Cardiovascular disease (CVD) has been the primary cause of death in women for almost a century, and more women than men have died of CVD every year since 1984. Although CVD incidence can be reduced by adherence to a heart-healthy lifestyle and detection and treatment of major risk factors, preventive recommendations have not been consistently or optimally applied to women. The American Heart Association guidelines for CVD prevention in women provide physicians with a clear plan for assessment and treatment of CVD risk and personalization of treatment recommendations. The emphasis of preventive efforts has shifted away from treatment of individual CVD risk factors in isolation toward assessment of a woman’s overall or “global” CVD risk. In addition to accounting for the presence or absence of preexisting coronary heart disease or its equivalents (e.g., diabetes, chronic kidney disease), cardiovascular risk can be further calculated with the Framingham risk score, which is based on age, sex, smoking history, and lipid and blood pressure levels. Intervention intensity and treatment goals are tailored to overall risk, with those at highest risk receiving the most intense risk-lowering interventions. Women at high risk for CVD and without contraindications should receive aspirin, beta blockers, and an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker in addition to pharmacologic therapy for hyperlipidemia, hypertension, and diabetes. Women who already are at optimal or low risk for CVD should be encouraged to maintain or further improve their healthy lifestyle practices. Optimal application of these preventive practices significantly reduces the burden of death and disability caused by heart attack and stroke in women.
Cardiovascular disease (CVD) remains the most common cause of death in women and men in the developed world, despite the multiple epidemiologic and interventional studies that demonstrate significant declines in CVD incidence and prevalence with adherence to a healthy lifestyle and identification and treatment of risk factors.1 Women account for more than one half of the almost 1 million deaths caused by CVD in the United States annually2; yet historically, CVD risk factors in women have been insufficiently recognized, diagnosed, and treated.3–9
The basis for this lack of recognition and less aggressive management of cardiovascular risk in women has largely been a result of the misperception by women and physicians that women are at an inherently low risk for developing heart disease.10,11 A 2003 American Heart Association (AHA) survey found that only 13 percent of U.S. women (7 percent in 1997) believe heart disease is their major health threat.10 Black and Hispanic women are less likely than white women to be aware that heart disease is the primary cause of death in women. Only about one third of women recall discussing heart-disease risk with their physician10; and women receive fewer preventive recommendations, such as lipid-lowering therapy, aspirin, and lifestyle advice, than do men with similar Framingham risk scores.11,12
In 2004, the AHA published evidence-based guidelines for CVD prevention in women.1 Several factors led to the development of these guidelines. First, much of the data used to develop previous prevention guidelines came from trials that enrolled few or no women, making it difficult to perform a meaningful analysis of results by sex. In practice, the response to this evidence gap has been to withhold preventive therapies from women because of the lack of sex-specific proof of benefit, or to provide “gender-blind” preventive care using data from studies involving men to guide treatment in women. Neither approach is evidence based and both have the potential for harm. In addition, new guidelines were necessitated by the publication of results from several large CVD prevention trials, most notably the Women’s Health Initiative trial, which confirmed the lack of cardiovascular protection afforded by estrogen use in women after menopause.13–15 Finally, acceptance of the concept of “global risk,” codified by the National Cholesterol Eduction Program – Adult Treatment Program III (NCEP– ATP III) report as a mechanism to assign CVD risk levels and guide the aggressiveness of preventive interventions using the Framingham risk score,16 provided a validated framework for the recommendations.
Methods of the AHA Guidelines
Systematic review topics of the AHA guidelines included dietary modification; physical activity; tobacco cessation; management of hyperlipidemia, blood pressure, weight, and diabetes; cardiac rehabilitation; angiotensin-converting enzyme (ACE) inhibitor/angiotensin receptor blocker (ARB), antiplatelet, and beta-blocker therapy; warfarin (Coumadin) for atrial fibrillation; aspirin for primary prevention; treatment for depression; hormone therapy; and antioxidant, omega-3 fatty acid, and folic acid supplementation.1 Recent guidelines for treating hypertension,17,18 diabetes,19 hyperlipidemia,16 obesity,20 tobacco dependence,21 and atrial fibrillation22 were incorporated where applicable.
Each major topic was reviewed for content, strength of recommendation, level of evidence, and generalizability to women (where data in women were insufficient). The classification of efficacy and level of evidence rating systems (Table 11) were based on methods used in AHA and American College of Cardiology (ACC) clinical practice guidelines.23 Class I and IIa interventions are generally recommended and have adequate levels of evidence to support their implementation; class IIb interventions typically have potential benefit but are supported by weaker or conflicting data. The final recommendations were divided into five main categories: lifestyle interventions, major risk factor interventions, preventive drug interventions, atrial fibrillation and stroke prevention, and contraindicated (potentially harmful) interventions.
TABLE 1 AHA/ACC Recommendation Classifications and Levels of Evidence
| Strength of recommendation | |
|---|---|
| Classification | |
| Class I | Intervention is useful and effective. |
| Class IIa | Weight of evidence/opinion is in favor of usefulness/efficacy. |
| Class IIb | Usefulness/efficacy is less well established by evidence/opinion. |
| Class III | Intervention is not useful/effective and may be harmful. |
| Level of evidence | |
| A | Sufficient evidence from multiple randomized trials |
| B | Limited evidence from single randomized trial or other nonrandomized studies |
| C | Based on expert opinion, case studies, or standard of care |
| Generalizability index | |
| 1 | Very likely that results generalize to women |
| 2 | Somewhat likely that results generalize to women |
| 3 | Unlikely that results generalize to women |
| 0 | Unable to project whether results generalize to women |
AHA = American Heart Association; ACC = American College of Cardiology.
Reprinted with permission from Mosca L, Appel LJ, Benjamin EJ, Berra K, Chandra-Strobos N, Fabunmi RP, et al., for the American Heart Association. Evidence-based guidelines for cardiovascular disease prevention in women. Circulation 2004;109:675.
Dissemination and use of the guidelines was recognized as critical. Most women who could benefit from CVD preventive measures are not cared for by cardiovascular subspecialists until they develop symptoms or signs of CVD. Previous guidelines were published primarily in cardiology journals, which limited knowledge and adoption of preventive practices by family physicians, gynecologists, and internists, who care for most women at risk.
Cardiovascular Risk Assessment
The first step in applying the guidelines to individual women is the assessment of overall CVD risk. The Framingham 10-year coronary heart disease (CHD) risk score is central to making appropriate CHD preventive recommendations.16 Risk level can be calculated using age, sex, total and high-density lipoprotein (HDL) cholesterol levels, smoking history, and blood pressure (Figure 1).1 An online calculator is available athttp://hin.nhlbi.nih.gov/atpiii/calculator.asp?usertype=prof. Persons at high risk have a greater than 20 percent 10-year CHD risk, and include women with established CVD as well as those with CHD equivalents such as diabetes and chronic renal disease (Table 2).1 Persons at intermediate risk have a 10 to 20 percent 10-year CHD risk, and persons at lower risk have a less than 10 percent 10-year CHD risk. “Optimal risk” is defined as optimal levels of all risk factors and adherence to a heart-healthy lifestyle. The guidelines or recommendations for each risk category based on a person’s risk score determine the intensity of intervention.
Figure 1. Framingham Point Score Calculator: Estimate of 10-Year CHD Risk for Women
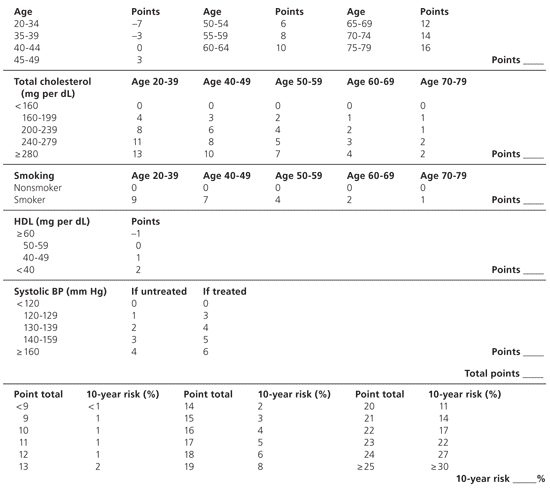
Framingham point score calculator for the 10-year risk of CHD in women. Points from each category are added to calculate risk. (CHD = coronary heart disease; HDL = high-density lipoprotein cholesterol; BP = blood pressure.)
note: An online risk calculator is available athttp://hin.nhlbi.nih.gov/atpiii/calculator.asp?usertype=prof.
Reprinted with permission from Mosca L, Appel LJ, Benjamin EJ, Berra K, Chandra-Strobos N, Fabunmi RP, et al., for the American Heart Association. Evidence-based guidelines for cardiovascular disease prevention in women. Circulation 2004;109:681.
TABLE 2 Spectrum of Cardiovascular Risk in Women
| Risk group | Framingham global risk (10-year absolute CHD risk) | Clinical examples |
|---|---|---|
| High risk | > 20% | Established CHD |
| Peripheral arterial disease | ||
| Abdominal aortic aneurysm | ||
| Diabetes mellitus | ||
| Chronic kidney disease | ||
| Intermediate risk | 10 to 20% | Subclinical CVD* (e.g., coronary calcification, carotid stenosis) |
| Metabolic syndrome (e.g., obesity, hypertension, hyperlipidemia) | ||
| Multiple risk factors | ||
| Markedly elevated levels of a single risk factor | ||
| First-degree relative with early-onset (i.e., younger than 55 years in men and younger than 65 years in women) atherosclerotic disease | ||
| Lower risk | < 10% | Modest or mild elevation of one or more risk factors; mayinclude women with metabolic syndrome or no risk factors |
| Optimal risk | < 10% | Optimal levels of risk factors and heart-healthy lifestyle |
*—Some patients with subclinical CVD have a greater than 20 percent 10-year CHD risk and should be elevated to the high-risk category.
CHD = coronary heart disease; CVD = cardiovascular disease.
Adapted with permission from Mosca L, Appel LJ, Benjamin EJ, Berra K, Chandra-Strobos N, Fabunmi RP, et al., for the American Heart Association. Evidence-based guidelines for cardiovascular disease prevention in women. Circulation 2004;109:673.
Final AHA Recommendations
The AHA CVD prevention recommendations for women are outlined in Tables 3 through 6.1 Interventions categorized as ineffective and potentially harmful, including prophylactic aspirin use in women at low risk, anti-oxidants,24 and hormone therapy, are listed in Table 7.1 A framework for applying the recommendations based on individual risk is given in Table 8,1 which lists the recommended interventions for each level of CVD risk.
TABLE 3 Clinical Recommendations for Lifestyle Interventions for the Prevention of CVD in Women
| Lifestyle intervention | AHA/ACC class, level | GI | References |
|---|---|---|---|
| Cigarette smoking | |||
| Consistently encourage women not to smoke and to avoid environmental tobacco. | I, B | 1 | 21 |
| Physical activity | |||
| Consistently encourage women to accumulate a minimum of 30 minutes of moderate-intensity physical activity (e.g., brisk walking) on most, and preferably all, days of the week. | I, B | 1 | |
| Cardiac rehabilitation | |||
| Women with a recent acute coronary syndrome or coronary intervention, or new-onset or chronic angina should participate in a comprehensive risk-reduction regimen, such as cardiac rehabilitation or a physician-guided home- or community-based program. | I, B | 2 | |
| Heart-healthy diet | |||
| Consistently encourage an overall healthy eating pattern including a variety of fruits, vegetables, grains, low-fat or nonfat dairy products, fish, legumes, and sources of protein low in saturated fat (e.g., poultry, lean meats, plant sources). Limit saturated fat intake to less than 10 percent of calories, limit cholesterol intake to less than 300 mg per day, and limit intake of trans-fatty acids. | I, B | 1 | 18 |
| Weight maintenance/reduction | |||
| Consistently encourage weight maintenance/reduction through an appropriate balance of physical activity, caloric intake, and formal behavioral programs when indicated to maintain or achieve a body mass index between 18.5 and 24.9 kg per m2 and a waist circumference less than 35 inches. | I, B | 1 | 20 |
| Psychosocial factors | |||
| Women with CVD should be evaluated for depression and referred or treated when indicated. | IIa, B | 2 | 29 |
| Omega-3 fatty acids | |||
| As an adjunct to diet, omega-3 fatty acid supplementation may be considered in women at high risk. | IIb, B | 2 | |
| Folic acid | |||
| As an adjunct to diet, folic acid supplementation may be considered in women at high risk (except after revascularization procedure) if a higher-than-normal level of homocysteine has been detected. | IIb, B | 2 | |
CVD = cardiovascular disease; AHA = American Heart Association; ACC = American College of Cardiology; GI = generalizability index.
note: For definitions of risk categories, see Table 2.
Adapted with permission from Mosca L, Appel LJ, Benjamin EJ, Berra K, Chandra-Strobos N, Fabunmi RP, et al., for the American Heart Association. Evidence-based guidelines for cardiovascular disease prevention in women. Circulation 2004;109:676.
TABLE 4 Clinical Recommendations for Major Risk Factor Interventions for Prevention of CVD in Women
| Risk factor intervention | AHA/ACC class, level | GI | References |
|---|---|---|---|
| Blood pressure—lifestyle | |||
| Encourage an optimal blood pressure of less than 120/80 mm Hg through lifestyle approaches. | I, B | 1 | 17 |
| Blood pressure—drugs | |||
| Pharmacotherapy is indicated when blood pressure is 140/90 mm Hg or greater, or lower in patients with blood pressure–related target-organ damage or diabetes. Thiazide diuretics should be part of the drug regimen for most patients unless contraindicated. | I, A | 1 | 17 |
| Lipid, lipoproteins | |||
| Optimal levels of lipids and lipoproteins in women are LDL cholesterol less than 100 mg per dL (2.59 mmol per L), HDL cholesterol greater than 50 mg per dL (1.29 mmol per L), triglycerides less than 150 mg per dL (1.69 mmol per L), and non–HDL cholesterol (total cholesterol minus HDL cholesterol) less than 130 mg per dL (3.36 mmol per L); these should be encouraged through lifestyle approaches. | I, B | 1 | 16 |
| Lipids—diet therapy | |||
| In women at high risk or with elevated LDL cholesterol, saturated fat intake should be reduced to less than 7 percent of calories, cholesterol should be reduced to less than 200 mg per day, and trans-fatty acid intake should be reduced. | I, B | 1 | 16 |
| Lipids—pharmacotherapy—high risk | |||
| Initiate LDL cholesterol–lowering therapy (preferably a statin) simultaneously with lifestyle therapy in women at high risk with LDL cholesterol 100 mg per dL or greater. | I, A | 1 | 16,26 |
| Initiate statin therapy in women at high risk with an LDL cholesterol less than 100 mg per dL unless contraindicated. | I, B | 1 | 16,26 |
| Initiate niacin or fibrate therapy when HDL cholesterol is low, or non–HDL cholesterol is elevated in women at high risk. | I, B | 1 | 16,26 |
| Lipids—pharmacotherapy—intermediate risk | |||
| Initiate LDL cholesterol–lowering therapy (preferably a statin) if LDL cholesterol level is 130 mg per dL or greater even with lifestyle therapy. | I, A | 1 | 16 |
| Initiate niacin or fibrate therapy when HDL cholesterol is low or non–HDL cholesterol elevated after LDL cholesterol goal is reached. | I, B | 1 | 16 |
| Lipids—pharmacotherapy—lower risk | |||
| Consider LDL cholesterol–lowering therapy in women at low risk with no more than one risk factor when LDL cholesterol level is 190 mg per dL (4.91 mmol per L) or greater, and in women with multiple risk factors when LDL cholesterol is 160 mg per dL (4.14 mmol per L) or greater; consider niacin or fibrate therapy when HDL cholesterol is low or non–HDL cholesterol elevated after LDL cholesterol goal is reached. | IIa, B | 1 | 16 |
| Diabetes | |||
| Lifestyle and pharmacotherapy should be used to achieve near-normal A1C levels (i.e., less than 7 percent) in women with diabetes. | I, B | 1 | 19 |
CVD = cardiovascular disease; AHA = American Heart Association; ACC = American College of Cardiology; GI = generalizability index; LDL = low-density lipoprotein; HDL = high-density lipoprotein.
note:For definitions of risk categories, seeTable 2.
Adapted with permission from Mosca L, Appel LJ, Benjamin EJ, Berra K, Chandra-Strobos N, Fabunmi RP, et al., for the American Heart Association. Evidence-based guidelines for cardiovascular disease prevention in women. Circulation 2004;109:676–7.
HEALTHY LIFESTYLE
Counseling on lifestyle interventions, including smoking cessation, physical activity, a heart-healthy diet, and weight maintenance, is important for all women, regardless of risk level, even if only to reinforce established healthy behaviors (Table 3).1 Women at higher risk may need additional consultation with a dietitian and prescription of a therapeutic diet or professional guidance in designing an exercise program. All women should be encouraged to perform at least 30 minutes of moderate-intensity physical activity (e.g., brisk walking) every day; to maintain or achieve a healthy weight; and to eat a diet low in saturated fat, cholesterol, trans-fatty acids, and sodium, and one that is rich in a variety of fruits, vegetables, whole grains, low-fat and nonfat dairy products, legumes, and fish.
RISK FACTOR MANAGEMENT
Risk factor interventions such as control of blood pressure and treatment of hyperlipidemia apply to all risk groups. The treatment targets and intensity of interventions are based on established guidelines as outlined in the Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure and the NCEP–ATP III report (Table 41).16,17 Ideal blood pressure is defined as less than 120/80 mm Hg. If weight loss, dietary modification, and exercise are insufficient to control blood pressure, pharmacologic therapy should be considered when blood pressure is 140/90 mm Hg or greater, or 130/80 mm Hg or greater in women with diabetes or chronic kidney disease.17
The ideal low-density lipoprotein (LDL) cholesterol level in women is less than 100 mg per dL (2.59 mmol per L), and the ideal HDL cholesterol level is greater than 50 mg per dL (1.29 mmol per L). The primary target for lipid-lowering therapy is LDL cholesterol. Women at high risk whose LDL cholesterol level is greater than 100 mg per dL should receive statin therapy to reach the goal level, and those at very high risk (e.g., multiple uncontrolled risk factors) should be considered for a target LDL cholesterol level of less than 70 mg per dL (1.81 mmol per L).25,26 Consideration also should be given to initiating pharmacologic therapy in women at high risk whose LDL cholesterol level is less than 100 mg per dL, because these women may benefit from further reduction. Pharmacologic therapy should be considered in women at intermediate risk whose LDL cholesterol levels are 130 mg per dL (3.36 mmol per L) or greater and in those at lower risk whose LDL cholesterol levels are 160 mg per dL (4.14 mmol per L) or greater. In women who have no more than one risk factor, a treatment threshold of LDL cholesterol 190 mg per dL (4.91 mmol per L) or greater should be used. Triglyceride levels should be less than 150 mg per dL (1.69 mmol per L), and combination therapy with niacin or a fibrate should be considered in women whose triglyceride levels remain elevated after LDL cholesterol level goals have been met.
Tight control of diabetes (i.e., normalized A1C level) is associated with a reduction in CVD events compared with less-stringent glucose control.27 Although the current A1C goal for most persons with diabetes is less than 7 percent, recent data suggest that normalization of A1C level (i.e., less than 6 percent) is associated with better outcomes and should be considered in patients who do not experience frequent hypoglycemic episodes.28
Women at high risk, especially those with overt CHD, should be screened and treated for depression (Table 3).1 Depression confers a significantly increased risk of adverse outcome after a diagnosis of heart disease and is a major barrier to adoption of healthy lifestyle behaviors. It is associated with a higher risk for first and recurrent myocardial infarction and is more prevalent in women than in men. Diagnosing and treating depression is critical, because treatment with selective serotonin reuptake inhibitors is safe and effective and may be associated with a reduced risk of adverse outcomes in women and men with heart disease.29,30
Except for generally higher levels of evidence to support CVD risk-lowering interventions in men, differences in treatment goals and recommendations for men and women are slight. For example, the desirable level of HDL cholesterol for men is 40 mg per dL (1.0 mmol per L) or greater. As in women, the goal levels of lipids, blood pressure, and diabetes control for men are dependent on estimated CVD risk, calculated using a separate Framingham risk table to reflect sex-based differences in risk intensity.
PHARMACOTHERAPY
In addition to pharmacologic therapy for hyperlipidemia, hypertension, and diabetes, all women at high risk for CVD should receive aspirin, beta blockers, and an ACE inhibitor or ARB unless contraindicated (Table 51).31,32 The use of omega-3 fatty acid and folic acid supplementation also should be considered in women at high risk, but there is insufficient evidence to support universal recommendations (Table 3).1
Aspirin has proven benefit and is recommended for all women at high risk without contraindications and for select women at intermediate risk (Table 5).1 Previously, there were few data on the use of aspirin for primary prevention of CVD in women. Earlier recommendations were based on extrapolation from large, men-only studies that demonstrated a reduction in risk for myocardial infarction but not stroke in men of middle age or older at intermediate or high risk. Data from the aspirin arm of the Women’s Health Initiative, released subsequent to the AHA guidelines, support the practice of not recommending aspirin to women at lower risk.33 In that study, almost 40,000 women were randomized to receive low-dose aspirin (100 mg every two days) or placebo. The overall risk of significant bleeding complications was increased by 40 percent in those taking aspirin, whereas ischemic stroke incidence was reduced by 24 percent. In contrast to the effects of aspirin for primary prevention of CVD in men, the risks of fatal and nonfatal myocardial infarction were not reduced in women except in those older than 65 years. A meta-analysis of five major aspirin primary prevention trials had similar findings.34 As a result, widespread use of aspirin is not recommended for women at low risk because of the excess risk of cerebral and gastrointestinal hemorrhage and the lack of definite benefit for prevention of CVD (Table 7).1
STROKE PREVENTION IN ATRIAL FIBRILLATION
Women with atrial fibrillation, paroxysmal or permanent, are at significantly increased risk for stroke; although systemic anticoagulation, particularly in older women, reduces this risk,22 women are less likely than men to receive it.35–37 As with other interventions, the decision to treat depends on risk level, and treatment guidelines are similar for women and men (Table 6).1 Women with atrial fibrillation who are at low risk for stroke (i.e., less than 1 percent per year) should be treated with aspirin at a dosage of 325 mg per day. Women at low risk include those younger than 60 years without heart disease or risk factors (e.g., hypertension, congestive heart failure, left ventricular dysfunction) and select older women with no risk factors for thromboembolism.22 Those at high or intermediate risk (i.e., 1 percent per year or greater) should receive warfarin anticoagulation with a goal International Normalized Ratio of 2 to 3. Persons with contraindications to warfarin anticoagulation should receive aspirin.
MANAGING HEALTH
Women must become more actively informed and work with physicians to manage their health, not just their illnesses. The mnemonic ALOHA can help physicians and women use the new guidelines effectively (Table 9).1,38 Several useful patient education pieces also have been developed.38,39 With wider dissemination and adoption of the AHA guidelines,1 CVD prevention practices in women may be enhanced, and CVD incidence and prevalence may decline. The gaps in sex-specific data identified in the systematic review process serve as a guide for researchers of future clinical trials to identify opportunities for reducing CVD risk in women.
TABLE 9 ALOHA: CVD Prevention Strategies
| Assess and stratify women into high, intermediate, lower, or optimal risk categories. |
| Lifestyle approaches (i.e., smoking cessation, regular exercise, weight management, and heart-healthy diet) to prevent CVD are AHA/ACC class I (SORT A*) recommendations for all women and a top priority in clinical practice. |
| Other CVD risk-reducing interventions should be prioritized on the basis of strength of recommendation and level of evidence, with the exception of lifestyle, which is a top priority for all women. |
| Highest priority is intervention in women at high risk. |
| Avoid interventions designated as AHA/ACC class III (e.g., hormone therapy, antioxidants; see Table 7). |
CVD = cardiovascular disease; AHA = American Heart Association; ACC = American College of Cardiology; SORT = strength of recommendation taxonomy.
*—A = consistent, good-quality patient-oriented evidence. For information about the SORT evidence rating system, see page 1263 orhttps://www.aafp.org/afpsort.xml.
note: For definitions of risk categories, see Table 2.
Adapted with permission from Mosca L, Appel LJ, Benjamin EJ, Berra K, Chandra-Strobos N, Fabunmi RP, et al., for the American Heart Association. Evidence-based guidelines for cardiovascular disease prevention in women. Circulation 2004;109:678, with additional information from reference 38.

