Gout is caused by monosodium urate crystal deposition in joints and tissues. Risk factors include male sex; obesity; hypertension; alcohol intake; diuretic use; a diet rich in meat and seafood; chronic kidney disease; a diet heavy in fructose-rich food and beverages; being a member of certain ethnic groups, including Taiwanese, Pacific Islander, and New Zealand Maori; and living in high-income countries. Gout is characterized by swelling, pain, or tenderness in a peripheral joint or bursa, including the development of a tophus. Diagnosis of gout can be made using several validated clinical prediction rules. Arthrocentesis should be performed when suspicion for an underlying septic joint is present; synovial fluid or tophus analysis should be performed if the diagnosis is uncertain. Colchicine, nonsteroidal anti-inflammatory drugs, and corticosteroids relieve pain in adults with acute gout episodes. Indications for long-term urate-lowering therapy include chronic kidney disease, two or more flare-ups per year, urolithiasis, the presence of tophus, chronic gouty arthritis, and joint damage. Allopurinol and febuxostat are used to prevent flare-ups, although febuxostat is associated with an increase in all-cause and cardiovascular mortality and is therefore not routinely recommended.
Gout, caused by monosodium urate crystal deposition in joints and tissues, is commonly encountered in primary care. This article provides a review of patient-oriented evidence to guide the diagnosis and management of gout.
SORT: TABLE OF KEY CLINICAL RECOMMENDATIONS
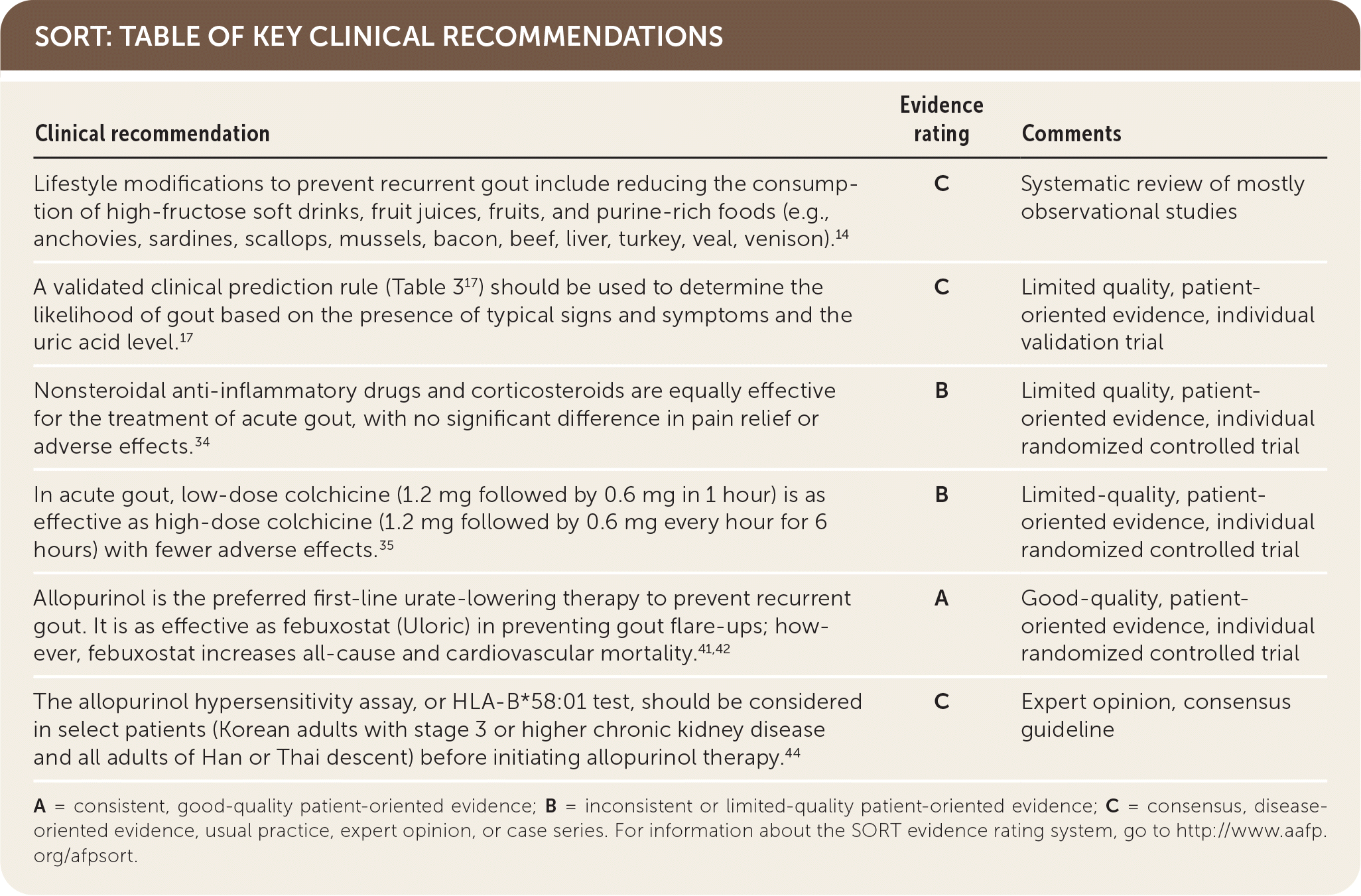
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to http://www.aafp.org/afpsort.
Epidemiology
- In western high-income countries, the prevalence of gout is 3% to 6% in men and 1% to 3% in women.1 In 2015 and 2016, the incidence of gout was 3.9% among U.S. adults (2.7% in women and 5.2% in men).2
- Gout is rare before the age of 20; prevalence increases linearly until plateauing after the age of 80.3
- Risk factors are summarized in Table 1.1–14
- Consumption of purine-rich foods is a risk factor for gout. These include some fish (e.g., anchovies, sardines, scallops, mussels) and meats (e.g., bacon, beef, liver, turkey, veal, venison).
- Beer is associated with a larger increase in risk of gout compared with wine or hard liquor.14
- Use of loop or thiazide diuretic or tacrolimus (Prograf) is associated with increased risk of gout.14
- Consumption of two or more sugar-sweetened soft drinks per day, fruits high in fructose, or any fruit juices is associated with an increased risk of gout in men. Consumption of diet soft drinks does not appear to increase risk.14
TABLE 1. Risk Factors for Gout

| Comorbidities |
| Cardiovascular disease |
| Diabetes mellitus |
| Diuretic use (loop and thiazide) |
| Elevated triglyceride and cholesterol levels |
| Hyperuricemia |
| Menopause |
| Obesity |
| Renal disease, including renal insufficiency and chronic kidney disease |
| Demographic factors |
| Certain ethnic groups, including indigenous Taiwanese, Pacific Islander, and New Zealand Maori |
| Living in high-income countries (specifically North America and western Europe) |
| Male sex (incidence 2 to 6 times higher than in females) |
| Dietary factors |
| Alcohol consumption |
| Diet rich in meat |
| Diet rich in seafood |
| Fructose-rich food and drink consumption |
Diagnosis
TABLE 2. Differential Diagnosis of Gout
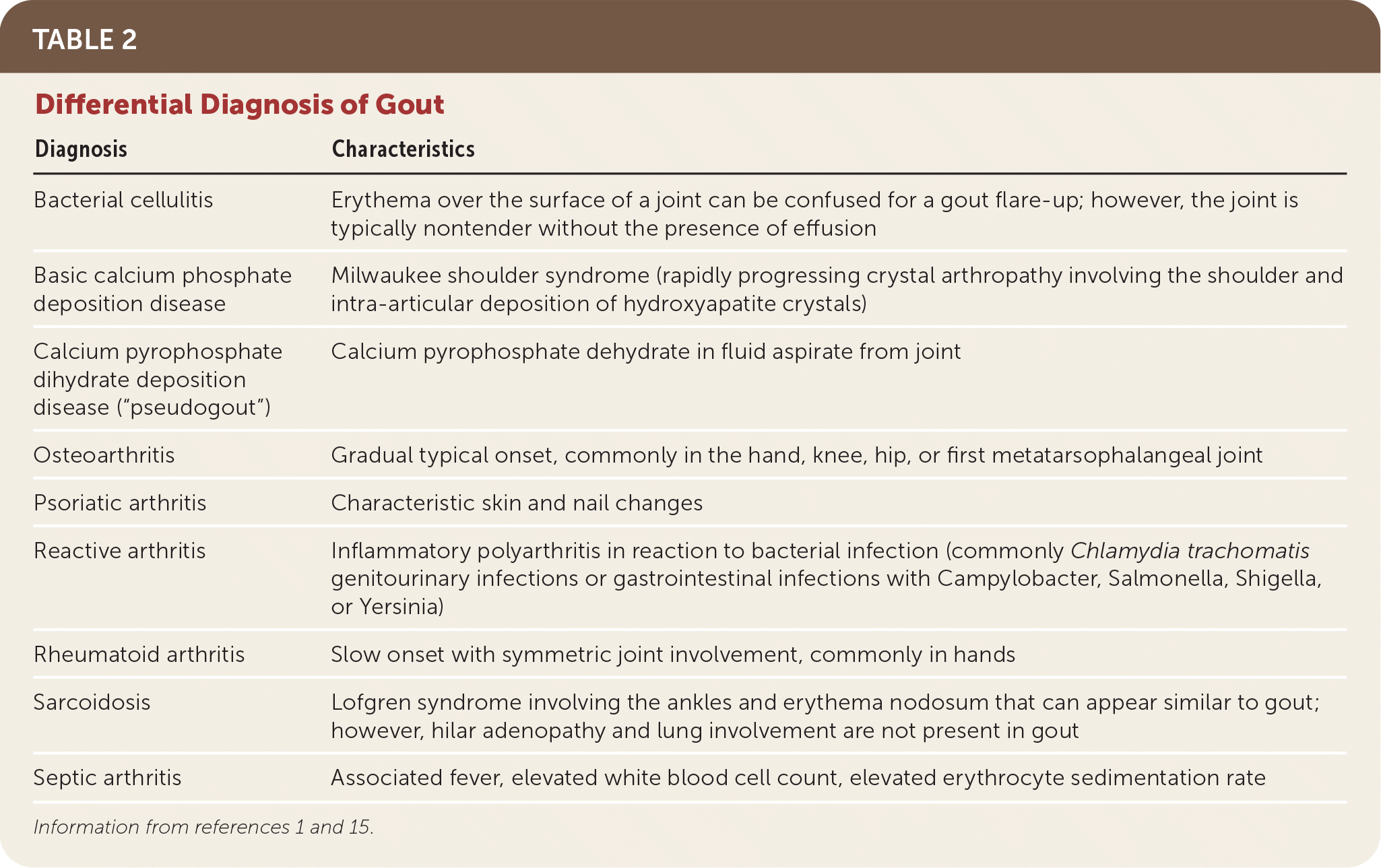
| Diagnosis | Characteristics |
|---|---|
| Bacterial cellulitis | Erythema over the surface of a joint can be confused for a gout flare-up; however, the joint is typically nontender without the presence of effusion |
| Basic calcium phosphate deposition disease | Milwaukee shoulder syndrome (rapidly progressing crystal arthropathy involving the shoulder and intra-articular deposition of hydroxyapatite crystals) |
| Calcium pyrophosphate dihydrate deposition disease (“pseudogout”) | Calcium pyrophosphate dehydrate in fluid aspirate from joint |
| Osteoarthritis | Gradual typical onset, commonly in the hand, knee, hip, or first metatarsophalangeal joint |
| Psoriatic arthritis | Characteristic skin and nail changes |
| Reactive arthritis | Inflammatory polyarthritis in reaction to bacterial infection (commonly Chlamydia trachomatis genitourinary infections or gastrointestinal infections with Campylobacter, Salmonella, Shigella, or Yersinia) |
| Rheumatoid arthritis | Slow onset with symmetric joint involvement, commonly in hands |
| Sarcoidosis | Lofgren syndrome involving the ankles and erythema nodosum that can appear similar to gout; however, hilar adenopathy and lung involvement are not present in gout |
| Septic arthritis | Associated fever, elevated white blood cell count, elevated erythrocyte sedimentation rate |
SIGNS AND SYMPTOMS
- Swelling, pain, and tenderness in a peripheral joint or bursa, most commonly the first metatarsophalangeal joint (56% to 78% of patients), are symptoms associated with gout. Other joints, including the midfoot (25% to 50%), ankle (18% to 60%), upper limb (13% to 46%), and interphalangeal joints (6% to 25%), may be involved.16
- Draining of a chalk-like substance from a subcutaneous nodule under transparent skin, often with overlying vascularity, indicates the presence of a tophus.1 Typical locations are the ear, olecranon bursa, and the finger pulps.
- Several validated clinical prediction rules do not require joint fluid analysis.17,18 One was derived in 338 patients presenting to their primary care physician with monoarthritis suspected to be gout; it was then prospectively validated in another 338 patients17,19 (Table 317 ).
- The American College of Rheumatology/European League Against Rheumatism 2015 gout classification criteria20 are available in an online tool (https://www.mdcalc.com/acr-eular-gout-classification-criteria).
TABLE 3. Clinical Prediction Rule for Gout
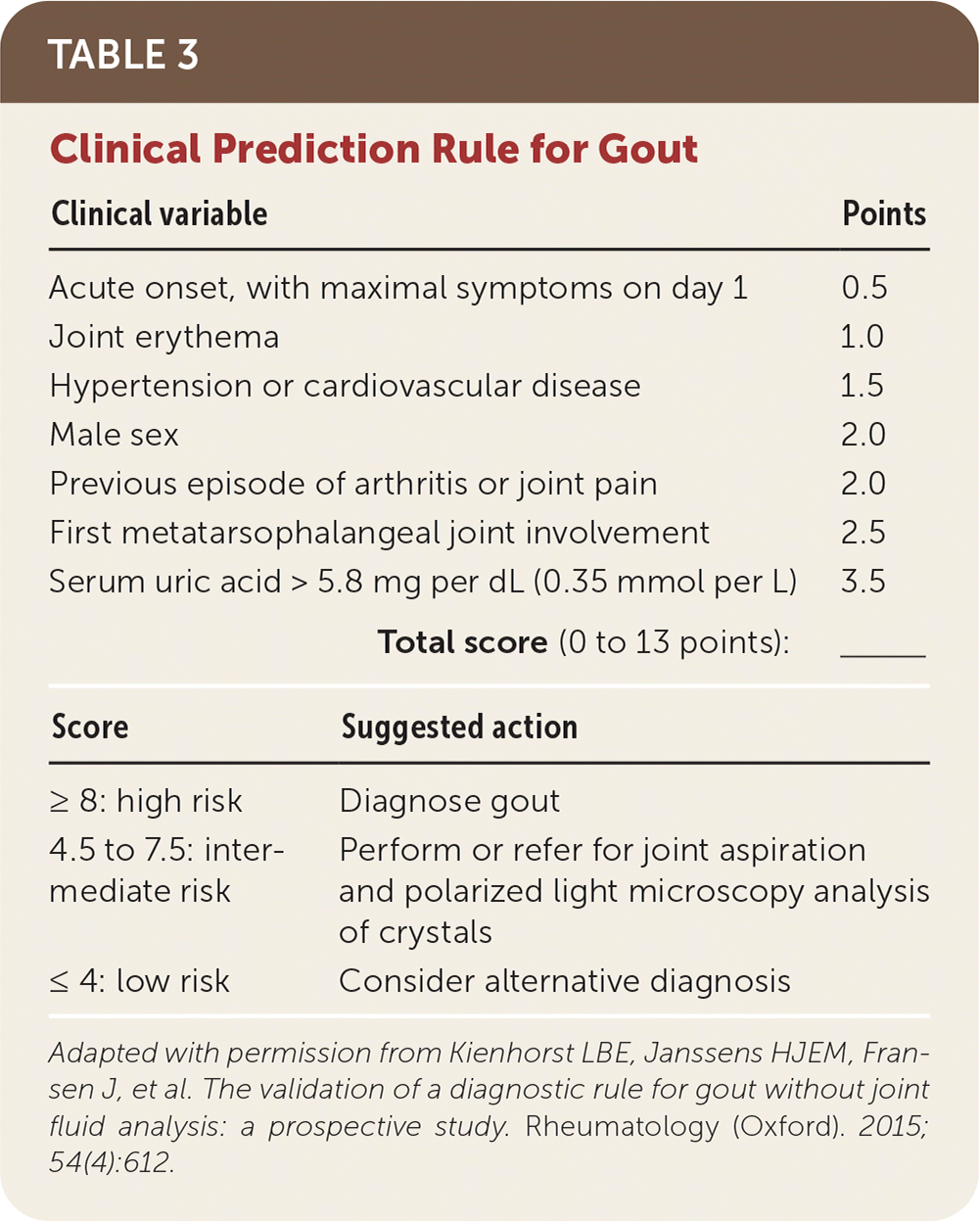
| Clinical variable | Points | |
|---|---|---|
| Acute onset, with maximal symptoms on day 1 | 0.5 | |
| Joint erythema | 1.0 | |
| Hypertension or cardiovascular disease | 1.5 | |
| Male sex | 2.0 | |
| Previous episode of arthritis or joint pain | 2.0 | |
| First metatarsophalangeal joint involvement | 2.5 | |
| Serum uric acid > 5.8 mg per dL (0.35 mmol per L) | 3.5 | |
| Total score (0 to 13 points): | ——— | |
| Score | Suggested action | |
| ≥ 8: high risk | Diagnose gout | |
| 4.5 to 7.5: intermediate risk | Perform or refer for joint aspiration and polarized light microscopy analysis of crystals | |
| ≤ 4: low risk | Consider alternative diagnosis | |
Adapted with permission from Kienhorst LBE, Janssens HJEM, Fransen J, et al. The validation of a diagnostic rule for gout without joint fluid analysis: a prospective study. Rheumatology (Oxford). 2015;54(4):612.
DIAGNOSTIC TESTING
- Diagnosis of gout is made clinically unless a septic joint is suspected based on acute onset of a hot, painful, red, tender, swollen joint with systemic symptoms such as fever or elevated white blood cell count.21
- Synovial fluid or tophus analysis demonstrating monosodium urate crystals by polarized light microscopy is the definitive test for gout but is recommended only when the diagnosis is uncertain (i.e., intermediate risk on acute gout diagnosis tool) or a septic joint is suspected.21
- If septic arthritis is suspected, blood culture and arthrocentesis with synovial fluid analysis (Gram stain and culture) should be performed.22,23
- Serum uric acid levels are usually elevated in people with gout; however, they may be lower during an acute episode and therefore should not be measured in this circumstance.24 In a pooled study of two randomized controlled trials (N = 339), 14% of patients had a serum uric acid level of less than 6 mg per dL (0.36 mmol per L) during an acute gout episode.25
- Ultrasonography and dual-energy computed tomography (CT) aid in the diagnosis of gout, whereas radiography and CT assess structural damage.26
- In a study of 824 patients, the presence of any one of three ultrasound findings (double contour sign, tophus, and snowstorm appearance) was moderately accurate when synovial fluid analysis was the reference standard (positive likelihood ratio = 4.8; negative likelihood ratio = −0.27).27
- Radiography of the affected joint is often normal with nonspecific soft tissue swelling.26
- Conventional CT may be used to visualize and measure tophi and to assess bony erosions.28
- Dual-energy CT can aid in detection of monosodium urate deposits in patients with gout. A diagnostic study of 40 patients with crystal-proven nontophaceous gout and 41 patients with other rheumatic diseases found that dual-energy CT was 90% sensitive and 83% specific.26,29
Treatment
Figure 1 presents a suggested approach to the treatment of gout.
Figure 1.
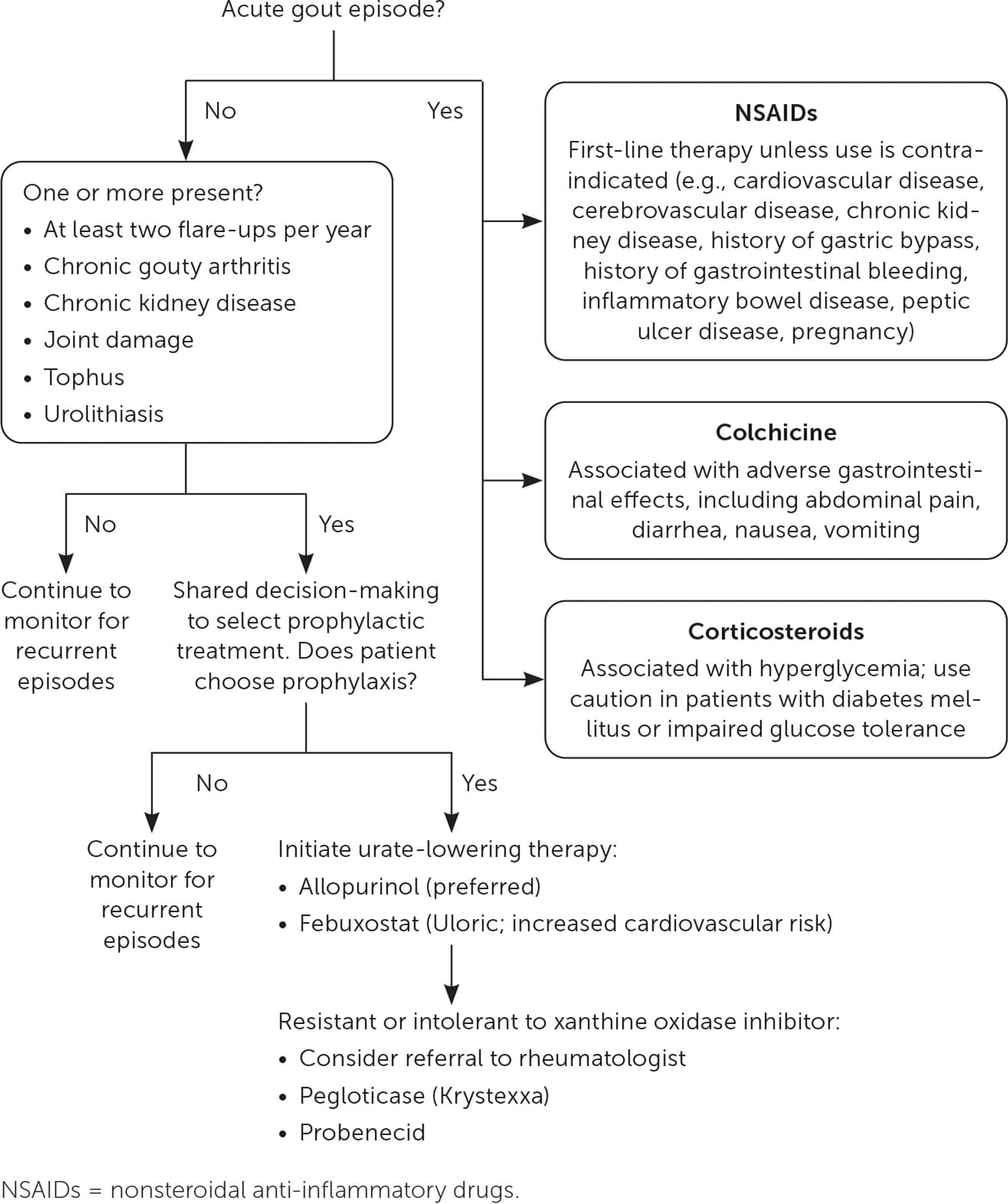
Treatment algorithm for gout.
LIFESTYLE THERAPY
- The Dietary Approaches to Stop Hypertension (DASH) diet was associated with a lower risk of developing gout in a prospective cohort study in men during 26 years of follow-up (N = 44,444; adjusted relative risk when comparing the highest fifth to the lowest fifth of the DASH dietary pattern score = 0.68; 95% CI, 0.57 to 0.80).30
- Behavior modifications should be recommended, including a low-fat, low-purine diet; avoidance of alcohol; weight loss if overweight; increased exercise; and avoidance of soft drinks and foods containing fructose. Consumption of skim milk and low-fat yogurt, vegetables, soybeans, vegetable sources of protein, and cherries is recommended.31
- Local ice therapy reduces pain in acute gouty arthritis.32
PHARMACOLOGIC THERAPY:ACUTE EPISODE
- Colchicine, nonsteroidal anti-inflammatory drugs, and corticosteroids relieve pain in adults with acute gout.33
- Nonsteroidal anti-inflammatory drugs are first-line treatment unless use is contraindicated (e.g., cardiovascular disease, cerebrovascular disease, chronic kidney disease, history of gastric bypass, history of gastrointestinal bleeding, inflammatory bowel disease, peptic ulcer disease, pregnancy).8
- Oral prednisolone (35 mg once daily) is equal to naproxen (500 mg twice daily) in the treatment of acute gout, with no significant difference in pain relief or adverse effects.34
- In acute gout, low-dose colchicine (1.2 mg orally followed by 0.6 mg one hour later) was as effective as high-dose colchicine (1.2 mg followed by 0.6 mg every hour for six hours) and had fewer adverse effects. Nearly 77% of patients treated with high-dose colchicine reported adverse effects (mostly diarrhea) compared with 36% in the low-dose colchicine group and 27% in the placebo group.35
- Further investigation is required to support routine use of interleukin-1 inhibitors (anakinra [Kineret], canakinumab [Ilaris], or rilonacept [Arcalyst]).36 Interleukin-1 inhibitors may benefit select patients, including those with frequent gout flare-ups resistant to standard treatments or in whom current therapies are contraindicated.
PHARMACOLOGIC THERAPY: CHRONIC MANAGEMENT AND PROPHYLAXIS
- The 2017 American College of Physicians guideline endorsed by the American Academy of Family Physicians recommends a discussion of the benefits, harms, costs, and individual preferences with patients who experience recurrent gout episodes before starting prophylactic treatment and urate-lowering therapy.21,37
- There are several indications for urate-lowering therapy31,37–39 (Figure 1).
- Allopurinol is the drug of choice for patients choosing urate-lowering therapy. The dosage of allopurinol will vary with the severity of disease, with a typical initial dosage of 100 mg orally once per day. The daily dosage should be titrated gradually (100-mg increments each week) to avoid precipitating a flare-up. Dosages of more than 300 mg per day are given in divided doses after meals, with a maximum of 800 mg per day.40
- Febuxostat (Uloric) is noninferior to allopurinol in preventing acute gout episodes.41,42 However, in a randomized, double-blind trial comparing allopurinol and febuxostat (N = 6,190), cardiovascular mortality was increased in the febuxostat group (4.3% vs. 3.2%; P = .03; number needed to harm over 2.7 years = 90). Similarly, all-cause mortality was higher in the febuxostat group compared with the allopurinol group (7.8% vs. 6.4%; P = .04; number needed to harm over 2.7 years = 71).41
- The American College of Physicians does not recommend monitoring 24-hour urinary excretion of uric acid or serum uric acid levels in patients taking standard doses of allopurinol or febuxostat.21 The British Society for Rheumatology recommends treating serum uric acid to less than 0.3 mmol per L (5 mg per dL) to prevent further uric acid crystal formation and to prevent recurrent flare-ups.31
- The European League Against Rheumatism and the American College of Rheumatology recommend prophylaxis while initiating urate-lowering therapy, although the recommended regimens differ.38,39,43
- Specialty organizations (the American College of Rheumatology and the Clinical Pharmacogenetics Implementation Consortium) recommend that the allopurinol hypersensitivity assay, or HLA-B*58:01 test, be considered in Korean adults with stage 3 or higher chronic kidney disease and in all adults of Han or Thai descent before initiating allopurinol therapy.39 This test is not recommended for other patients.44
- The U.S. Food and Drug Administration has approved pegloticase (Krystexxa), a pegylated recombinant form of urate-oxidase enzyme that converts uric acid to an inactive water-soluble form, for use in cases of refractory gout. Further study is needed to evaluate patient-oriented outcomes and adverse effects.45
CORTICOSTEROID INJECTIONS
- There is no evidence from clinical trials to support the use of intra-articular corticosteroid injection in the treatment of acute gout.46 It could be considered in patients for whom nonsteroidal anti-inflammatory drugs or oral steroids are contraindicated.
COMPLEMENTARY THERAPIES
Prognosis
This article updates previous articles by Hainer, et al.8; Eggebeen9; Pittman and Bross50; and Harris, et al.10
Data Sources: A PubMed search was completed using the MeSH terms gout and gout suppressants. This search included meta-analyses, randomized controlled trials, and reviews. The authors also searched the Cochrane database, Dynamed, and Essential Evidence Plus. Reference lists were also searched for additional articles. Search dates: February 24, 2020; April 25, 2020; and September 13, 2020.

