Bronchiolitis is a common lower respiratory tract infection in infants and young children, and respiratory syncytial virus (RSV) is the most common cause of this infection. RSV is transmitted through contact with respiratory droplets either directly from an infected person or self-inoculation by contaminated secretions on surfaces. Patients with RSV bronchiolitis usually present with two to four days of upper respiratory tract symptoms such as fever, rhinorrhea, and congestion, followed by lower respiratory tract symptoms such as increasing cough, wheezing, and increased respiratory effort. In 2014, the American Academy of Pediatrics updated its clinical practice guideline for diagnosis and management of RSV bronchiolitis to minimize unnecessary diagnostic testing and interventions. Bronchiolitis remains a clinical diagnosis, and diagnostic testing is not routinely recommended. Treatment of RSV infection is mainly supportive, and modalities such as bronchodilators, epinephrine, corticosteroids, hypertonic saline, and antibiotics are generally not useful. Evidence supports using supplemental oxygen to maintain adequate oxygen saturation; however, continuous pulse oximetry is no longer required. The other mainstay of therapy is intravenous or nasogastric administration of fluids for infants who cannot maintain their hydration status with oral fluid intake. Educating parents on reducing the risk of infection is one of the most important things a physician can do to help prevent RSV infection, especially early in life. Children at risk of severe lower respiratory tract infection should receive immunoprophylaxis with palivizumab, a humanized monoclonal antibody, in up to five monthly doses. Prophylaxis guidelines are restricted to infants born before 29 weeks' gestation, infants with chronic lung disease of prematurity, and infants and children with hemodynamically significant heart disease.
Bronchiolitis is a common lower respiratory tract infection in infants and young children, and respiratory syncytial virus (RSV) is the most common cause. Although the exact mechanism is unclear, it is likely that direct viral cytotoxic injury has a role in the pathogenesis of RSV infections. This leads to necrosis of the epithelial cells of the small airways, and the sloughed cells and mucus cause plugging of the bronchioles that leads to hyperinflation and atelectasis.1–3 RSV bronchiolitis typically affects children in the first two years of life. In 2014, the American Academy of Pediatrics updated its clinical practice guideline on RSV bronchiolitis to minimize unnecessary diagnostic testing and interventions.4 This guideline was accepted by the American Academy of Family Physicians and incorporated in its updated policy.5
SORT: KEY RECOMMENDATIONS FOR PRACTICE

| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Routine viral testing and chest imaging are not recommended for patients with presumed RSV bronchiolitis. | B | 4, 15, 16 |
| Bronchodilators, systemic or inhaled corticosteroids, and epinephrine should not be administered to infants and children with bronchiolitis. | A | 4, 24, 28, 30 |
| Antibiotics should not be administered to children with RSV bronchiolitis unless a bacterial infection is confirmed or suspected. | B | 4, 37 |
| Palivizumab (Synagis) should be given in the first year of life to infants born before 29 weeks' gestation or to infants born before 32 weeks' gestation who have chronic lung disease. | B | 42 |
RSV = respiratory syncytial virus.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
BEST PRACTICES IN INFECTIOUS DISEASE: RECOMMENDATIONS FROM THE CHOOSING WISELY CAMPAIGN
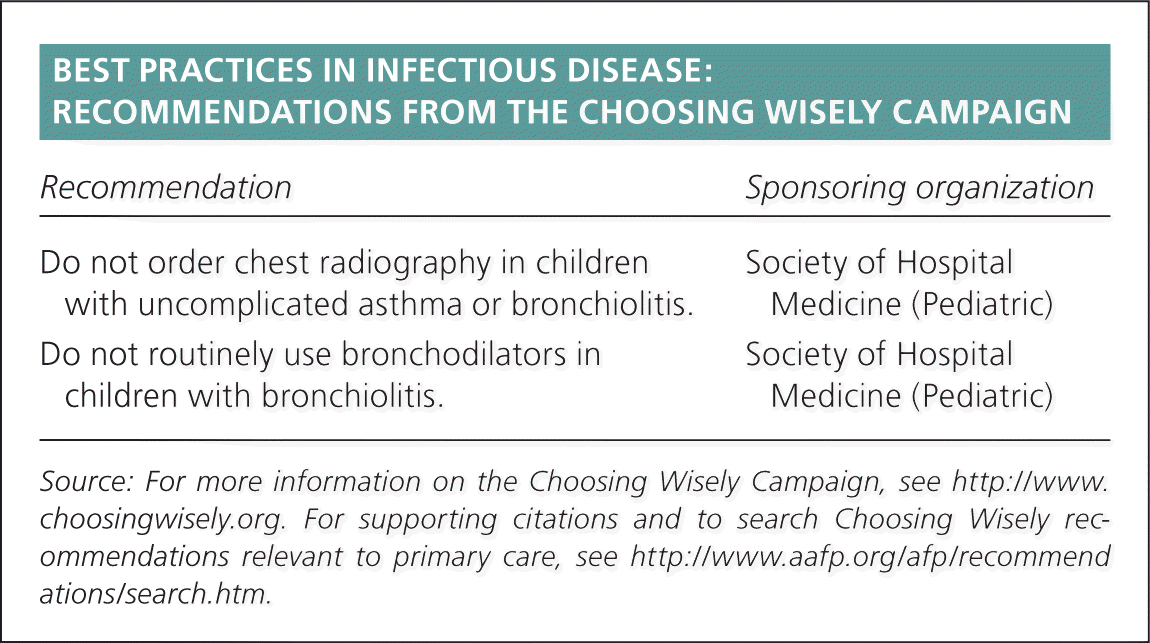
| Recommendation | Sponsoring organization |
|---|---|
| Do not order chest radiography in children with uncomplicated asthma or bronchiolitis. | Society of Hospital Medicine (Pediatric) |
| Do not routinely use bronchodilators in children with bronchiolitis. | Society of Hospital Medicine (Pediatric) |
Source: For more information on the Choosing Wisely Campaign, see http://www.choosingwisely.org. For supporting citations and to search Choosing Wisely recommendations relevant to primary care, see https://www.aafp.org/afp/recommendations/search.htm.
Epidemiology and Natural History
There are seasonal differences in the incidence of RSV infection in the United States, although the highest incidence occurs from December to March.6 Nearly all children have been infected with RSV at least once by two years of age. Most children have a self-limited course that responds to supportive home care; however, approximately 2% to 3% of infants younger than 12 months are hospitalized with an RSV infection each year in the United States.7 It is estimated that more than 57,500 hospitalizations and 2.1 million outpatient visits are associated with RSV infections each year in U.S. children younger than five years.6 The strongest predictor of hospitalization is chronologic age, with the highest rate of hospitalization occurring in the first 90 days of life.8
Although RSV infection is the most common cause of bronchiolitis, there are many other viral pathogens that can lead to bronchiolitis. A large multicenter, multiyear prospective study found that 30% of hospitalized children had multiple pathogen infections with RSV, with human rhinovirus being the most common.9 It is unclear if having multiple pathogen infection increases the severity of the clinical course.9 Most children recover uneventfully, although underlying chronic lung disease, congenital anomalies, immunocompromised state, and hemodynamically significant heart disease in patients with bronchiolitis are associated with progression to severe disease or mortality.4 There are few long-term risks associated with bronchiolitis, but infection with rhinovirus or severe RSV bronchiolitis is associated with an increased risk of asthma that can persist into adulthood.10,11
Clinical Manifestations
Patients with RSV bronchiolitis usually present with two to four days of upper respiratory tract symptoms, such as fever, rhinorrhea, and congestion, followed by lower respiratory tract symptoms. Increasing cough is usually the first sign of lower respiratory tract involvement; later symptoms include tachypnea, dyspnea, increased respiratory effort, and difficulty feeding.1,2 Patients with a normal respiratory rate are at low risk of significant viral or bacterial lower respiratory tract infection or pneumonia, although the presence of tachypnea cannot be used to distinguish between viral and bacterial infections.4 Physical examination findings usually include auscultation of wheezing and crackles, and may include evidence of increased respiratory effort such as grunting, nasal flaring, or retractions.1,2 In infants presenting with lower respiratory tract symptoms, the history and physical examination findings help distinguish viral bronchiolitis from other causes and can help estimate disease severity.4
Common predictors of the need for hospital admission are shown in Table 1.12,13 Another study found that age of two months or more, no history of intubation, a history of eczema, lower respiratory rate, no or only mild retractions, initial oxygen saturation of at least 94%, and adequate oral intake predicted safe discharge.13 Apnea may be an early manifestation of RSV bronchiolitis and other viral etiologies, and is more common in infants two to eight weeks of age and in those with birth weight less than 5 lb, 1 oz (2,300 g).14 When assessing disease severity, an important factor is the effect of the patient's symptoms on mental status, feeding, and hydration.4
Table 1. Predictors of Severity in Bronchiolitis
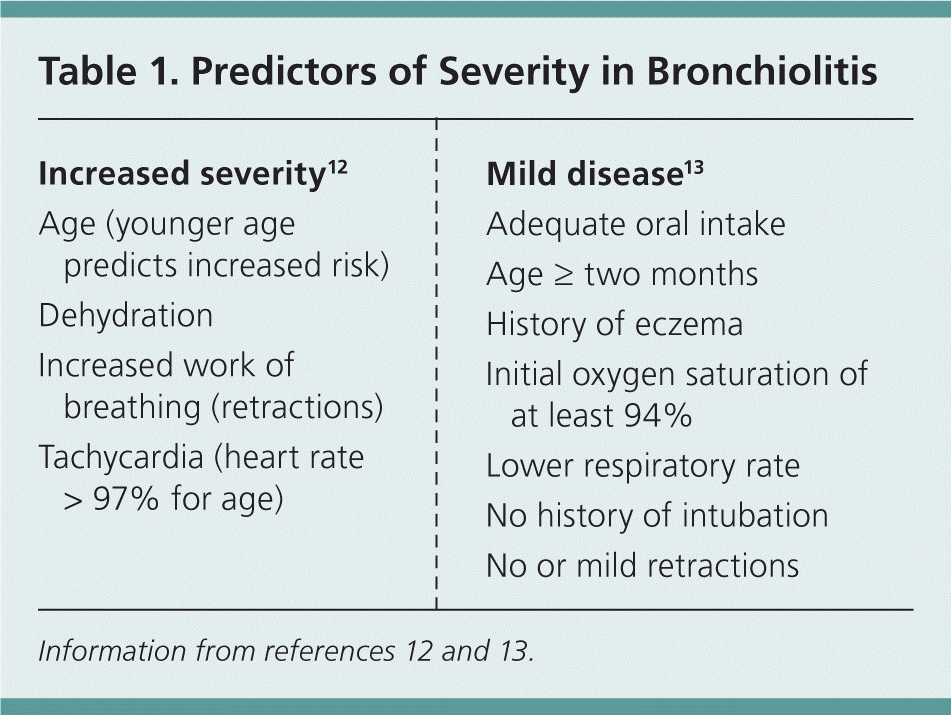
| Increased severity12 | Mild disease13 |
|---|---|
| Age (younger age predicts increased risk) | Adequate oral intake |
| Dehydration | Age ≥ two months |
| Increased work of breathing (retractions) | History of eczema |
| Tachycardia (heart rate > 97% for age) | Initial oxygen saturation of at least 94% |
| Lower respiratory rate | |
| No history of intubation | |
| No or mild retractions |
Diagnostic Testing
RSV bronchiolitis has a more severe clinical course than non-RSV bronchiolitis, including a longer hospital stay.15 However, virologic testing on an individual basis is insufficient to predict outcomes and does not affect management decisions.15 Therefore, RSV testing should not be routinely performed except in hospitalized infants who are receiving monthly prophylaxis (so that further prophylaxis can be discontinued because of the low risk of having a second RSV infection in the same year).4,15 Chest radiography findings in patients with bronchiolitis include peribronchial markings, hyperinflation, and atelectasis. However, chest radiography should not be performed routinely because it does not improve clinical outcomes and is associated with increased antibiotic use.15,16 Pulse oximetry is often used to identify children with hypoxemia, although it has been implicated in increased rates of hospital admission with no other clinical outcome data supporting its use.4,17,18 Use of a bronchiolitis guideline in the emergency department may decrease chest radiography, RSV testing, and albuterol use, as well as length of hospital stay and total cost.19
Treatment
The mainstay of therapy for acute RSV bronchiolitis in infants and children is supportive care (Table 2). Oxygen saturation of 90% or more is sufficient for children with bronchiolitis.4 No data support the use of supplemental oxygen to maintain higher oxygen saturation, and doing so only prolongs hospitalization because of an assumed need for oxygen.20 Furthermore, any transient desaturation is considered normal in otherwise healthy infants.21 A small study showed that high-flow nasal cannula oxygen may be a viable option for infants with persistent hypoxemia (oxygen saturation less than 90% with fraction of inspired oxygen of 0.4, or respiratory rate of 60 breaths or more per minute).22
Table 2. Treatment for Acute Respiratory Syncytial Virus Bronchiolitis

| Recommended therapies |
| Maintain nutrition and hydration status |
| Maintain oxygen saturation ≥ 90% |
| Therapies not routinely recommended |
| Antibiotics |
| Bronchodilators |
| Chest physiotherapy |
| Epinephrine |
| Excessive nasal suction of secretions |
| Nebulized hypertonic saline* |
| Systemic or inhaled corticosteroids |
*—When the hospital stay is shorter than three days.
Pulse oximetry alone is an unreliable measure of hypoxemia and respiratory distress, and continuous use is optional in infants and children with bronchiolitis.23 Continuous pulse oximetry lends to negative sleep effects on patients' families and alarm fatigue for families and staff. Its use is implicated in increased hospitalization rates without significant change in mortality.24
Maintaining the nutrition and hydration status of infants with bronchiolitis is an important factor in management. Infants with respiratory rates greater than 60 breaths per minute may have poor feeding secondary to difficulty breathing or copious nasal secretions.4 Oral rehydration in these patients may increase the risk of aspiration.25 For infants who cannot maintain oral hydration, intravenous or nasogastric fluids should be administered.4 In patients without impending respiratory failure, poor ventilation, or oxygenation, nasogastric rehydration was tolerated as well as intravenous fluid administration, with no difference in supplemental oxygen administration or length of hospitalization.26
Bronchodilators should not be administered to infants and children with bronchiolitis. Despite short-term improvement in clinical symptom scores, they have no effect on the need for hospitalization, oxygen saturation, length of hospitalization, or disease resolution.24,27 A 2014 Cochrane review of 30 randomized controlled trials found that infants with bronchiolitis who received bronchodilators did not have significant improvements in oxygen saturation, rate of hospitalization, or duration of hospitalization.24 In the outpatient setting, a short-term improvement was seen in clinical symptom scores using different scoring systems, some of which were partially validated. This analysis also included studies of children with recurrent wheezing, such as those with asthma, who are known to respond to bronchodilators.24 Ultimately, the adverse effects of bronchodilators, including high cost, tachycardia, and tremors, likely outweigh the possible short-term benefits.24
Epinephrine should not be administered to children with bronchiolitis in the inpatient setting.28,29 Outpatient epinephrine use is controversial. A 2011 Cochrane review of first-time wheezing among infants showed a statistically significant reduction in admission rates in patients who received nebulized epinephrine compared with placebo. However, this reduction was found on only the initial day of presentation, and there was no statistically significant reduction in the overall admission rate, likely because of the relatively short-acting effects of epinephrine.30
Nebulized hypertonic saline (3% or 7%) also should not be administered to infants with bronchiolitis in the emergency department or when the hospital stay is less than three days.4,31,32 Although it decreases the length of stay in patients hospitalized for more than three days, this benefit may not be applicable to patient populations in the United States, where the average length of stay is less than three days.31 Although the mechanism of action of hypertonic saline supports its benefit in chronic lung disease, it should not be administered to infants with bronchiolitis in the emergency department; however, it may be beneficial if administered in the inpatient setting.4
Systemic or inhaled corticosteroids should not be used to treat bronchiolitis in any setting.4,28,33 One study showed a possible reduction in hospitalization rates with simultaneous administration of high-dose dexamethasone and epinephrine for seven days in the outpatient setting, but this evidence is inconsistent.28 Furthermore, data for prolonged (three months) use of high-dose inhaled glucocorticoids started in the inpatient setting did not improve long-term outcomes in lung function, diagnosis of asthma, or atopic disease, further fueling the lack of evidence for corticosteroid use.34
Antibiotics should not be administered to infants and children with bronchiolitis and should be reserved for those with concomitant bacterial infection.4,35,36 Overall, there is a very low rate of bacteremia in patients diagnosed with bronchiolitis.35 Antibiotic therapy during acute bronchiolitis or in the postacute phase (after 14 days) does not reduce length of hospitalization or improve clinical outcomes.37 Antibiotic therapy may be administered in some subpopulations, including children with bronchiolitis who require intubation and mechanical ventilation for respiratory failure, because their risk of bacterial pneumonia is higher.38
Other treatments with no clear clinical benefit include chest physiotherapy and excessive nasal suction of secretions.39,40 Passive slow expiration technique may provide some short-term relief, but neither vibration nor percussion is beneficial. Although nasal suctioning provides relief of symptoms, excessive or deep nasal suctioning is associated with longer hospitalization. On the other hand, failure to perform routine external suctioning every four hours is also associated with increased length of stay.40
Prevention
Educating parents on reducing the risk of infection is one of the most important things a physician can do to help prevent RSV infection, especially early in life. RSV is highly contagious and is transmitted through direct contact with respiratory droplets. Secretions can remain infectious for more than six hours on hard surfaces such as tabletops, cribs, and toys.2,4 Strict hand hygiene must be adhered to, including washing hands before and after contact with a patient infected with RSV, after contact with surfaces near the patient, and after removal of gloves. Alcohol-based hand solutions are recommended for health care professionals; soap and water should be used if these are not available.4,41 In addition to frequent hand washing by caregivers, avoidance of large crowds and day care settings can also decrease the risk of infection.42,43 The morbidity of respiratory infections can be lowered by encouraging exclusive breastfeeding for at least six months and preventing exposure to secondhand smoke.4,44,45
Infants at risk of severe lower respiratory tract infection should receive immunoprophylaxis with palivizumab (Synagis). Five doses provide six months of coverage, which is sufficient protection for the RSV season. The initial recommendations for palivizumab in 1998 and again in 2003 were based on the results of two randomized, double-blind, placebo-controlled trials that reported a reduction in RSV hospitalization among high-risk infants who received palivizumab.42 A prospective, population-based surveillance study in 2005 found that the rate of RSV hospitalization was increased only among infants born before 30 weeks' gestation.8 In 2014, palivizumab prophylaxis guidelines were updated, with the goal of restricting its use to infants and children at greatest risk (Table 3).42
Table 3. Recommendations for the Use of Palivizumab Prophylaxis for Respiratory Syncytial Virus Infection

| Palivizumab (Synagis) is not recommended for otherwise healthy infants born at or after 29 weeks, 0 days' gestation. |
| Palivizumab is recommended in the first year of life for infants born before 29 weeks, 0 days' gestation. |
| Palivizumab is recommended in the first year of life for infants with hemodynamically significant heart disease. |
| Palivizumab is recommended in the first year of life for preterm infants with chronic lung disease of prematurity (born before 32 weeks, 0 days' gestation with an oxygen requirement of more than 21% for at least 28 days after birth). Palivizumab is recommended in the second year of life for children with chronic lung disease who require continued medical intervention (e.g., supplemental oxygen, chronic corticosteroid therapy, diuretics). |
| Consider prophylaxis for infants younger than one year who have pulmonary abnormalities or neuromuscular disease that impairs the ability to clear upper airway secretions, or for children younger than two years who are profoundly immunocompromised. |
Information from reference 42.
This article updates previous articles on this topic by Dawson-Caswell and Muncie,46 and by Steiner.47
Data Sources: A PubMed search was completed using the key terms respiratory syncytial virus, RSV, and bronchiolitis. The searches included meta-analyses, randomized controlled trials, clinical trials, and reviews. We also searched the Cochrane database, Essential Evidence Plus, and the National Guideline Clearinghouse. In addition, references in these resources were searched. Search dates: June 2015, April 2016, and November 2016.
The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Department of the Navy, the Department of Defense, or the U.S. government.
The authors are military service members. This work was prepared as part of their official duties. Title 17 U.S.C. 105 provides that “Copyright protection under this title is not available for any work of the United States Government.” Title 17 U.S.C. 101 defines United States Government work as a work prepared by a military service member or employee of the United States Government as part of that person's official duties.

