |||
| Tuberculosis of the reproductive tract | |||
| Turner syndrome | |||
The most common causes of abnormal uterine bleeding are described with the acronym PALM-COEIN.5 The etiologies in the PALM group (polyp, adenomyosis, leiomyoma, malignancy and hyperplasia) are structural and can be imaged or biopsied. The etiologies in the COEIN group (coagulopathy, ovulatory dysfunction, endometrial, iatrogenic, not otherwise classified) are nonstructural. These etiologies are not mutually exclusive, and patients may have more than one cause.
POLYP
The lifetime prevalence of endometrial polyps ranges from 8% to 35%, and their incidence increases with age.12 Intermenstrual bleeding is the most common presenting symptom, but many polyps are asymptomatic. Physical examination findings are typically unremarkable, except for cases in which the polyps prolapse through the cervix.13 Although they can develop into malignancy, approximately 95% of symptomatic polyps are benign, and the risk of malignancy is even lower in premenopausal women.14
ADENOMYOSIS
The presence of endometrial tissue in the myometrium is known as adenomyosis. Its prevalence ranges from 5% to 70%, and its association with abnormal uterine bleeding is unclear.15 Many patients are asymptomatic, but those who have symptoms typically report painful, heavy, or prolonged menstrual bleeding. Examination may reveal a dense, enlarged uterus.
LEIOMYOMA
Leiomyomas (also called fibroids) are benign tumors arising from the uterine myometrium. Their prevalence increases with age; they are eventually found in up to 80% of all women.16 Most leiomyomas are asymptomatic, but bleeding is a common presenting symptom and typically involves heavy or prolonged menses. Larger leiomyomas are more likely to be associated with abnormal uterine bleeding.17 Patients may report pelvic pain or pressure, and on examination the uterus may be enlarged or irregularly contoured. More information on the diagnosis and treatment of leiomyomas is available in a previous American Family Physician article (https://www.aafp.org/afp/2017/0115/p100.html).
MALIGNANCY AND HYPERPLASIA
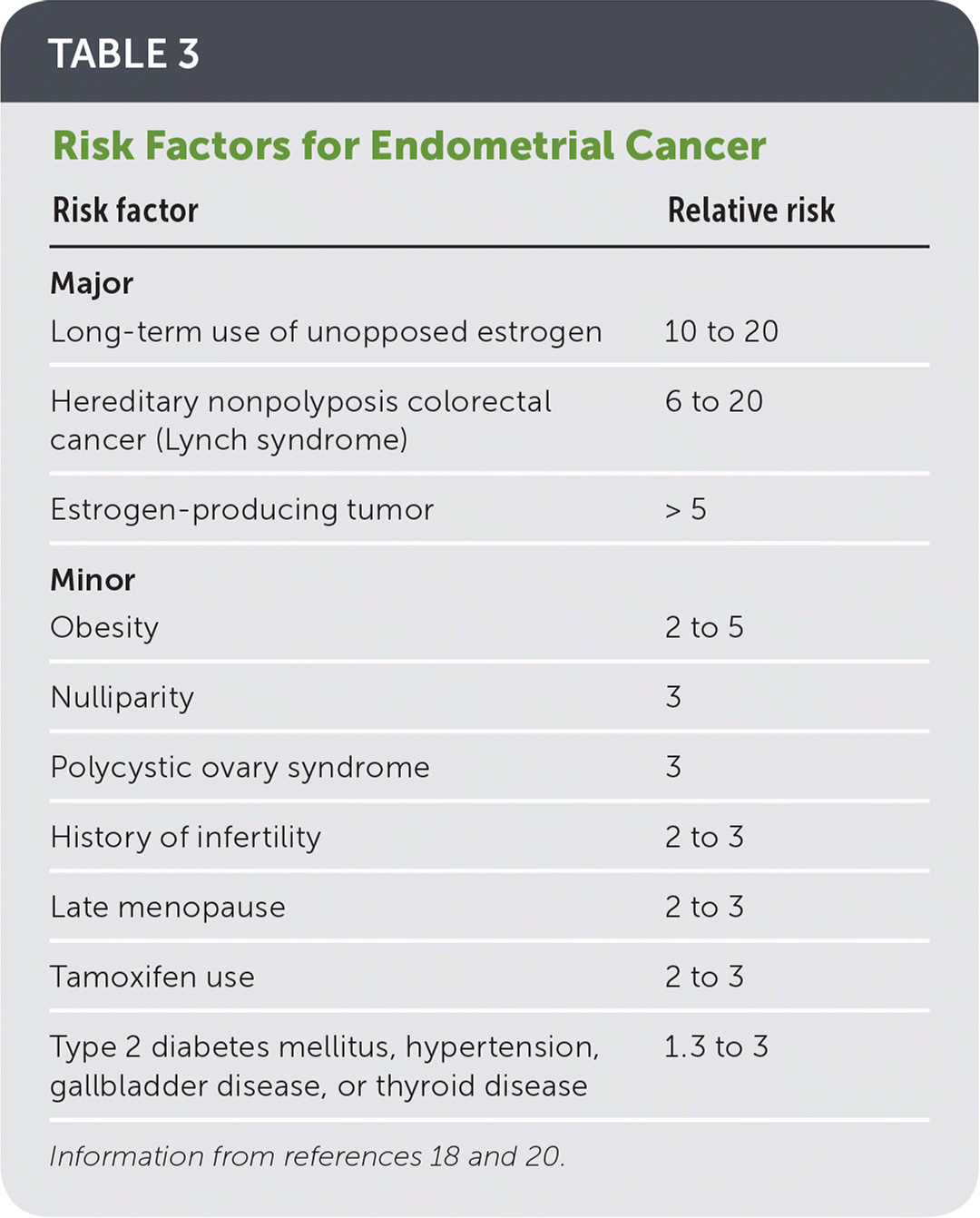
Abnormal uterine bleeding is the most common symptom of endometrial cancer.18 Although the prevalence of endometrial cancer increases with age, close to one-fourth of new diagnoses occur in patients younger than 55 years.19 Long-term unopposed estrogen exposure is the primary risk factor (Table 3).18,20 Bleeding patterns in patients with uterine malignancy are highly variable. More information on the diagnosis and management of endometrial cancer is available in a previous American Family Physician article (https://www.aafp.org/afp/2016/0315/p468.html).
TABLE 3 Risk Factors for Endometrial Cancer
| Risk factor | Relative risk |
|---|---|
| Major | |
| Long-term use of unopposed estrogen | 10 to 20 |
| Hereditary nonpolyposis colorectal cancer (Lynch syndrome) | 6 to 20 |
| Estrogen-producing tumor | > 5 |
| Minor | |
| Obesity | 2 to 5 |
| Nulliparity | 3 |
| Polycystic ovary syndrome | 3 |
| History of infertility | 2 to 3 |
| Late menopause | 2 to 3 |
| Tamoxifen use | 2 to 3 |
| Type 2 diabetes mellitus, hypertension, gallbladder disease, or thyroid disease | 1.3 to 3 |
COAGULOPATHY
Approximately 20% of patients with heavy menstrual bleeding have a bleeding disorder, and the prevalence in adolescent girls who bleed heavily is even higher.21–23 Von Willebrand disease and platelet dysfunction are the most common coagulopathies associated with abnormal uterine bleeding.24 In addition to heavy menstrual bleeding, adolescents with bleeding disorders may report irregular menstrual bleeding.25
OVULATORY DYSFUNCTION
A variety of endocrine disorders can lead to ovulatory dysfunction (Table 2).9–11 Infrequent or absent ovulation during the first few years after menarche and during perimenopause is common and not necessarily a sign of underlying pathology.26 Menstrual bleeding caused by ovulatory dysfunction is often irregular, heavy, or prolonged.
ENDOMETRIAL
Primary disorders of endometrial hemostasis typically occur in the setting of predictable ovulatory cycles and are likely due to vasoconstriction disorders, inflammation, or infection. Endometrial dysfunction is poorly understood; there are no reliable diagnostic methods, and it should be considered only after other causes are excluded.5
IATROGENIC
A variety of medical treatments can provoke abnormal uterine bleeding. Hormonal contraception is the most common cause of iatrogenic uterine bleeding (i.e., breakthrough bleeding).5 Other causative agents include noncontraceptive hormone therapy, drugs that interfere with sex steroid hormone function or synthesis (e.g., tamoxifen), anticoagulants, and dopamine antagonists (e.g., tricyclic antidepressants, some antipsychotics).
NOT OTHERWISE CLASSIFIED
This category contains poorly understood conditions, rare disorders (e.g., arteriovenous malformations), and conditions that do not otherwise fit into the classification system, such as cesarean scar defects, which can cause postmenstrual spotting when blood collects in the niche caused by the scar.
Diagnostic Evaluation
The approach to patients presenting with abnormal uterine bleeding includes assessing for hemodynamic instability and anemia, identifying the source of bleeding, pregnancy testing, and determining whether evaluation for endometrial carcinoma is indicated (Figure 1).6,18,26–30 The broad differential diagnosis necessitates a detailed history and physical examination.
FIGURE 1
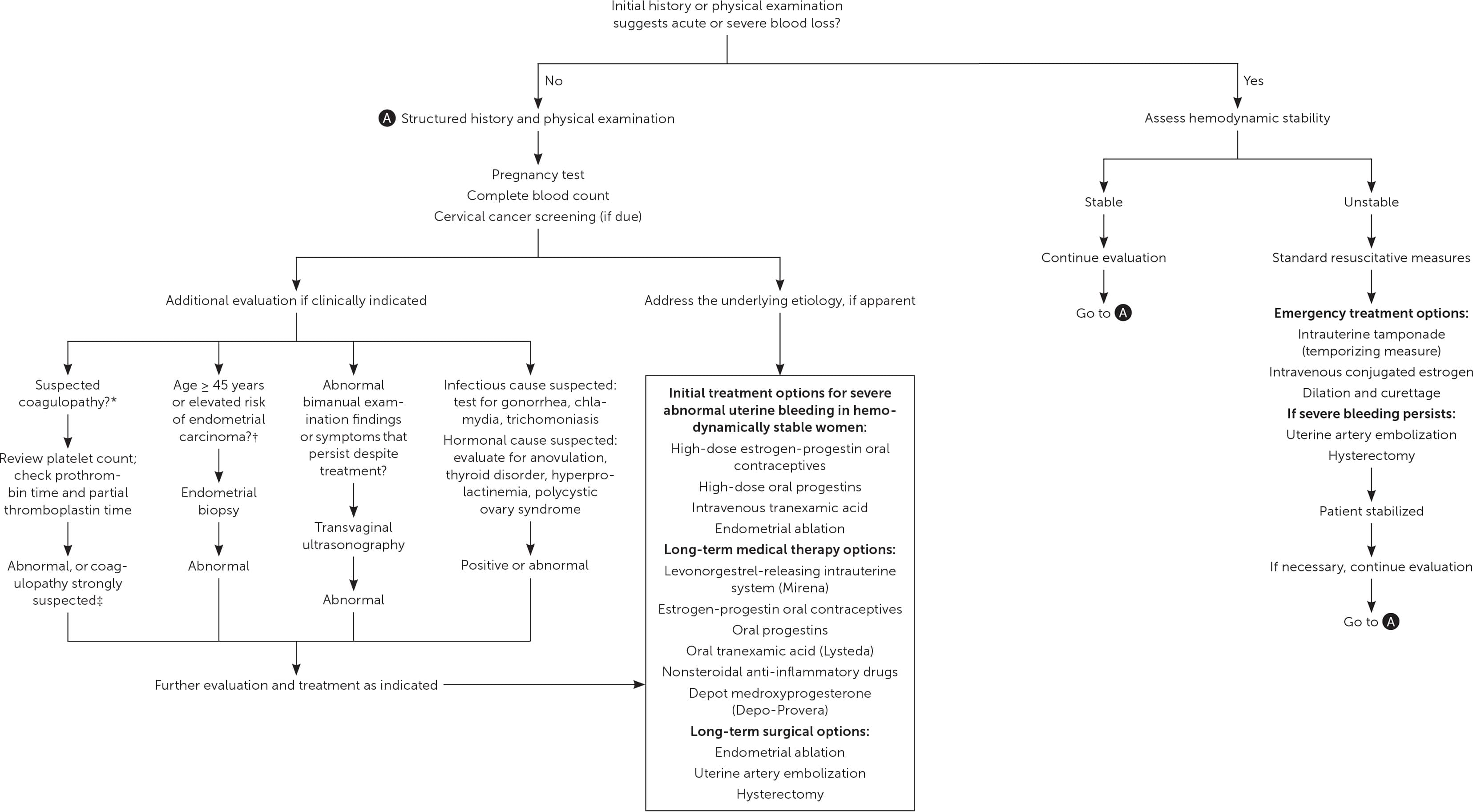
Evaluation and management of abnormal uterine bleeding.
*—The likelihood of a bleeding disorder increases if any of the following historical clues are present: heavy menstrual bleeding since menarche; history of postpartum hemorrhage, surgical bleeding, or bleeding with dental procedures; or two or more of the following: frequent gum bleeding, bruising > 5 cm at least monthly, epistaxis at least monthly, or family history of abnormal bleeding.25
†—Risk factors include a history of exposure to unopposed estrogen (Table 3), failed medical management, and persistent bleeding.
‡—Initial screening tests may be normal in the setting of some coagulopathies, with diagnosis requiring further testing and possibly hematology consultation.
Information from references 6, 18, and 26 through 30.
BLEEDING HISTORY
A description of the bleeding pattern should be elicited, including frequency, duration, regularity, and volume. Heavy menstrual bleeding is defined as more than 80 mL of total blood loss, but quantitative assessment is impractical in routine clinical practice. Historical clues such as passing blood clots or changing pads/tampons at least hourly suggest heavy menstrual bleeding.31 A history of postcoital bleeding may indicate cervicitis, ectropion, or, rarely, cervical cancer, whereas abdominopelvic pain may suggest infection, structural lesions, or endometriosis.
Clinicians may underestimate the prevalence of coagulopathies among patients with abnormal uterine bleeding.32 These conditions should be considered in women with a family history of abnormal bleeding or a personal history of heavy menstrual bleeding since menarche, or symptoms such as frequent bruising, bleeding gums, epistaxis, postpartum hemorrhage, or bleeding with surgical and dental procedures.27
PHYSICAL EXAMINATION
An examination of the pelvis, including speculum and bimanual examinations, is an important aspect of the evaluation of abnormal uterine bleeding. Care should be taken to examine all potential bleeding sites, including the urethra, perineum, and anus. Cervical cancer screening should be performed if it is not up to date. Pelvic examination can be deferred in adolescents if the patient is not sexually active, neither trauma nor infection is suspected, and the response to initial treatment is adequate.33
LABORATORY TESTING
All patients with abnormal uterine bleeding should be evaluated for pregnancy with a urine or serum human chorionic gonadotropin test, and for anemia and thrombocytopenia with a complete blood count.6 Thyroid function should be evaluated in patients with signs or symptoms of thyroid disease, or if the initial workup does not reveal a likely cause.6,28,34 Additional hormonal tests (e.g., prolactin, androgens, estrogen) are indicated only if history or examination findings suggest a specific hormonal cause.26,28 The platelet count, prothrombin time, and partial thromboplastin time can be initial screening tests when a bleeding disorder is suspected, but results may be normal in women with von Willebrand disease or other bleeding disorders. Diagnosing a bleeding disorder typically requires additional testing, often in consultation with a hematologist.27
Because older age is an important risk factor for endometrial cancer, all patients with abnormal uterine bleeding who are 45 years or older should undergo endometrial sampling.18 Younger women should undergo sampling if they have a history of unopposed estrogen exposure, if medical management fails, or if bleeding symptoms persist.6 Office-based endometrial biopsy is the preferred approach, with hysteroscopic dilation and curettage reserved for instances in which office sampling fails, is inadequate, or cannot be performed.35 Blind sampling may miss focal lesions, so hysteroscopic dilation and curettage should be performed if symptoms persist despite normal biopsy results.18
IMAGING
Indications for pelvic imaging include abnormalities palpated on bimanual examination or symptoms that persist despite initial treatment.6 Transvaginal ultrasonography is the first-line approach for most patients, although saline infusion sonohysterography (the infusion of sterile saline into the endometrial cavity while transvaginal ultrasonography is performed) is better at detecting intracavitary lesions.36 Routine use of magnetic resonance imaging is discouraged but can be considered if sonographic imaging is inadequate.6
Management
Multiple factors should be considered when choosing among treatment options for abnormal uterine bleeding (Table 4),37–42 including the cause and acuity of the bleeding, fertility and contraceptive preferences, medical comorbidities, adverse effects, cost, and relative effectiveness. If the underlying cause of bleeding can be identified and treated, symptoms may resolve without the need for additional intervention. Anemia is an indication for treatment, as is bleeding that negatively affects the patient's quality of life. Because exposure to unopposed estrogen increases the risk of endometrial cancer, treatment of anovulatory abnormal uterine bleeding involves inducing ovulatory cycles or administering supplemental progesterone to antagonize estrogen's proliferative effect on the endometrium.
TABLE 4 Treatment Options for Medical Management of Abnormal Uterine Bleeding
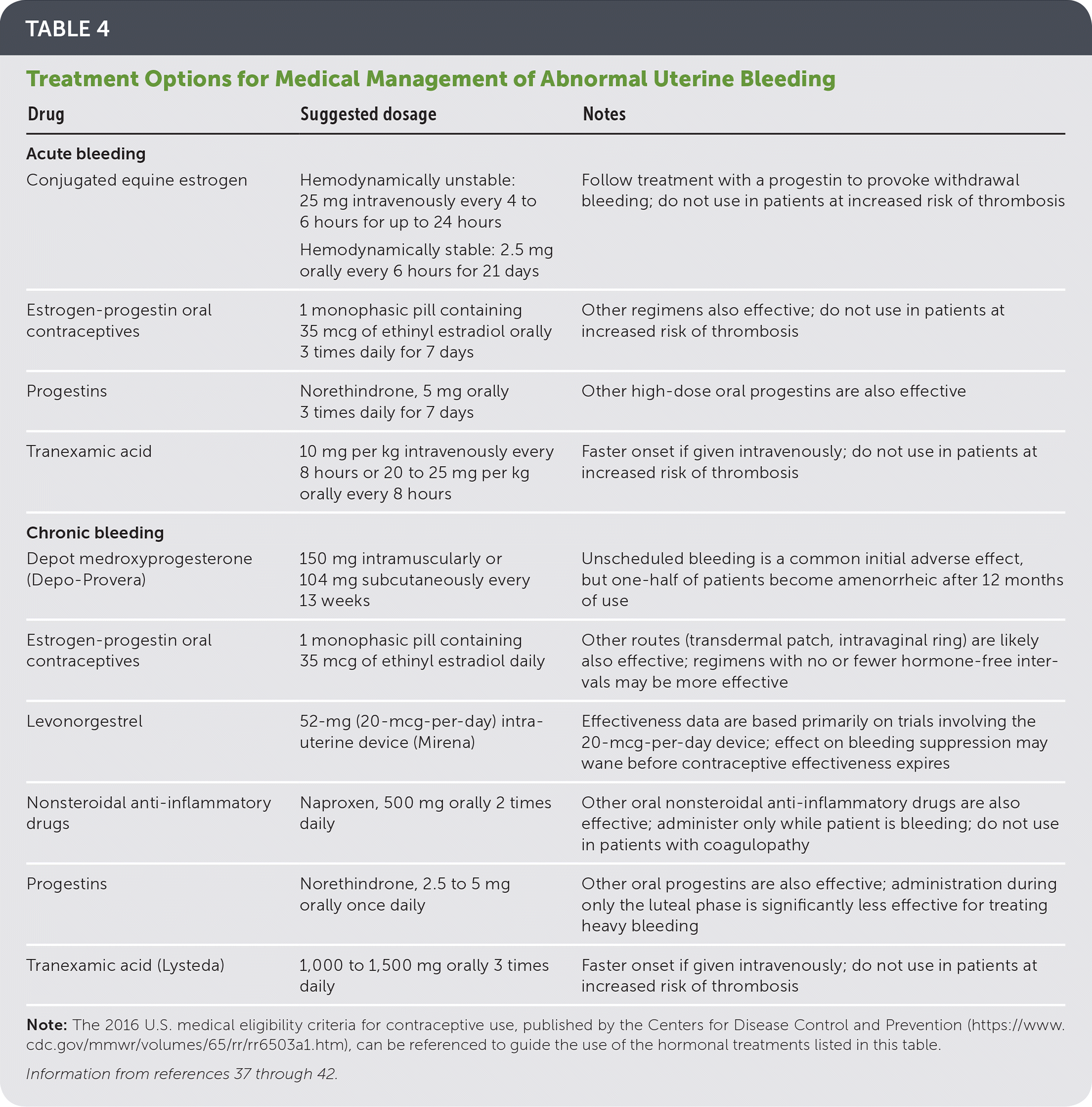
| Drug | Suggested dosage | Notes |
|---|---|---|
| Acute bleeding | ||
| Conjugated equine estrogen | Hemodynamically unstable: 25 mg intravenously every 4 to 6 hours for up to 24 hours | Follow treatment with a progestin to provoke withdrawal bleeding; do not use in patients at increased risk of thrombosis |
| Hemodynamically stable: 2.5 mg orally every 6 hours for 21 days | ||
| Estrogen-progestin oral contraceptives | 1 monophasic pill containing 35 mcg of ethinyl estradiol orally 3 times daily for 7 days | Other regimens also effective; do not use in patients at increased risk of thrombosis |
| Progestins | Norethindrone, 5 mg orally 3 times daily for 7 days | Other high-dose oral progestins are also effective |
| Tranexamic acid | 10 mg per kg intravenously every 8 hours or 20 to 25 mg per kg orally every 8 hours | Faster onset if given intravenously; do not use in patients at increased risk of thrombosis |
| Chronic bleeding | ||
| Depot medroxyprogesterone (Depo-Provera) | 150 mg intramuscularly or 104 mg subcutaneously every 13 weeks | Unscheduled bleeding is a common initial adverse effect, but one-half of patients become amenorrheic after 12 months of use |
| Estrogen-progestin oral contraceptives | 1 monophasic pill containing 35 mcg of ethinyl estradiol daily | Other routes (transdermal patch, intravaginal ring) are likely also effective; regimens with no or fewer hormone-free intervals may be more effective |
| Levonorgestrel | 52-mg (20-mcg-per-day) intrauterine device (Mirena) | Effectiveness data are based primarily on trials involving the 20-mcg-per-day device; effect on bleeding suppression may wane before contraceptive effectiveness expires |
| Nonsteroidal anti-inflammatory drugs | Naproxen, 500 mg orally 2 times daily | Other oral nonsteroidal anti-inflammatory drugs are also effective; administer only while patient is bleeding; do not use in patients with coagulopathy |
| Progestins | Norethindrone, 2.5 to 5 mg orally once daily | Other oral progestins are also effective; administration during only the luteal phase is significantly less effective for treating heavy bleeding |
| Tranexamic acid (Lysteda) | 1,000 to 1,500 mg orally 3 times daily | Faster onset if given intravenously; do not use in patients at increased risk of thrombosis |
Note: The 2016 U.S. medical eligibility criteria for contraceptive use, published by the Centers for Disease Control and Prevention (https://www.cdc.gov/mmwr/volumes/65/rr/rr6503a1.htm), can be referenced to guide the use of the hormonal treatments listed in this table.
EMERGENT TREATMENT
Occasionally, abnormal uterine bleeding is of sufficient quantity or duration that emergent attention is required. For hemodynamically unstable patients, uterine tamponade using a Foley catheter or gauze packing can achieve rapid but temporary control of blood loss.43 Further emergency interventions for hemodynamically unstable patients include intravenous estrogen, dilation and curettage, uterine artery embolization, and, rarely, hysterectomy. Medical therapy (e.g., oral estrogen, combined oral contraceptives, oral progestins, intravenous tranexamic acid) is usually adequate for treating hemodynamically stable patients with severe bleeding.
NONEMERGENT TREATMENT
A wider range of medical and surgical options are available for treatment of nonemergent uterine bleeding (Table 4).37–42 To avoid surgical risks and preserve fertility, medical management is the first-line approach for most patients.44 Among medical therapies, the 20-mcg-per-day formulation of the levonorgestrel-releasing intrauterine system (Mirena) is most effective for decreasing heavy menstrual bleeding (71% to 95% reduction in blood loss) and is as effective as hysterectomy when quality-adjusted life years are considered.39,45–47 Estrogen-progestin oral contraceptives are effective (35% to 69% reduction) and can also be used to regulate bleeding in patients with ovulatory dysfunction.39,48 Continuous dosing of oral progestins is another effective hormonal treatment option (87% reduction), but long-term patient satisfaction is low.39,49 Two effective, well-tolerated, nonhormonal choices are oral tranexamic acid (Lysteda; 26% to 54% reduction) and nonsteroidal anti-inflammatory drugs (10% to 52% reduction).39,50 Both are taken only when the patient is bleeding, and tranexamic acid has the added benefit of being safe while the patient is attempting to conceive.
Hysterectomy is the definitive and most effective treatment for abnormal uterine bleeding, and it yields a high level of patient satisfaction.44,47,51 A less invasive, lower-risk surgical option is endometrial ablation, which is as effective as the levonorgestrel-releasing intrauterine system.47 A variety of ablative techniques are available, and all are equivalent in terms of bleeding outcomes and patient satisfaction.52 Myomectomy and uterine artery embolization are treatment options for leiomyomas, and endometrial polyps can be treated with polypectomy. Except for myomectomy and polypectomy, surgical interventions for abnormal uterine bleeding are contraindicated in patients who wish to preserve fertility.
This article updates previous articles on this topic by Sweet, et al.53; Albers, et al.54; and Oriel and Schrager.55
Data Sources: A PubMed search was completed in Clinical Queries using the key terms abnormal uterine bleeding, heavy menstrual bleeding, irregular menstrual bleeding, menorrhagia, metrorrhagia, and dysfunctional uterine bleeding. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Also searched were the Agency for Healthcare Research and Quality evidence reports, Clinical Evidence, the Cochrane database, and UpToDate. Search dates: August 21, 2017, and November 10, 2018.

