To the Editor: I appreciated the article on medical management of heart failure with reduced ejection fraction.1 The number needed to treat (NNT) is provided in reference to the effectiveness of a medication on three separate occasions. For angiotensin-converting enzyme (ACE) inhibitors, the NNT to avoid one death (all-cause mortality) is 50 over 6 months and 63 over 12 months. An angiotensin receptor-neprilysin inhibitor has an NNT of 32 to prevent one cardiovascular death. For sodium-glucose cotransporter-2 inhibitors, the NNT is used in two instances: one combining all-cause mortality with worsening of first episode of heart failure and the other combining cardiovascular death with heart failure hospitalization. None of these NNTs are interchangeable with respect to the outcome or duration required to achieve these outcomes, making it challenging for the reader to compare various interventions on equal terms.
Standardizing NNT to all-cause mortality is preferable because it is easily measurable and allows inclusion of patients who may die early due to medication exposure. Specifying NNT to a standard time frame would be preferable for making comparisons. I recommend using the 1-year death NNT (ie, outcome time [years]/absolute risk reduction).2 It expresses the number of patients who would need to take the treatment for 1 year to prevent one death. The outcome time in years is the median duration of the study. An example of how the data regarding NNT could be presented is demonstrated in Table 1.3,4
TABLE 1. Sample of How to Present NNT Data
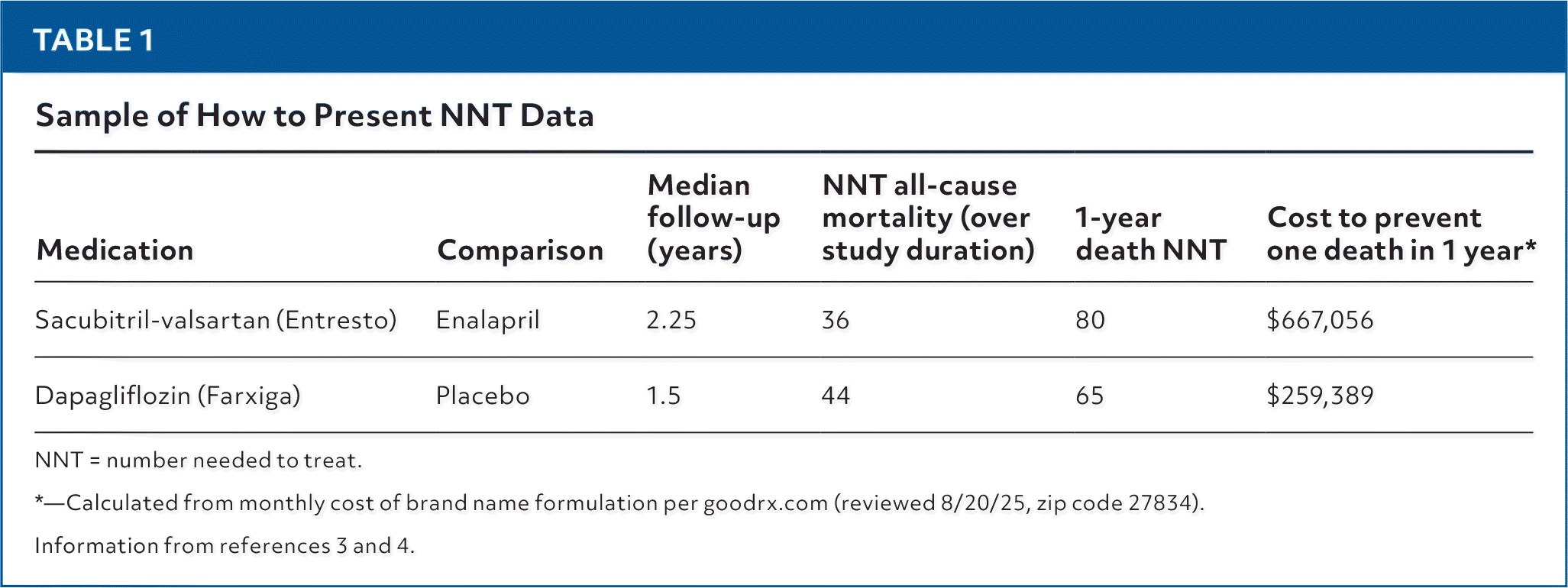
| Medication | Comparison | Median follow-up (years) | NNT all-cause mortality (over study duration) | 1-year death NNT | Cost to prevent one death in 1 year* |
|---|---|---|---|---|---|
| Sacubitril-valsartan (Entresto) | Enalapril | 2.25 | 36 | 80 | $667,056 |
| Dapagliflozin (Farxiga) | Placebo | 1.5 | 44 | 65 | $259,389 |
NNT = number needed to treat.
*—Calculated from monthly cost of brand name formulation per goodrx.com (reviewed 8/20/25, zip code 27834).
A study on empagliflozin (Jardiance) does not report all-cause mortality.5 Using NNT as shown in Table 1 allows comparison of interventions from separate trials in a standardized fashion and an understanding of cost vs benefit.3,4
There are weaknesses to this approach, one being the assumption that the intervention studied impacts death in a linear fashion. Patients may experience benefits from pharmaceutical interventions in a logarithmic or exponential fashion, whereby use of the 1-year death NNT would over- or underestimate the effectiveness. Acknowledging this weakness, the 1-year death NNT is an improvement over the use of random and varied NNTs that do not allow the reader to directly compare outcomes of various medications.
In Reply: I appreciate your insightful comment on standardizing the reporting of NNT. This has been a difficult area given the decades-long evolution of heart failure management, from the introduction of ACE inhibitors in 1987 to SGLT-2 inhibitors in 2019. Study heterogeneity, including variation in trial duration (10–42 months) and reliance on hazard ratios rather than absolute risk reduction, complicates consistent NNT calculations. For example, a 2022 systematic review of pharmacologic therapy in heart failure with reduced ejection fraction reported hazard ratios for all-cause mortality as follows: angiotensin receptor-neprilysin inhibitors: 0.75 (95% CI, 0.66–0.85), mineralocorticoid receptor antagonists: 0.76 (95% CI, 0.67–0.85), beta blockers: 0.78 (95% CI, 0.72–0.84), ACE inhibitors: 0.89 (95% CI, 0.82–0.96), SGLT-2 inhibitors: 0.88 (95% CI, 0.78–0.99), and angiotensin receptor blockers (ARBs): 0.95 (95% CI, 0.88–1.02).1 Importantly, NNT cannot be derived from relative risk alone without knowing the absolute risk in the control group, and hazard ratios add complexity by incorporating time-to-event analysis.
A 2025 study reported 12-month all-cause mortality NNT for individual classes of guideline-directed medical therapy (GDMT): 37 for ACE inhibitors, 78 for ARBs, and 18 for beta blockers, although SGLT-2 inhibitors and mineralocorticoid receptor antagonists were not included.2 A 2024 article reported an NNT of 28 for beta blockers, 77 for ACE inhibitor/ARBs, 38 for angiotensin receptor-neprilysin inhibitors, 59 for mineralocorticoid receptor antagonists, and 63 for SGLT-2 inhibitors.3 These discrepancies highlight how trial populations, duration, and methodology influence reported NNT.
In contemporary clinical practice, optimal management of heart failure with reduced ejection fraction prioritizes the integrated use of all four classes of GDMT. Accordingly, the NNT for combined therapy provides a more clinically relevant measure of therapeutic benefit than an NNT derived from individual drug classes in isolation. A 2024 retrospective cohort study based on the Get With the Guidelines - Heart Failure registry sponsored by the American Heart Association strengthened the importance of initiating quadruple therapy before hospital discharge.4 By applying relative risk reductions from pivotal clinical trials, investigators estimated that complete implementation of all four GDMT classes projected to yield an absolute reduction in 12-month mortality of 10.4% (NNT = 10) compared with therapy limited to ACE inhibitor/ARBs and beta blockers, and 24.8% (NNT = 4) compared with no GDMT.4 Emphasizing this combined benefit, rather than focusing solely on individual drug classes, better reflects real-world treatment strategies. Ultimately, the key message is that combined GDMT markedly reduces morbidity, heart failure readmissions, and mortality.

