Clinical Question
Do therapies that help patients who have heart failure with reduced ejection fraction (HFrEF) also improve morbidity and mortality in patients who have heart failure with preserved ejection fraction (HFpEF)?
Evidence-Based Answer
Mineralocorticoid receptor antagonists reduce hospitalizations for patients with HFpEF. Angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) have not been shown to change the morbidity or mortality in patients with HFpEF. Beta blockers may positively affect cardiovascular mortality, but they do not improve hospitalizations or all-cause mortality.1 (Strength of Recommendation: A, based on consistent, good-quality patient-oriented evidence.)
Practice Pointers
HFpEF is a heterogeneous clinical syndrome with signs and symptoms of heart failure with left ventricular ejection fraction greater than 40%.2 Six million adults in the United States have heart failure.3 Patients with HFpEF account for one-half of this population and share similar mortality rates as those with HFrEF.1,4 The authors of this Cochrane review sought to determine if therapies that improve morbidity and mortality in patients with HFrEF (i.e., beta blockers, mineralocorticoid receptor antagonists, ACE inhibitors, and ARBs) have similar benefits in those with HFpEF.1
SUMMARY TABLE: TREATMENTS FOR CHRONIC HEART FAILURE WITH PRESERVED EJECTION FRACTION
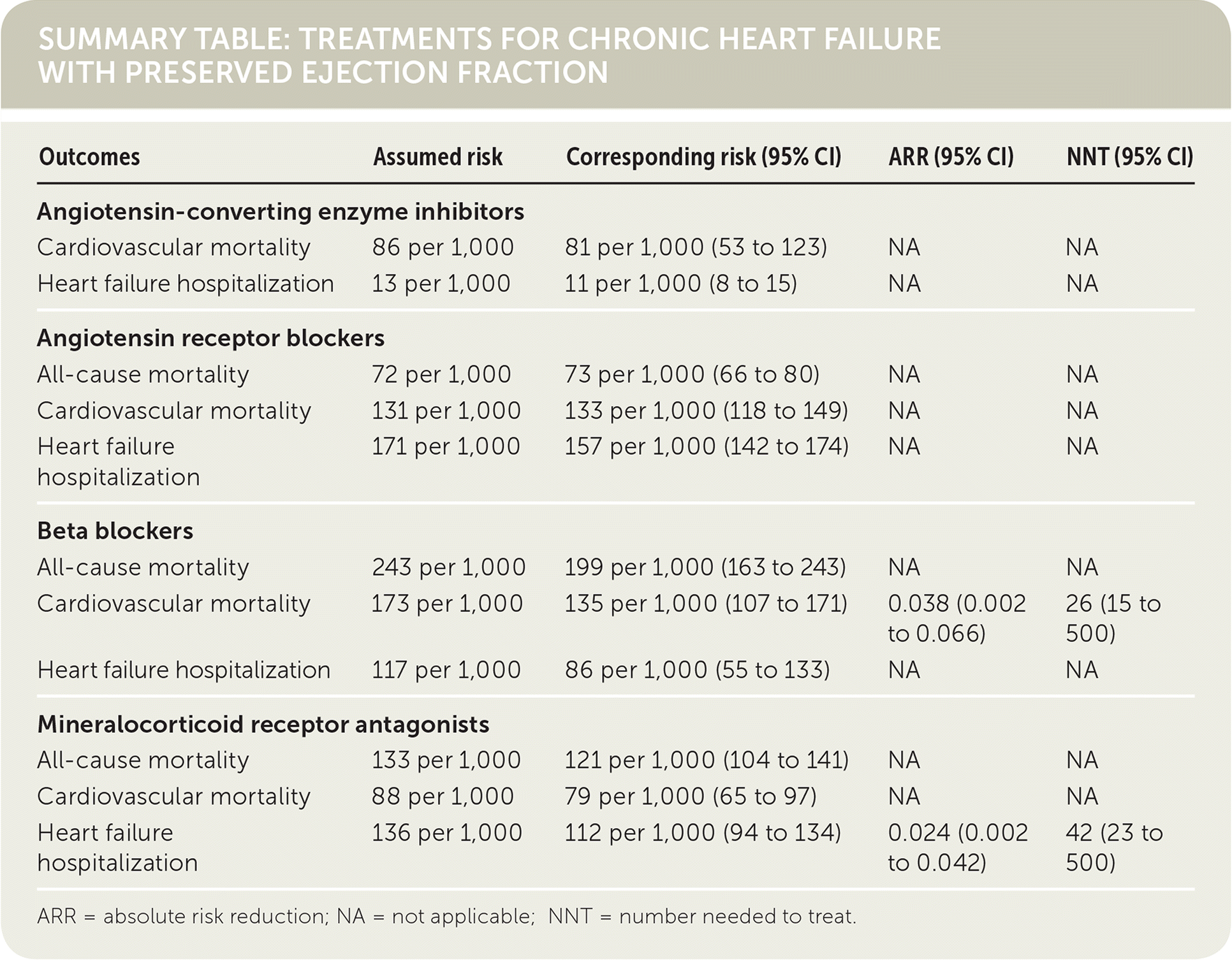
| Outcomes | Assumed risk | Corresponding risk (95% CI) | ARR (95% CI) | NNT (95% CI) |
|---|---|---|---|---|
| Angiotensin-converting enzyme inhibitors | ||||
| Cardiovascular mortality | 86 per 1,000 | 81 per 1,000 (53 to 123) | NA | NA |
| Heart failure hospitalization | 13 per 1,000 | 11 per 1,000 (8 to 15) | NA | NA |
| Angiotensin receptor blockers | ||||
| All-cause mortality | 72 per 1,000 | 73 per 1,000 (66 to 80) | NA | NA |
| Cardiovascular mortality | 131 per 1,000 | 133 per 1,000 (118 to 149) | NA | NA |
| Heart failure hospitalization | 171 per 1,000 | 157 per 1,000 (142 to 174) | NA | NA |
| Beta blockers | ||||
| All-cause mortality | 243 per 1,000 | 199 per 1,000 (163 to 243) | NA | NA |
| Cardiovascular mortality | 173 per 1,000 | 135 per 1,000 (107 to 171) | 0.038 (0.002 to 0.066) | 26 (15 to 500) |
| Heart failure hospitalization | 117 per 1,000 | 86 per 1,000 (55 to 133) | NA | NA |
| Mineralocorticoid receptor antagonists | ||||
| All-cause mortality | 133 per 1,000 | 121 per 1,000 (104 to 141) | NA | NA |
| Cardiovascular mortality | 88 per 1,000 | 79 per 1,000 (65 to 97) | NA | NA |
| Heart failure hospitalization | 136 per 1,000 | 112 per 1,000 (94 to 134) | 0.024 (0.002 to 0.042) | 42 (23 to 500) |
ARR = absolute risk reduction; NA = not applicable; NNT = number needed to treat.
This review included 37 randomized controlled trials with 18,311 patients. Primary outcomes were cardiovascular mortality and heart failure hospitalization, and secondary outcomes included all-cause mortality and quality of life. A limitation of this review was that a range of left ventricular ejection fraction cutoffs were used for HFpEF diagnosis (between 40% and 50%).
Beta blockers (i.e., carvedilol [Coreg], nebivolol [Bystolic], propranolol, metoprolol succinate, and bisoprolol) were compared with placebo in five studies and with usual care in five studies for a total of 3,087 patients. None of these studies were conducted in the United States or Canada. Only a few studies were included in the meta-analysis because of different outcome measures reported. There was an apparent reduction in cardiovascular mortality when examining the data across three studies (absolute risk reduction [ARR] = 0.038; 95% CI, 0.002 to 0.066; number needed to treat [NNT] = 26). However, the authors were cautious about drawing conclusions from this because when they removed the two studies that were considered to be at high risk of bias, the one remaining low-risk study did not demonstrate a difference in cardiovascular mortality between outcomes with beta blockers and placebo (relative risk = 0.81; 95% CI, 0.50 to 1.29; 643 participants).
Mineralocorticoid receptor antagonists (i.e., spironolactone, eplerenone [Inspra], and canrenone [not available in the United States]) were compared with placebo in eight studies and to usual care in four studies for a total of 4,408 participants. Some of these studies were conducted in the United States and Canada. Although most findings were driven by one trial with 3,445 patients, treatment with mineralocorticoid receptor antagonists led to a reduction in heart failure hospitalization (ARR = 0.024; 95% CI, 0.002 to 0.042; NNT = 42) with an increased risk of hyperkalemia. There was, however, no benefit on cardiovascular or all-cause mortality.
ACE inhibitors (i.e., enalapril [Vasotec], benazepril [Lotensin], perindopril [Aceon], ramipril [Altace], and quinapril [Accupril]) were compared with placebo in three studies and with usual care in five studies for a total of 2,061 patients with HFpEF. None of the studies were conducted in the United States or Canada, and the findings were again driven by one trial with 850 patients. ACE inhibitors showed no benefits on cardiovascular mortality, heart failure hospitalizations, all-cause mortality, or quality of life.
ARBs (i.e., candesartan [Atacand], irbesartan [Avapro], valsartan [Diovan], and olmesartan [Benicar]) were compared with placebo in five studies and with usual care in three studies for a total of 8,755 patients with HFpEF. There were no benefits with regard to cardiovascular mortality, heart failure hospitalizations, all-cause mortality, or quality of life.
Guidelines on the treatment of HFpEF call for consideration of mineralocorticoid receptor antagonists to decrease hospitalization in patients who meet the following criteria: ejection fraction of 45% or more, elevated brain natriuretic peptide or heart failure hospitalization within one year, estimated glomerular filtration rate greater than 30 mL per minute, creatinine levels less than 2.5 mg per dL (221 μmol per L), and potassium levels less than 5.0 mEq per L (5 mmol per L). The American College of Cardiology also continues to recommend that ARB treatment be considered to decrease hospitalizations, consistent with previous guidelines.5
The practice recommendations in this activity are available at http://www.cochrane.org/CD012721.
Editor's Note: The absolute risk reductions and numbers needed to treat reported in this Cochrane for Clinicians were calculated by the author based on raw data provided in the original Cochrane review.

