End-stage renal disease (ESRD) is diagnosed when kidney function is no longer adequate for long-term survival without kidney transplantation or dialysis. Primary care clinicians should refer people at risk of ESRD to nephrology to optimize disease management. Kidney transplantation typically yields the best patient outcomes, although most patients are treated with dialysis. The decision to initiate dialysis is best made through shared decision-making. Because most patients with ESRD elect to receive hemodialysis, the preservation of peripheral veins is important for those with stage III to V chronic kidney disease. A palliative approach to ESRD is a reasonable alternative to dialysis, particularly for individuals with limited life expectancy, with severe comorbid conditions, or who wish to avoid medical interventions. For patients with ESRD, vaccination against seasonal influenza, tetanus, hepatitis B, human papillomavirus (through 26 years of age), and Streptococcus pneumoniae is advised. Routine cancer screening for patients not receiving kidney transplantation is discouraged. Controlling blood pressure in patients receiving dialysis improves mortality. Volume control through adequate dialysis and sodium restriction can help optimize hypertension treatment in these patients. Insulin is the preferred treatment for patients with ESRD and diabetes mellitus requiring medication. Patients should be monitored for signs of protein-energy wasting and malnutrition. Clinicians must be aware of the many medical complications associated with ESRD.
End-stage renal disease (ESRD) is when kidney function is no longer adequate for long-term survival without kidney transplantation or dialysis.1,2 The estimated glomerular filtration rate (GFR) is usually less than 15 mL per minute per 1.73 m2 when this occurs.3 Kidney failure, a more concise term, may soon replace ESRD.4
SORT: KEY RECOMMENDATIONS FOR PRACTICE
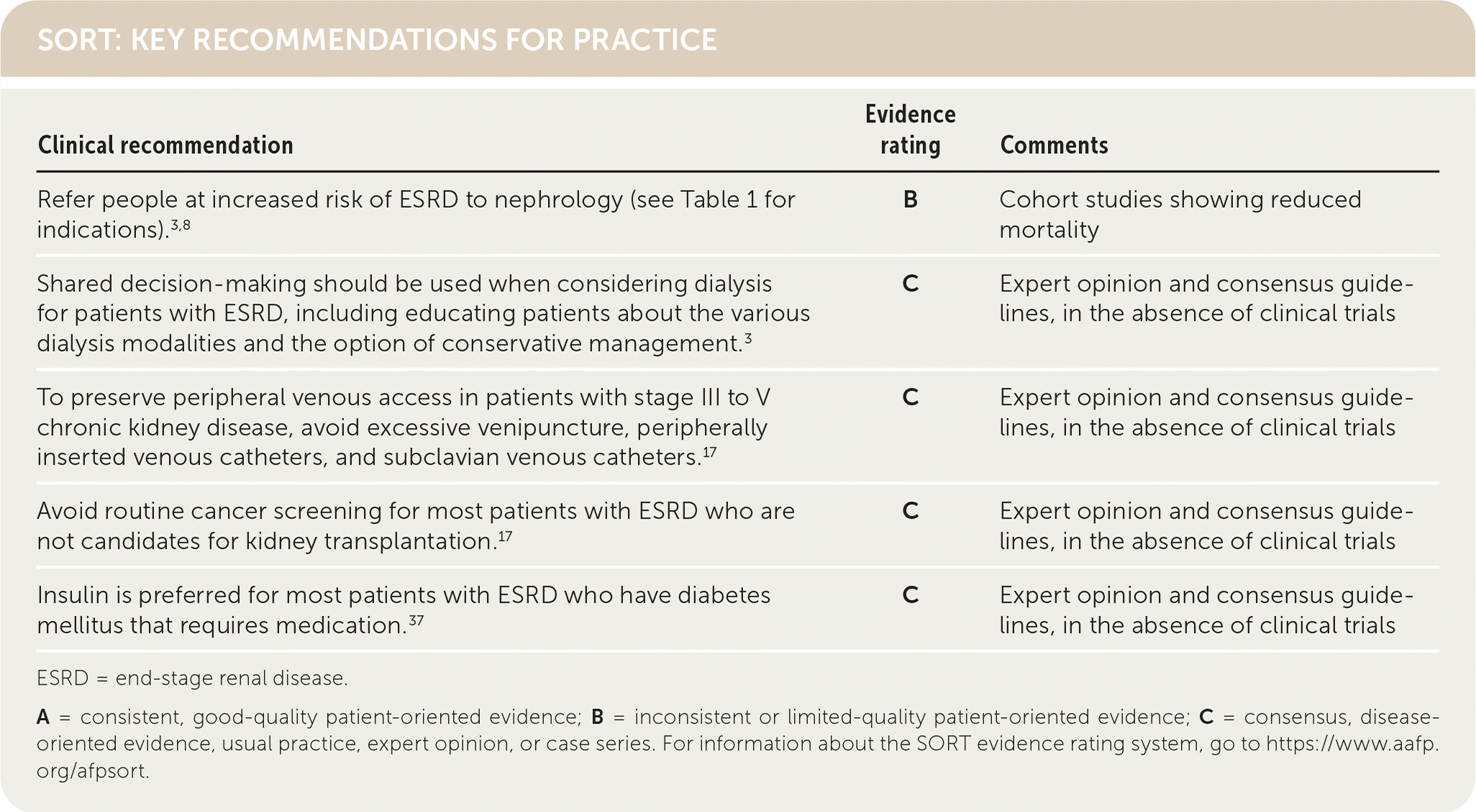
ESRD = end-stage renal disease.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
BEST PRACTICES IN NEPHROLOGY

| Recommendation | Sponsoring organization |
|---|---|
| Do not perform routine cancer screening in asymptomatic patients receiving dialysis if they have a limited life expectancy. | American Society of Nephrology |
| Do not place peripherally inserted central catheters in patients with stage III to V chronic kidney disease without consulting nephrology. | American Society of Nephrology |
| Do not initiate chronic dialysis without ensuring a shared decision-making process between patients, their families, and their physicians. | American Society of Nephrology |
Source: For more information on the Choosing Wisely Campaign, see https://www.choosingwisely.org. For supporting citations and to search Choosing Wisely recommendations relevant to primary care, see https://www.aafp.org/afp/recommendations/search.htm.
The incidence of ESRD increased more than threefold between 1980 and 2000 because of increasing numbers of patients with diabetes mellitus, hypertension, and related conditions. Although this increase has leveled off, the prevalence of ESRD has steadily increased, largely because of longer patient survival. By 2018, there were more than 750,000 individuals with ESRD in the United States. The disease is costly, accounting for approximately 10% of Medicare fee-for-service spending. It is also associated with high mortality; fewer than one-half of those who initiate hemodialysis survive for five years.5
Primary care clinicians play a key role in diagnosing chronic kidney disease, monitoring its progression, treating modifiable risk factors, and identifying and treating complications. The evaluation of chronic kidney disease was discussed in a previous issue of American Family Physician (AFP).6 ESRD often develops slowly and can be prevented in many cases. This article provides an overview of the medical management of ESRD, as well as its comorbidities and complications.
Nephrology Referral
Early nephrology referral for patients at increased risk of ESRD is vital because it is associated with improved patient-centered outcomes, including mortality.3,7 Approximately one-third of all patients receive little to no nephrology care before ESRD is diagnosed.5,8
Patients with chronic kidney disease should be referred to nephrology if their estimated GFR falls below 30 mL per minute per 1.73 m2.3 A full list of indications for referral is provided in Table 1.3
TABLE 1. Indications for Nephrology Referral in Patients with Chronic Kidney Disease

| Abrupt, sustained decrease in estimated GFR |
| Estimated GFR < 30 mL per minute per 1.73 m2 |
| Hereditary kidney disease |
| Persistent significant albuminuria (albumin-to-creatinine ratio 300 mg per g [≥ 30 mg per mmol] or albumin excretion rate ≥300 mg per 24 hours) |
| Persistent hypokalemia or hyperkalemia |
| Progression of chronic kidney disease (progressive decline in estimated GFR, particularly if decline is > 5 mL per minute per 1.73 m2 per year) |
| Recurrent or extensive nephrolithiasis |
| Resistant hypertension |
| Risk of end-stage renal disease within one year is 10% to 20% or higher as determined by a validated risk prediction tool, such as the Kidney Failure Risk Calculator (https://www.mdcalc.com/kidney-failure-risk-calculator) |
| Urinary red cell casts or persistent red blood cells > 20 per high power field (if otherwise unexplained) |
GFR = glomerular filtration rate.
Information from reference 3.
Goals of early referral include initiating disease-specific therapies; slowing the progression of chronic kidney disease; evaluating and treating comorbid conditions and complications; providing psychosocial support; and planning for kidney transplantation, dialysis, or conservative kidney management.3 Multidisciplinary care, involving primary care and other clinicians, pharmacists, nurses, dietitians, and social workers, may improve patient outcomes.9
Kidney Transplantation
A key consideration for patients with ESRD is establishing eligibility for kidney transplantation, which, compared with dialysis or conservative management, improves survival and quality of life.10 Referral to a transplantation program is advised when estimated GFR falls below 30 mL per minute per 1.73 m2 because receiving a transplant before dialysis is needed improves survival.11 Early referral allows time for medical and psychosocial evaluation, treatment of modifiable risk factors, and identification of a living donor; it also maximizes accrual of wait time on the transplant waiting list.10 The median wait time for a transplant is four years, and currently only 5% of patients who initiate dialysis were preemptively placed on the kidney transplant waiting list.5
Dialysis
Most patients elect to receive dialysis to treat their ESRD,5 and these patients tend to live longer than those choosing conservative management.12 Yet, because of the time commitment, discomfort, and complications associated with dialysis, shared decision-making should be used, with adequate time for patients to consider the various dialysis modalities and the option of conservative management.3 Many patients do not receive adequate education before starting dialysis, and one survey showed that 61% of patients who chose dialysis later regretted the decision.8
In the United States, nearly 90% of patients with ESRD are treated with center-based hemodialysis, but many patients are unaware that other options exist.5,13 Alternatives include in-home hemodialysis and peritoneal dialysis, which can provide flexibility in frequency and duration of dialysis sessions. Patients who receive targeted education before the initiation of dialysis are more likely to choose peritoneal dialysis.13–15
Alone, GFR does not determine when dialysis is initiated; instead, guidelines advise starting dialysis when irreversible signs or symptoms of ESRD manifest.3,14,16 Common indications for dialysis include persistent volume overload; refractory metabolic disorders such as metabolic acidosis and hyperkalemia; cognitive impairment; and worsening nutritional status.3 Table 2 lists indications for initiation of dialysis.3,14
TABLE 2. Indications for Initiating Dialysis

| Refractory acidosis, potassium disorders, or hyperphosphatemia | |
| Refractory volume overload | |
| Signs or symptoms of uremia* Alterations in sense of taste or smell Amenorrhea Anorexia Bleeding/bruising Confusion Encephalopathy Hiccups Insulin resistance Lethargy/fatigue Muscle cramps Nausea Protein-energy wasting Pruritus Reduced body core temperature Restless legs Seizures Serositis (pleuritis, pericarditis) Sleep disturbance Somnolence | |
| Uncontrolled hypertension |
*—The signs and symptoms of uremia are nonspecific and may require further evaluation to determine their etiology.
Adapted with permission from KDOQI Clinical Practice Guideline for Hemodialysis Adequacy: 2015 update [published correction appears in Am J Kidney Dis. 2016;67(3):534]. Am J Kidney Dis. 2015; 66(5):901, with additional information from reference 3.
Most patients with ESRD will be treated with hemodialysis, and arteriovenous access (fistula or graft) is usually preferred. Early referral allows for evaluation and creation of vascular access before dialysis is needed. For patients with stage III to V chronic kidney disease, strategies to preserve peripheral venous access are important. These include avoiding excessive venipuncture, peripherally inserted central venous catheters, and subclavian venous catheters.17 Once arteriovenous access is established, patients and clinicians should monitor the site for signs of infection, stenosis, and other complications (e.g., examining for tenderness, erythema, and swelling and confirming the presence of a palpable thrill).18
Conservative Kidney Management
Patients with ESRD can elect a palliative approach to managing their disease that does not involve dialysis. This approach emphasizes quality of life over the prolongation of life. Although dialysis offers a survival benefit, that benefit decreases, and may disappear, with increasing patient age and comorbidity.12 The overall symptom burden does not substantially improve in patients treated with dialysis, and they are more likely to receive medical interventions and to be hospitalized.19,20 Conservative kidney management may be a reasonable alternative to dialysis, particularly for individuals with limited life expectancy or severe comorbid conditions and for those who strongly wish to avoid medical interventions. This topic was discussed further in a previous issue of AFP.21
Other Considerations
VACCINATION
Because of immune suppression and increased susceptibility to infection in patients with ESRD, the Centers for Disease Control and Prevention (CDC) provides specific vaccination recommendations for this population. Routine vaccinations against seasonal influenza and tetanus should be continued in these patients. Additionally, the CDC recommends hepatitis B and pneumococcal vaccination (23-valent pneumococcal polysaccharide vaccine [Pneumovax-23] and 13-valent pneumococcal conjugate vaccine [Prevnar-13]).22,23 The human papillomavirus vaccine should be offered to men and women through 26 years of age.24 When vaccinating patients with ESRD against the herpes zoster virus, the recombinant vaccine (Shingrix) is preferred.25 Chronic kidney disease is considered a risk factor for severe COVID-19, and vaccination is advised.25 Updated vaccination guidance from the Advisory Committee on Immunization Practices can be found on the CDC website at https://www.cdc.gov/vaccines/hcp/acip-recs/index.html.
CANCER SCREENING
Given the limited life expectancy for most patients with ESRD who do not undergo kidney transplantation, routine cancer screening is discouraged in these patients.17,26
NUTRITION
People with ESRD are at high risk of developing protein-energy wasting and other malnutrition disorders and should receive nutritional counseling from a registered dietitian. Protein-energy wasting is strongly associated with increased mortality and other adverse outcomes.27,28 Signs of protein-energy wasting include body mass index less than 23 kg per m2, unintentional weight loss (5% or more over three months or 10% or more over six months), and a serum albumin level less than 3.8 g per dL (38 g per L).29 Although modest protein restriction may help prevent the progression of chronic kidney disease to ESRD, once ESRD develops, patients are typically counseled to consume a relatively high-protein diet (1.0 to 1.2 g of protein per kg of body weight per day).28 Dietary protein supplements are often prescribed to patients with protein-energy wasting. Routine micronutrient supplementation is not necessary but should be considered when dietary intake is inadequate or signs or symptoms of specific micronutrient deficiencies are present.28
HYPERTENSION
Most patients receiving dialysis have hypertension. Because blood pressure control is closely associated with volume status, modifying dialysis sessions to maintain normovolemia can improve blood pressure control.30,31 Likewise, sodium restriction (less than 2 g per day) can reduce blood pressure by limiting fluid retention, but patient compliance is usually poor.32
Blood pressure measurements provide more accurate prognostic information when obtained outside the dialysis setting (such as ambulatory or home blood pressure monitoring).33 Antihypertensive therapy in patients receiving dialysis improves mortality, although target blood pressure values and the optimal first-line antihypertensive medications have not been established.34
TYPE 2 DIABETES
Careful glucose monitoring in patients with ESRD and diabetes is important because insulin requirements are difficult to predict and the risk of hypoglycemia is increased in these patients.35 Hyperglycemia may resolve when patients start dialysis.36 The optimal A1C goal for patients with ESRD has not been established; however, maintaining an A1C between 6% and 9% may reduce mortality.35,36 The accuracy and precision of A1C measurements are reduced in patients with ESRD, particularly when undergoing dialysis. Therefore, continuous glucose monitoring is a reasonable alternative, especially when there is substantial discordance between fasting glucose measurements and A1C measurements.35
Insulin is preferred for most patients who require medication, although glipizide (Glucotrol) and repaglinide are acceptable oral alternatives.37 When estimated GFR falls below 30 mL per minute per 1.73 m2, many diabetes medications, including metformin, are contraindicated.37
Medical Complications of ESRD
Clinicians caring for patients with ESRD should be aware of its many complications. Patients with ESRD are at increased risk of bleeding due to platelet dysfunction, although dialysis may attenuate this risk.38 Anemia (hemoglobin less than 13 g per dL [130 g per L] in men and less than 12 g per dL [120 g per L] in women) is common. Treatment includes intravenous iron for patients who are iron deficient and erythropoiesis-stimulating agents for select patients who are not iron deficient.39 Patients who are not yet anemic should be monitored for anemia every three months.39
Chronic metabolic acidosis is typically resolved with dialysis, but because the condition is associated with increased mortality and other adverse outcomes, those with persistently low bicarbonate concentrations (less than 22 mEq per L [22 mmol per L]) may require oral bicarbonate supplementation.3 Potassium excretion by the kidneys is decreased in patients with ESRD, putting them at high risk of hyperkalemia and its complications.40 Secondary hyperparathyroidism is also common; therefore, serum calcium and phosphate levels should be measured every one to three months, parathyroid hormone levels every three to six months, and alkaline phosphatase levels at least every 12 months.41 Patients with ESRD should also be assessed for vitamin D deficiency.41
Although rare, calciphylaxis is a life-threatening condition that may occur in patients with ESRD. The condition is characterized by small vessel occlusions in skin and adipose tissue that present as painful necrotic skin lesions.42
Chronic kidney disease is considered a coronary heart disease risk equivalent, and patients with ESRD are at high risk of cardiovascular complications and death.43 Although statin therapy can continue during dialysis treatments if already established, initiating statin therapy has not been shown to improve outcomes.44 Aspirin is likely beneficial for individuals at very high risk of atherosclerotic events (e.g., those with recent myocardial infarction). However, there is a lack of conclusive evidence regarding the role of aspirin therapy for secondary prevention of cardiovascular disease in patients with ESRD.45,46
Pericarditis is not uncommon in patients with ESRD and can manifest differently in this population. Patients with ESRD are more likely to present with constitutional symptoms, such as fever, chills, and malaise, and electrocardiography often does not show classic widespread ST elevations.47
This article updates a previous article on this topic by O'Connor and Corcoran.21
Data Sources: A PubMed search was completed in Clinical Queries using the key terms end-stage renal disease, end-stage kidney disease, and kidney failure. The search included meta-analyses, randomized controlled trials, and reviews. Also searched were the Agency for Healthcare Research and Quality Effective Healthcare Reports, the Cochrane database, and Essential Evidence Plus. Search date: October 3, 2020, and July 9, 2021.

