Chronic tendon injuries are common athletic and occupational injuries that account for many physician visits. Tendons have a complex biology that provides a unique combination of strength, flexibility, and elasticity but also predisposes them to injury. The term tendinopathy is preferred to tendinitis because of the presence of a disordered and degenerative healing process—not inflammation—in the pathologic tendon. Insidious onset of pain and dysfunction is a common presentation for most tendinopathies, and patients typically report that a change in activity affected the use of the tendon. Diagnosis is typically based on history and physical examination findings, but radiography is an acceptable initial imaging modality. Ultrasonography and magnetic resonance imaging may be useful when the diagnosis is unclear. The mainstays of treatment are activity modification, relative rest, pain control, and protection. Early initiation of rehabilitative exercises that emphasize eccentric loading is also beneficial. Despite a lack of high-quality evidence, cryotherapy has a role in controlling pain. Nonsteroidal anti-inflammatory drugs and corticosteroids have a role in treatment despite the lack of histologic evidence of inflammation. Short-term use of these drugs reduces pain and increases range of motion, which can assist patients in completing rehabilitative exercises. Care should be taken when injecting corticosteroids into and near major load-bearing tendons because of the risk of rupture. Topical nitroglycerin, extracorporeal shock wave therapy, and platelet-rich plasma injections have varying levels of evidence in certain tendinopathies and are additional nonsurgical treatment options.
Tendon injuries account for at least 7% of physician visits in the United States.1 More than 30% of sports-related injuries involve tendons, but chronic tendinopathy can also occur via occupational or lifestyle activities and can impair work and recreational performance.1–3
SORT: KEY RECOMMENDATIONS FOR PRACTICE
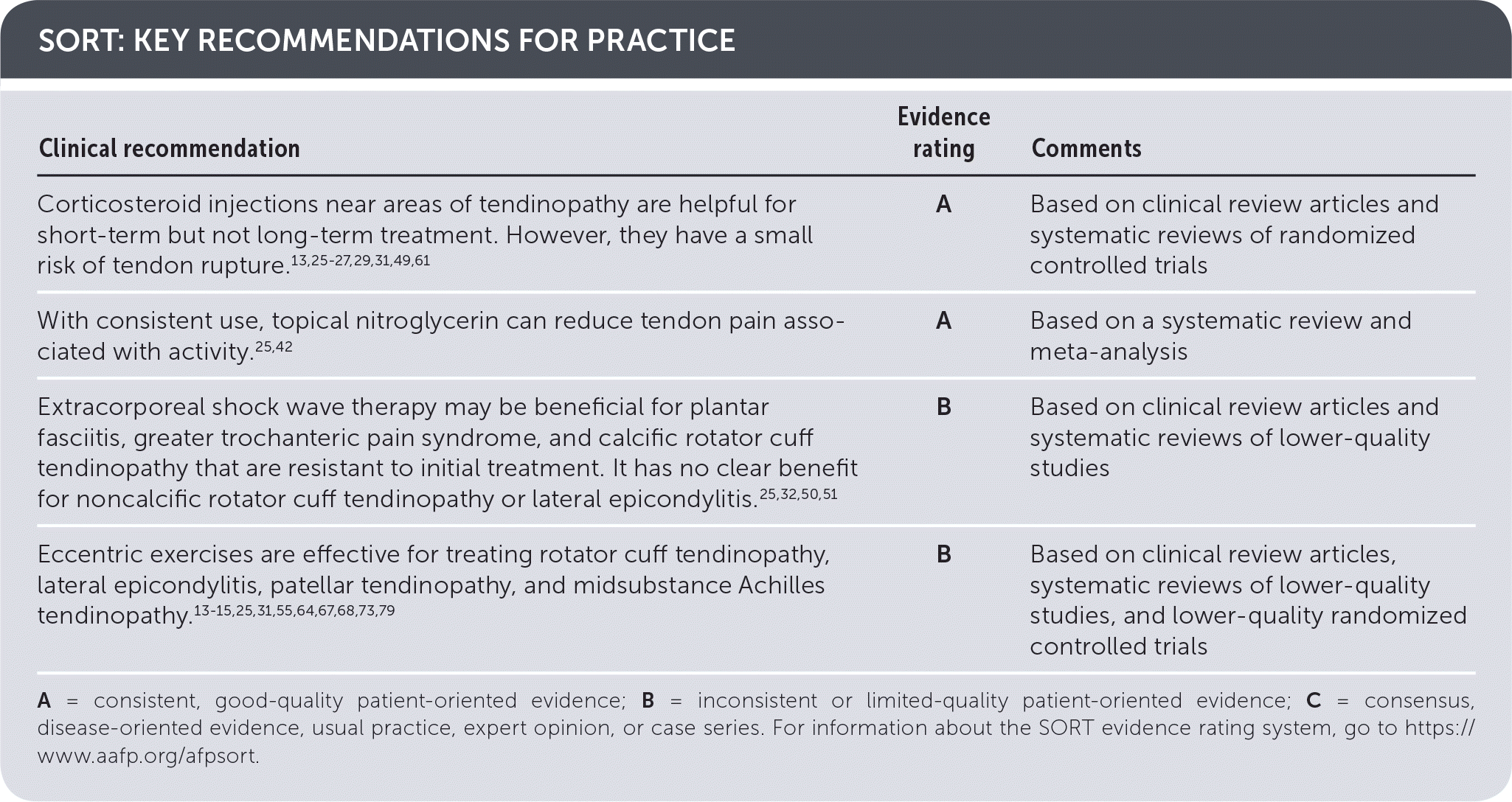
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
The terms tendinitis, tendinopathy, and tendinosis are commonly used when discussing tendon injuries, but these terms are often misconstrued (Table 1).4 Tendinopathy is the most accurate clinical term to describe tendon pain and dysfunction of unclear duration.5 Although inflammation may be present, tendinopathy is primarily a degenerative process in which a failed healing response leads to pain and dysfunction.6,7
TABLE 1. Definitions of Tendon Pathology

| Pathology | Definition |
|---|---|
| Tendinitis | Injury to the tendon that causes an inflammatory response and presence of inflammatory cells |
| Tendinopathy | Injury to the tendon that involves degeneration and failed healing; clinical syndrome of tendon pain and thickening; diagnosed primarily via history and physical examination findings |
| Tendinosis | Histopathologic term used when a tissue sample shows absence or minimal presence of inflammatory cells; synonymous with angiofibroblastic dysplasia |
Information from reference 4.
Pathophysiology
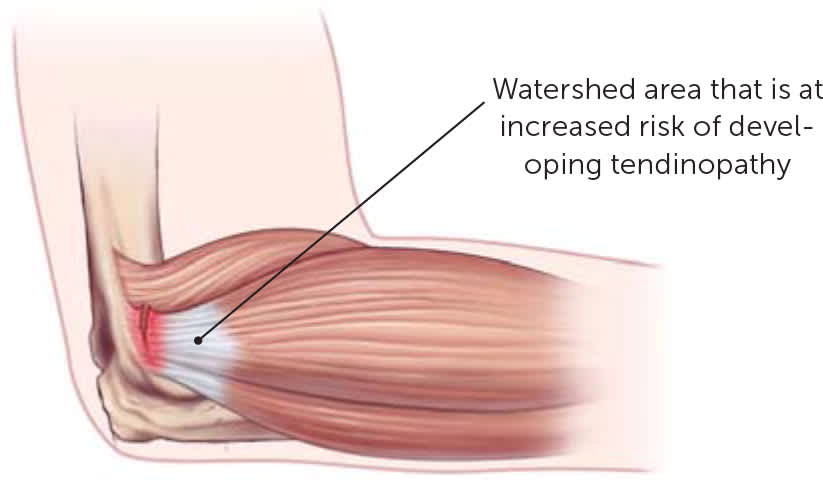
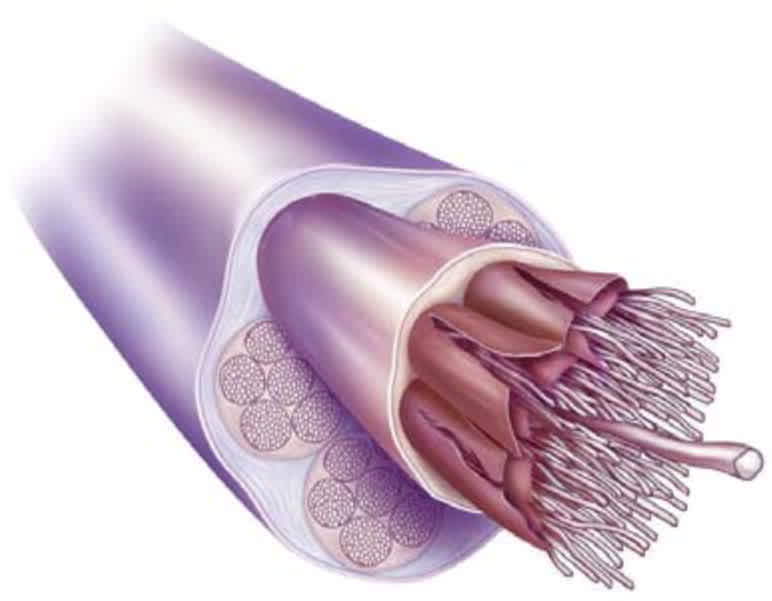
Tendons are biologically complex structures composed of multiple layers of tightly packed collagen fibers that are produced by tenocytes and tenoblasts5,8 (Figure 11 ). Tenocytes are mechano-receptive cells that are able to change their gene expression in response to demand.9 Tendons' unique combination of strength, flexibility, and elasticity allows them to carry loads and maintain tensile strength for long periods of time. Because they have a lower rate of oxygen consumption than skeletal muscle, they are also slow to heal. Tendons receive their blood supply from a variety of sources, and pathology usually occurs in the hypovascular or watershed areas between two sources of blood supply 1,2,10 (Figure 211 ). Appropriate loads on a tendon are anabolic, but overloading a tendon can be catabolic and catastrophic. Disrupting the homeostasis between tendon stress and repair damages the tendon8,12 (Figure 31 ).
FIGURE 1.
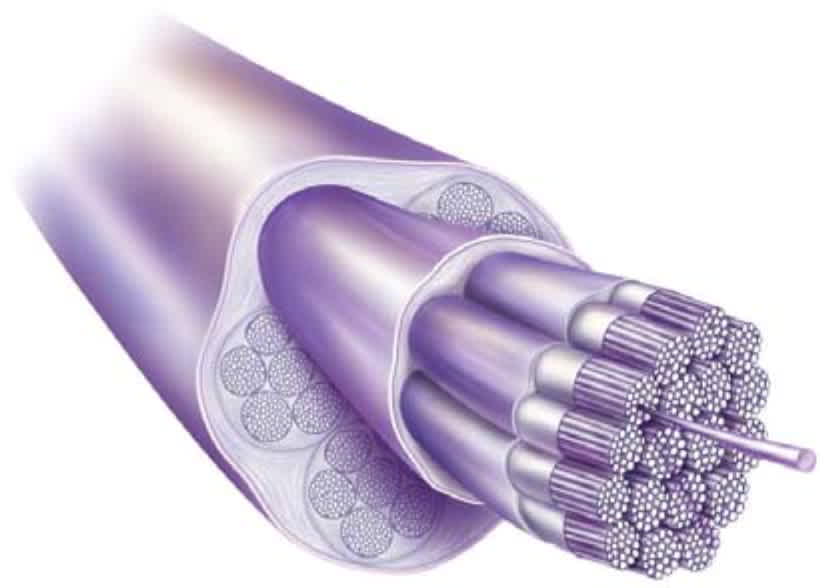
Histology of a normal tendon.
Illustration by Renee L. Cannon
Reprinted with permission from Wilson JJ, Best TM. Common overuse tendon problems: a review and recommendations for treatment. Am Fam Physician. 2005;72(5):813.
FIGURE 2.
Lateral epicondyle and the origin of the common extensor tendon.
Illustration by Myriam Kirkman-Oh
Reprinted with permission from Kane SF, Lynch JH, Taylor JC. Evaluation of elbow pain in adults. Am Fam Physician. 2014;89(8):653.
FIGURE 3.
Histology of a damaged tendon. Note the collagen disorientation and fiber separation.
Illustration by Renee L. Cannon
Reprinted with permission from Wilson JJ, Best TM. Common overuse tendon problems: a review and recommendations for treatment. Am Fam Physician. 2005;72(5):813.
The etiology of pain associated with chronic tendinopathy is unclear but has been attributed to neovascularization, neurochemicals, and mechanical breakdown. Patients typically present with localized pain of insidious onset that is often associated with beginning a new activity or increasing the intensity of regular activities.2,8,13 In addition to activity changes, there are other modifiable and nonmodifiable risk factors that should be evaluated (Table 2).14–16 Ideally, tendinopathy can be prevented by gradually modifying activity or movement patterns. If prevention is not possible, early identification and treatment of an improper tendon response to loading activities will minimize the chance of progression to a chronic injury.4
TABLE 2. Risk Factors for Tendinopathy
Imaging
The diagnosis of chronic tendinopathy is based on history, pain on palpation, and findings from provocative physical examination maneuvers. When alternative diagnoses are being considered, plain radiography can be performed initially for atraumatic joint pain. Magnetic resonance imaging or ultrasonography can be performed for follow-up imaging if pain persists and initial radiography findings were unremarkable or inconclusive.17–19
No single imaging technique can definitively diagnose tendinopathy. The specificity of ultrasonography and magnetic resonance imaging for the diagnosis of tendinopathy ranges from 63% to 83% and 68% to 70%, respectively. Both modalities can show the tendon thickening or disorganized fiber orientation that occurs with chronic tendinopathy, but tendons of asymptomatic patients can also exhibit these changes. Similarly, the enthesophytes and calcifications often seen on plain radiography of patients with tendon pain can be present in asymptomatic patients. Ultrasonography can show tendon hypoechogenicity or increased vascularity, which are associated with tendinopathy; however, the diagnostic power of ultrasonography is user dependent.20
Treatment
The mainstays of initial treatment for tendinopathy are activity modification, relative rest, pain control, rehabilitative exercise, and protection (Table 3).3,4,13–15,21–46 It is important for the patient to understand that it may take more than six months for complete symptom resolution.1,47 Early initiation of selective and specific rehabilitative exercises is beneficial because appropriate tendon loading is critical for healing, which is why relative rest—not total rest—is recommended.4,47 Cryotherapy with ice reduces pain in chronic tendinopathy, possibly by decreasing the neovascularization process, and is widely used as a simple and low-cost remedy with few adverse effects.28,29
TABLE 3. Summary of Treatments for Chronic Tendon Injuries
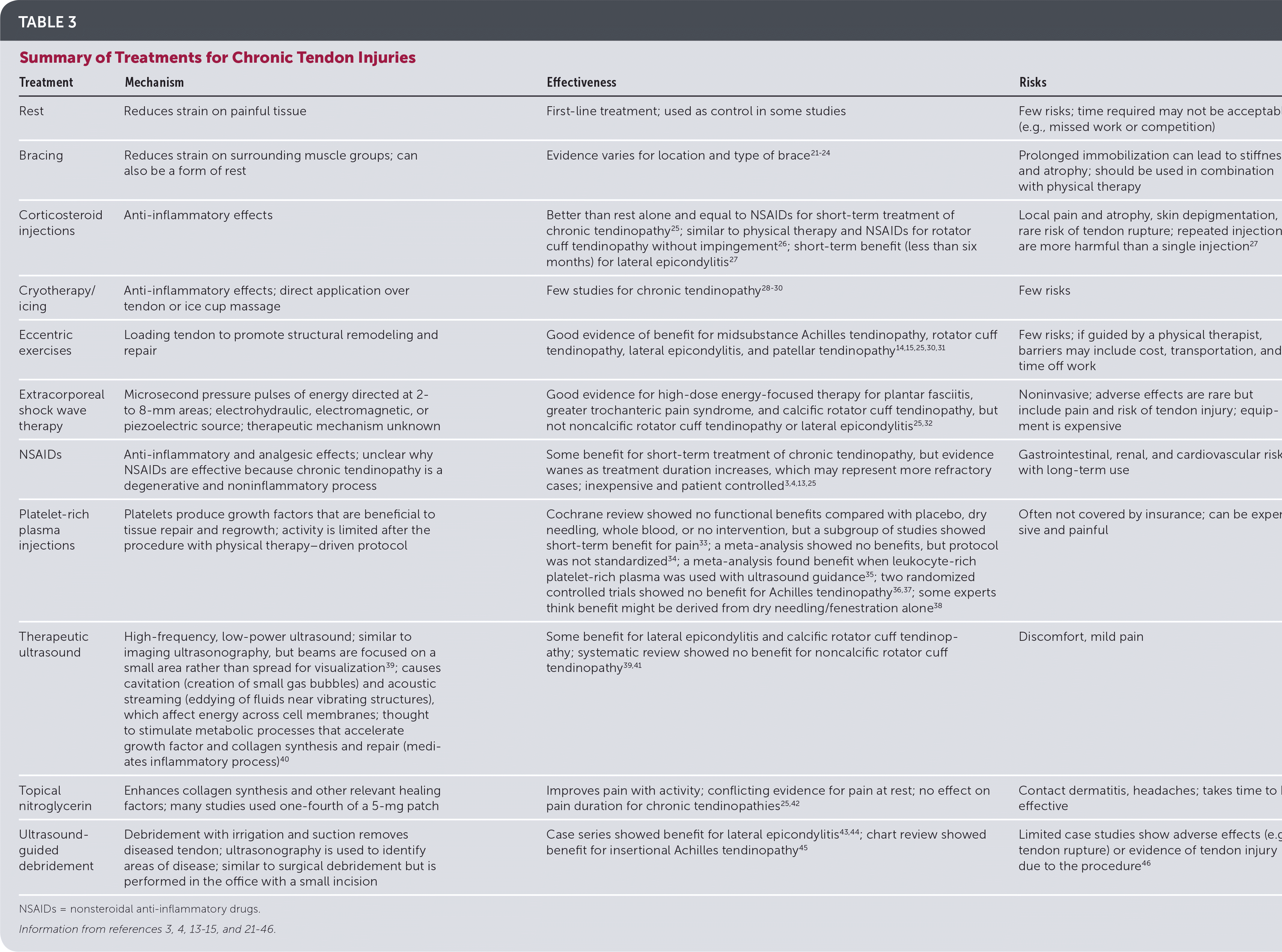
| Treatment | Mechanism | Effectiveness | Risks |
|---|---|---|---|
| Rest | Reduces strain on painful tissue | First-line treatment; used as control in some studies | Few risks; time required may not be acceptable (e.g., missed work or competition) |
| Bracing | Reduces strain on surrounding muscle groups; can also be a form of rest | Evidence varies for location and type of brace21–24 | Prolonged immobilization can lead to stiffness and atrophy; should be used in combination with physical therapy |
| Corticosteroid injections | Anti-inflammatory effects | Better than rest alone and equal to NSAIDs for short-term treatment of chronic tendinopathy25; similar to physical therapy and NSAIDs for rotator cuff tendinopathy without impingement26; short-term benefit (less than six months) for lateral epicondylitis27 | Local pain and atrophy, skin depigmentation, rare risk of tendon rupture; repeated injections are more harmful than a single injection27 |
| Cryotherapy/icing | Anti-inflammatory effects; direct application over tendon or ice cup massage | Few studies for chronic tendinopathy28–30 | Few risks |
| Eccentric exercises | Loading tendon to promote structural remodeling and repair | Good evidence of benefit for midsubstance Achilles tendinopathy, rotator cuff tendinopathy, lateral epicondylitis, and patellar tendinopathy14,15,25,30,31 | Few risks; if guided by a physical therapist, barriers may include cost, transportation, and time off work |
| Extracorporeal shock wave therapy | Microsecond pressure pulses of energy directed at 2- to 8-mm areas; electrohydraulic, electromagnetic, or piezoelectric source; therapeutic mechanism unknown | Good evidence for high-dose energy-focused therapy for plantar fasciitis, greater trochanteric pain syndrome, and calcific rotator cuff tendinopathy, but not noncalcific rotator cuff tendinopathy or lateral epicondylitis25,32 | Noninvasive; adverse effects are rare but include pain and risk of tendon injury; equipment is expensive |
| NSAIDs | Anti-inflammatory and analgesic effects; unclear why NSAIDs are effective because chronic tendinopathy is a degenerative and noninflammatory process | Some benefit for short-term treatment of chronic tendinopathy, but evidence wanes as treatment duration increases, which may represent more refractory cases; inexpensive and patient controlled3,4,13,25 | Gastrointestinal, renal, and cardiovascular risks with long-term use |
| Platelet-rich plasma injections | Platelets produce growth factors that are beneficial to tissue repair and regrowth; activity is limited after the procedure with physical therapy–driven protocol | Cochrane review showed no functional benefits compared with placebo, dry needling, whole blood, or no intervention, but a subgroup of studies showed short-term benefit for pain33; a meta-analysis showed no benefits, but protocol was not standardized34; a meta-analysis found benefit when leukocyte-rich platelet-rich plasma was used with ultrasound guidance35; two randomized controlled trials showed no benefit for Achilles tendinopathy36,37; some experts think benefit might be derived from dry needling/fenestration alone38 | Often not covered by insurance; can be expensive and painful |
| Therapeutic ultrasound | High-frequency, low-power ultrasound; similar to imaging ultrasonography, but beams are focused on a small area rather than spread for visualization39; causes cavitation (creation of small gas bubbles) and acoustic streaming (eddying of fluids near vibrating structures), which affect energy across cell membranes; thought to stimulate metabolic processes that accelerate growth factor and collagen synthesis and repair (mediates inflammatory process)40 | Some benefit for lateral epicondylitis and calcific rotator cuff tendinopathy; systematic review showed no benefit for noncalcific rotator cuff tendinopathy39,41 | Discomfort, mild pain |
| Topical nitroglycerin | Enhances collagen synthesis and other relevant healing factors; many studies used one-fourth of a 5-mg patch | Improves pain with activity; conflicting evidence for pain at rest; no effect on pain duration for chronic tendinopathies25,42 | Contact dermatitis, headaches; takes time to be effective |
| Ultrasound-guided debridement | Debridement with irrigation and suction removes diseased tendon; ultrasonography is used to identify areas of disease; similar to surgical debridement but is performed in the office with a small incision | Case series showed benefit for lateral epicondylitis43,44; chart review showed benefit for insertional Achilles tendinopathy45 | Limited case studies show adverse effects (e.g., tendon rupture) or evidence of tendon injury due to the procedure46 |
NSAIDs = nonsteroidal anti-inflammatory drugs.
REHABILITATIVE EXERCISE
Rehabilitative exercise is the cornerstone of tendinopathy treatment. It focuses first on returning to a normal pain-free range of motion, then on increasing strength. Therapy consists of eccentric exercises (Figure 42 and Figure 52 ), which consist of loading a muscle and tendon as they are being lengthened, and/or concentric exercises, which consist of loading while the muscle and tendon shorten. Although eccentric exercises are preferred in the treatment of some tendinopathies, there is no clear consensus on a specific rehabilitation program, and most programs consist of a combination of eccentric and concentric exercises.48
FIGURE 4.
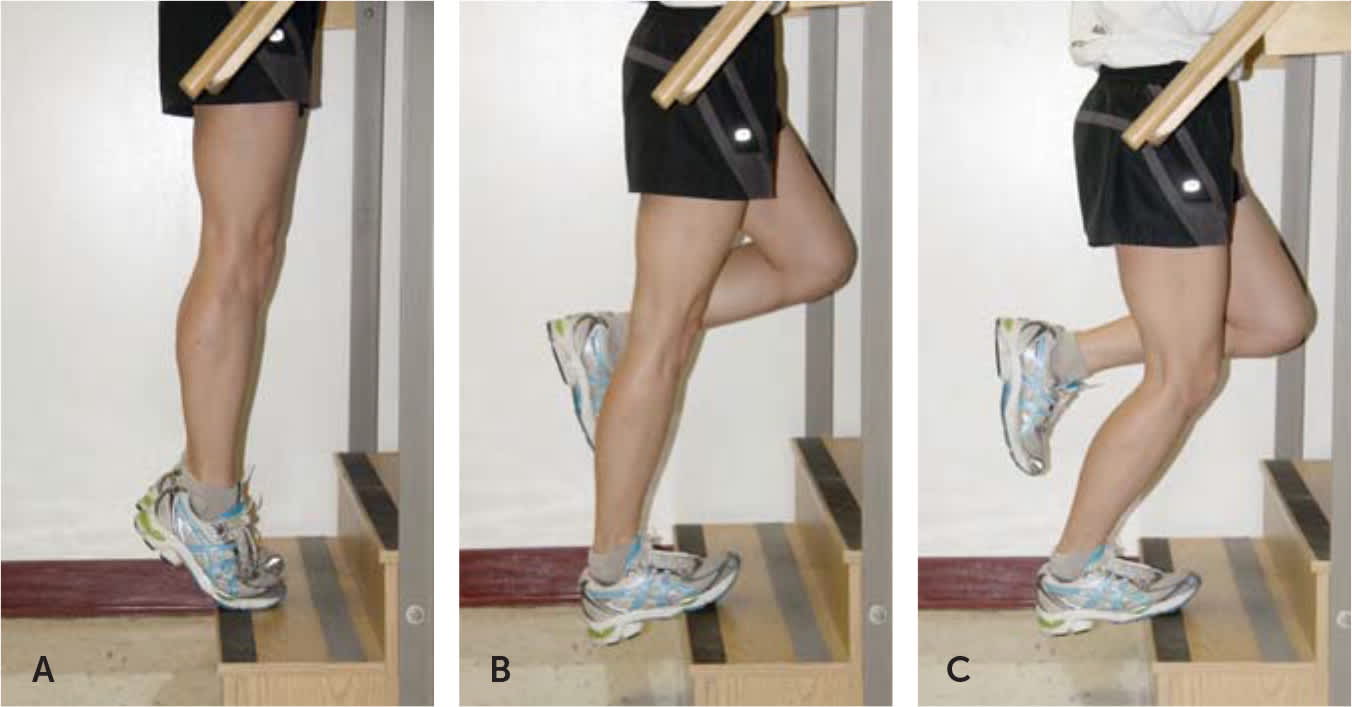
Eccentric exercises for midsubstance Achilles tendinopathy. (A) Patients begin with a straight leg and the ankle in flexion. (B) The ankle of the injured leg is then lowered to full dorsiflexion and returned to its original position with the assistance of the uninjured leg. (C) The exercise is repeated with the knee bent to approximately 45 degrees.
Reprinted with permission from Childress MA, Beutler A. Management of chronic tendon injuries. Am Fam Physician. 2013;87(7):488.
FIGURE 5.
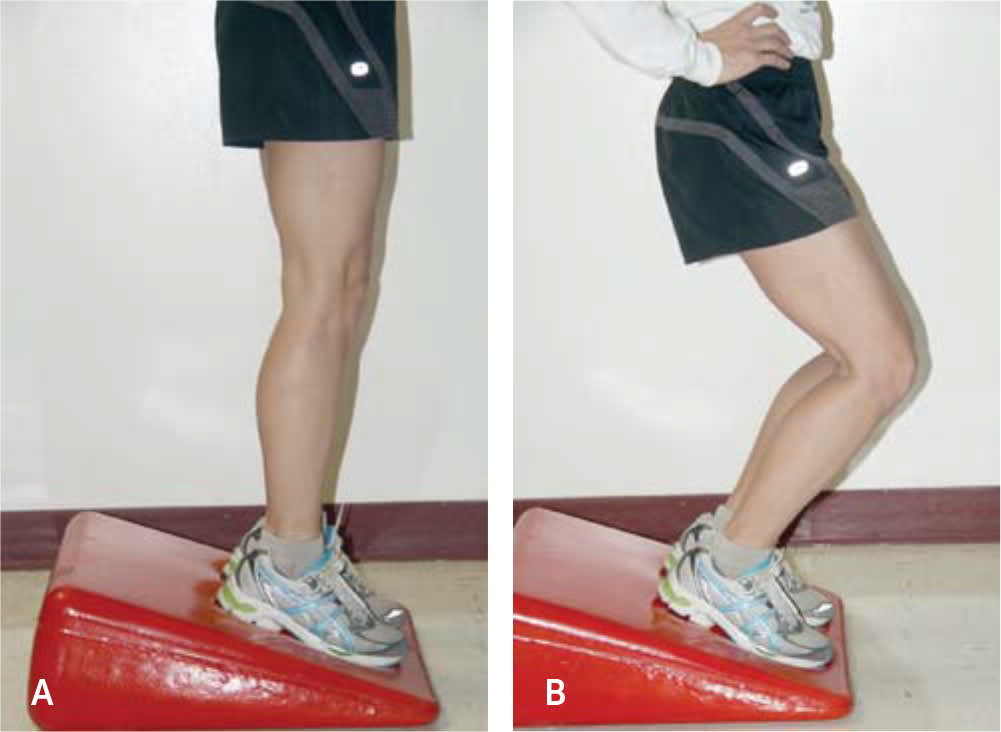
Eccentric exercises for the patellar tendon. (A) Patients are instructed to begin in an extended position and (B) to slowly bend the knees to approximately 45 degrees, then return to an extended position.
Reprinted with permission from Childress MA, Beutler A. Management of chronic tendon injuries. Am Fam Physician. 2013;87(7):488.
NSAIDS, CORTICOSTEROIDS, AND TOPICAL NITROGLYCERIN
Although tendinopathy is a degenerative noninflammatory process, corticosteroids and nonsteroidal anti-inflammatory drugs (NSAIDs) are commonly used in its treatment.1,2 NSAIDs can be administered orally or topically and have benefit with short-term use (seven to 14 days). Risks of long-term use include gastrointestinal bleeding, impaired renal function, and mildly increased risk of cardiovascular disease.3,4,13 A single peritendinous corticosteroid injection is as effective as NSAIDs for acute pain relief, but repeated injections may worsen chronic pain and result in tendon rupture.3,4,13 Many clinicians perform tendon fenestration with corticosteroid injections. This involves rapidly moving the needle throughout the area of injured tendon to create microtrauma, which increases bleeding and recruits local regenerative factors. Fenestration is effective as a solo technique.38 It differs from dry needling, in which a thin needle is placed directly into a thickened or painful area of tendon or muscle (found by palpation, not ultrasonography). Dry needling also is an effective treatment and can be performed with or without electrical stimulation.49 Daily application of topical nitroglycerin (usually administered as one-fourth of a 5-mg patch) can improve strength and decrease pain with activity. Although risks include headaches and localized skin reactions, topical nitroglycerin is a useful option for primary care physicians in cases of refractory tendinopathy.42
OTHER TREATMENTS
Extracorporeal shock wave therapy is another nonsurgical option that can be considered for the treatment of refractory tendinopathy. Studies have shown that it has similar effectiveness as traditional physiotherapy, eccentric exercises, corticosteroid injections, platelet-rich plasma injections, and surgery.50,51 Table 3 reviews other treatment options and the evidence to support their effectiveness.3,4,13–15,21–46 Some techniques that are not included in the table have limited evidence but are low risk (e.g., deep tissue massage, iontophoresis, phonophoresis), whereas others (e.g., stem cells or other orthobiologics) are more invasive and costly, or have unclear evidence of effectiveness.25,52
SURGERY
Depending on the anatomic location, 10% to 45% of patients with tendinopathy will not improve with conservative management.53 There is no firm timeline for when to refer them for surgery, but it is reasonable after three to six months of conservative treatment when there has been little or no improvement.53,54
Common Tendinopathies
UPPER EXTREMITIES
Rotator Cuff Tendinopathy. Shoulder pain is one of the top three causes of musculoskeletal symptoms, with an estimated incidence of 19 cases per 1,000 person years.55,56 Rotator cuff pathology is the most common cause. The rotator cuff comprises four muscles: the supraspinatus, infraspinatus, teres minor, and subscapularis. Its primary function is to maintain the position of the humeral head during glenohumeral movement. The supraspinatus is the most likely tendon to show signs of injury.57,58 The etiology of rotator cuff tears (partial or complete) is a complicated interaction of intrinsic, extrinsic, and environmental factors. A chronic degenerative injury can lead to significant pain, disability, or a complete tear.4,57
Patients with rotator cuff tendinopathy typically report weakness, stiffness, constant pain that is exacerbated with certain movements, and pain that makes it difficult to sleep on the affected side. The mainstay of treatment is rehabilitative exercise focusing on scapular stabilization and biomechanics to decrease subacromial impingement.59 Eccentric exercise has not been proven superior to conventional exercise in treating rotator cuff tendinopathy, and home-based programs are as effective as clinic-based programs.55,60 Subacromial corticosteroid injections are commonly used to provide short-term pain relief, which can be useful when initiating physical therapy.61 If the patient remains symptomatic after six months of therapy, other treatments such as extracorporeal shock wave therapy, platelet-rich plasma injections, or dextrose prolotherapy can be considered. If these treatments are ineffective, surgery may be a consideration.33,62
Biceps Tendinopathy. The long head of the biceps (LHB) is the major source of pain in the anterior aspect of the shoulder. The LHB tendon sheath is a continuum of the glenohumeral joint and is closely associated with rotator cuff pathology; it is estimated that chronic LHB tendinopathy is the primary injury in only 5% of patients with shoulder pain.63,64 Repetitive traction, friction, and shear forces in the narrow bicipital groove from constant glenohumeral rotation lead to the insidious and progressive pain that can occur with repetitive overhead movements. Reproduction of pain with direct palpation of the tendon in the bicipital groove is the most common physical examination finding. In some patients, palpation is painful even in nonpathologic tendons, so it is important to also palpate the unaffected, contralateral side. Several other tests have been developed to diagnose LHB tendinopathy, but none has a high predictive value, whether used alone or in specific combinations.64,65
Therapeutic injections may be helpful in establishing the diagnosis of LHB tendinopathy vs. rotator cuff pathology. Persistent pain of the LHB tendon after a subacromial injection or improvement in pain after an injection directly into the bicipital groove can confirm LHB tendinopathy. Because of the high correlation with rotator cuff pathology, most rehabilitative exercises for LHB tendinopathy also focus on addressing problems with the rotator cuff. Primary LHB tendon rehabilitation focuses on range of motion, muscle balance, and dynamic stability of the shoulder.63,65
Epicondylitis. Epicondylitis is a misnomer; there is no inflammation at the humeral epicondyles. These conditions are tendinopathies of the common extensor and flexor tendons of the forearm. Medial epicondylitis is not as common as lateral epicondylitis, but the treatments are generally the same.66 Lateral epicondylitis, or tendinopathy of the extensor carpi radialis brevis, affects 1% to 3% of Americans each year.11,31 Patients report pain with wrist extension and reduced grip strength, and they have reproducible pain with palpation approximately 1 cm distal to the lateral epicondyle. Although it is also called tennis elbow, lateral epicondylitis is more commonly associated with occupational overuse injuries than athletic activities.11
Physical therapy with an emphasis on eccentric exercises has short- and long-term benefit for epicondylitis compared with no treatment or corticosteroid injections.31,67 Bracing can treat pain and does not negatively affect strength.21 A comparison of cock-up wrist braces vs. counterforce straps showed that wrist braces may have an advantage because of complete immobilization, but the strap is more practical for daily use.22–24 If conservative treatment is ineffective, corticosteroid injections, deep friction massage, dry needling, and platelet-rich plasma injections are appropriate. Referral for surgery is appropriate if these treatments are ineffective.27,68,69
LOWER EXTREMITIES
Greater Trochanteric Pain Syndrome. Greater trochanteric pain syndrome (GTPS) is lateral hip pain with tenderness over the greater trochanter, and has been attributed to trochanteric bursitis.70 GTPS is an inaccurate diagnosis because the primary etiology of the pain is a degenerative tendinopathy of the gluteus medius or gluteus minimus tendons with some secondary bursal distention.71,72 GTPS is a common condition, affecting 1.8 to 5.6 per 1,000 adults each year with a 4: 1 female-to-male prevalence.73 Risk factors include being 40 to 60 years of age or obese, and having low back pain, knee or hip osteoarthritis, a sedentary lifestyle, or poor running biomechanics with increased hip adduction.71,74 Patients with GTPS report insidious onset of chronic intermittent lateral hip pain that worsens with activity, sitting with legs crossed, or lying on the affected side. GTPS is a clinical diagnosis that is confirmed with pain over the greater trochanter and other findings such as pain with resisted hip abduction, nonradicular pain extending down the lateral thigh, and pain with FABER testing (hip flexion, abduction, and external rotation).75
There is no universally accepted physical therapy program for GTPS. Therapy should focus on patient-specific deficits in combination with stretching of the piriformis and iliotibial band and strengthening of the gluteal and core muscle groups. In addition to therapy, corticosteroid injections provide short- and medium-term pain control, and extracorporeal shock wave therapy provides medium- to long-term pain control.49 GTPS is a self-limiting condition, and conservative treatment is successful in up to 90% of patients.70,73,74,76 However, relapse is common if the underlying cause is not addressed.
Patellar Tendinopathy. Patellar tendinopathy, or jumper's knee, is a common cause of anterior knee pain. The highest incidence is in jumping sports such as basketball, volleyball, and soccer. As with most tendinopathies, its etiology is multifactorial. Risk factors include high body mass index, leg length discrepancies, pes cavus, weak and inflexible quadriceps, and inflexible hamstrings.29 Patients report insidious onset of anterior knee pain that is exacerbated with knee flexion and prolonged activity. Localized tenderness at the inferior pole of the patella is a consistent physical examination finding. The decline squat test is the most specific maneuver (Figure 6)1; patients with patellar tendinopathy will be limited by pain in the number and depth of squats they can perform.
FIGURE 6.
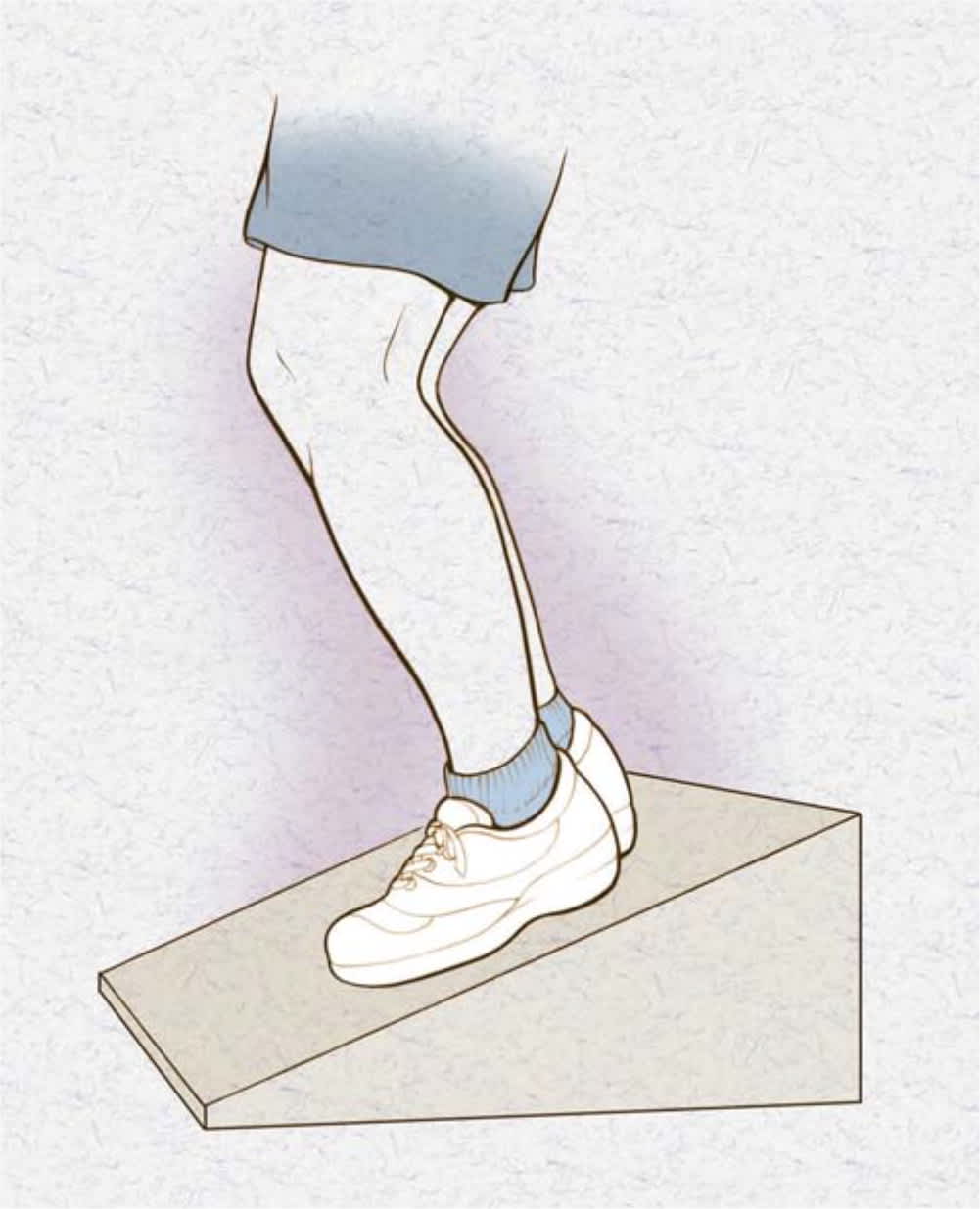
Decline squat test for patellar tendinopathy. This test places greater load on the patellar tendon than a squat performed on level ground.
Illustration by Marcia Hartsock
Reprinted with permission from Wilson JJ, Best TM. Common overuse tendon problems: a review and recommendations for treatment. Am Fam Physician. 2005;72(5):817.
Initial management of patellar tendinopathy includes relative rest and pain control with NSAIDs or cryotherapy. A patellar counterforce brace, which works by positively altering tendon biomechanics, helps decrease pain and increase function14,29,77,78 (Figure 777 ). Eccentric exercise is another proven treatment modality and should be part of any physical therapy program for patellar tendinopathy.14 Up to 45% of patients will not respond to eccentric exercise alone; in these cases, treatments such as corticosteroid injections, topical nitroglycerin, extracorporeal shock wave therapy, and other therapeutic injections are options before surgery.14,29
FIGURE 7.

Patellar tendon strap for patellar tendinopathy.
Reprinted with permission from Sprouse RA, McLaughlin AM, Harris GD. Braces and splints for common musculoskeletal conditions. Am Fam Physician. 2018;98(10):572.
Achilles Tendinopathy. Achilles tendinopathy is common, particularly in patients who engage in regular running-related activities. Studies have estimated an annual incidence of 7% to 9% in current runners and a lifetime prevalence of 52% in former runners.15 However, 30% of referred patients report not regularly participating in exercise, which suggests a multifactorial etiology.16,54 Achilles tendinopathy is a clinical diagnosis characterized by pain, swelling, and impaired tendon performance. The pain initially starts at the beginning of and at the termination of exercise; as the condition becomes more chronic, the pain persists throughout exercise and can impair activities of daily living. The pathology typically occurs in two locations: 55% to 65% at the midportion of the tendon (2 cm to 6 cm proximal to the insertion), and 20% to 25% at the insertion (Figure 8).2 If the location is uncertain based on examination findings, ultrasonography or magnetic resonance imaging can be performed.54,79
FIGURE 8.
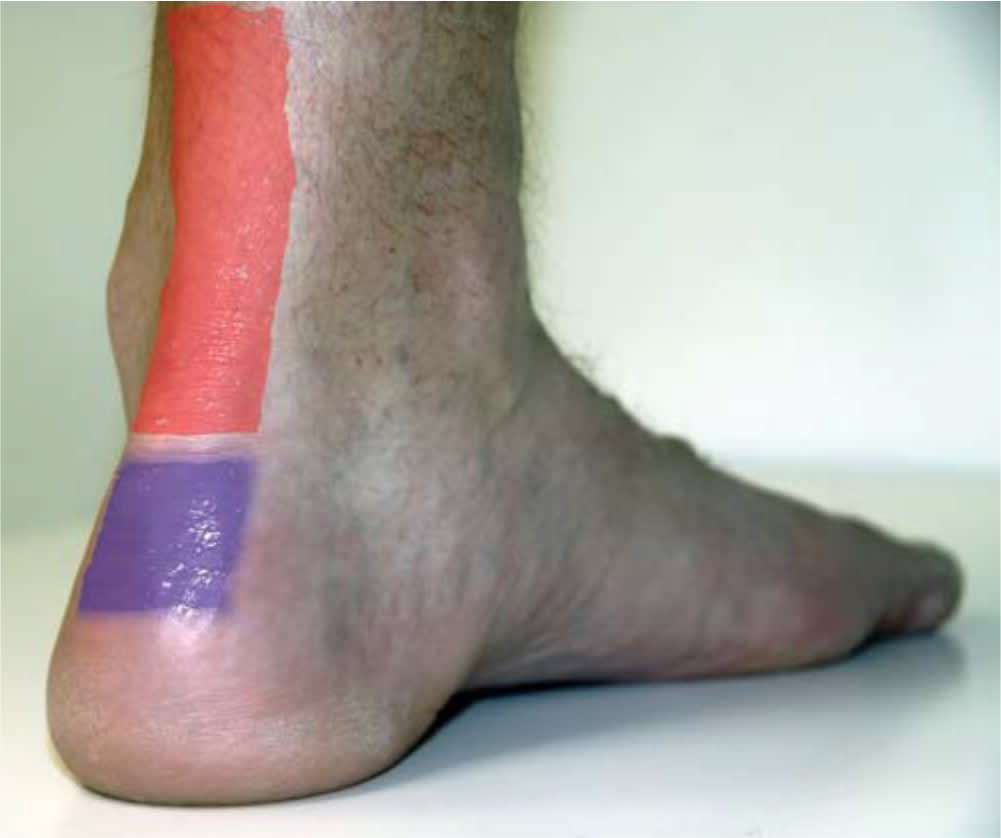
Location of pain in midsubstance Achilles tendinopathy (red) and insertional Achilles tendinopathy (purple).
Reprinted with permission from Childress MA, Beutler A. Management of chronic tendon injuries. Am Fam Physician. 2013;87(7):487.
Initial management of Achilles tendinopathy begins with risk factor modification, relative rest, and location-specific rehabilitative exercise. Midportion tendinopathy is treated with an eccentric exercise program initially (https://www.youtube.com/watch?v=omMw8Lhfmck), then a combination of eccentric and concentric exercises.79 Insertional tendinopathy does not respond as well to eccentric exercise, so rehabilitation focuses on concentric exercise.14,15,79 Orthotics to correct foot malalignment are another noninvasive and inexpensive modality that can be used for initial treatment.1 As with other tendinopathies, corticosteroid injections, topical nitroglycerin, extracorporeal shock wave therapy, and platelet-rich plasma injections are options if initial conservative treatments are not successful.
This article updates previous articles on this topic by Wilson and Best,1 and by Childress and Beutler.2
Data Sources: A PubMed search was completed in Clinical Queries using the key terms tendinitis, tendinopathy, chronic tendinopathy, and management of tendinopathy. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. We also searched the Agency for Healthcare Research and Quality evidence reports, Clinical Evidence, the Cochrane database, Essential Evidence Plus, the Institute for Clinical Systems Improvement, the National Guideline Clearinghouse database, and the evidence summary provided by AFP. Search dates: January 2019 to April 2019.