Upper gastrointestinal (GI) bleeding is defined as hemorrhage from the mouth to the ligament of Treitz. Common risk factors for upper GI bleeding include prior upper GI bleeding, anticoagulant use, high-dose nonsteroidal anti-inflammatory drug use, and older age. Causes of upper GI bleeding include peptic ulcer bleeding, gastritis, esophagitis, variceal bleeding, Mallory-Weiss syndrome, and cancer. Signs and symptoms of upper GI bleeding may include abdominal pain, lightheadedness, dizziness, syncope, hematemesis, and melena. Physical examination includes assessment of hemodynamic stability, presence of abdominal pain or rebound tenderness, and examination of stool color. Laboratory tests should include a complete blood count, basic metabolic panel, coagulation panel, liver tests, and type and screen. [corrected] A bolus of normal saline or lactated Ringer solution should be rapidly infused to correct hypovolemia and to maintain blood pressure, and blood should be transfused when hemoglobin is less than 8 g per dL. [updated] Clinical prediction guides (e.g., Glasgow-Blatchford bleeding score) are necessary for upper GI bleeding risk stratification and to determine therapy. Patients with hemodynamic instability and signs of upper GI bleeding should be offered urgent endoscopy, performed within 24 hours of presentation. A common strategy in patients with failed endoscopic hemostasis is to attempt transcatheter arterial embolization, then proceed to surgery if hemostasis is not obtained. Proton pump inhibitors should be initiated upon presentation with upper GI bleeding. Guidelines recommend high-dose proton pump inhibitor treatment for the first 72 hours post-endoscopy because this is when rebleeding risk is highest. Deciding when to restart antithrombotic therapy after upper GI bleeding is difficult because of lack of sufficient data.
Upper gastrointestinal (GI) bleeding is defined as hemorrhage from the mouth to the ligament of Treitz.1 The incidence of upper GI bleeding varies from 48 to 160 cases per 100,000 individuals.2 Upper GI bleeding mortality in the United States is decreasing. Upper GI bleeding accounts for 300,000 hospitalizations annually with a direct in-hospital economic burden of $3.3 billion.1,2 Patients with significant upper GI bleeding often have hemodynamic compromise and usually present to or are rapidly transported to the emergency department for resuscitation, stabilization, and hospitalization.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
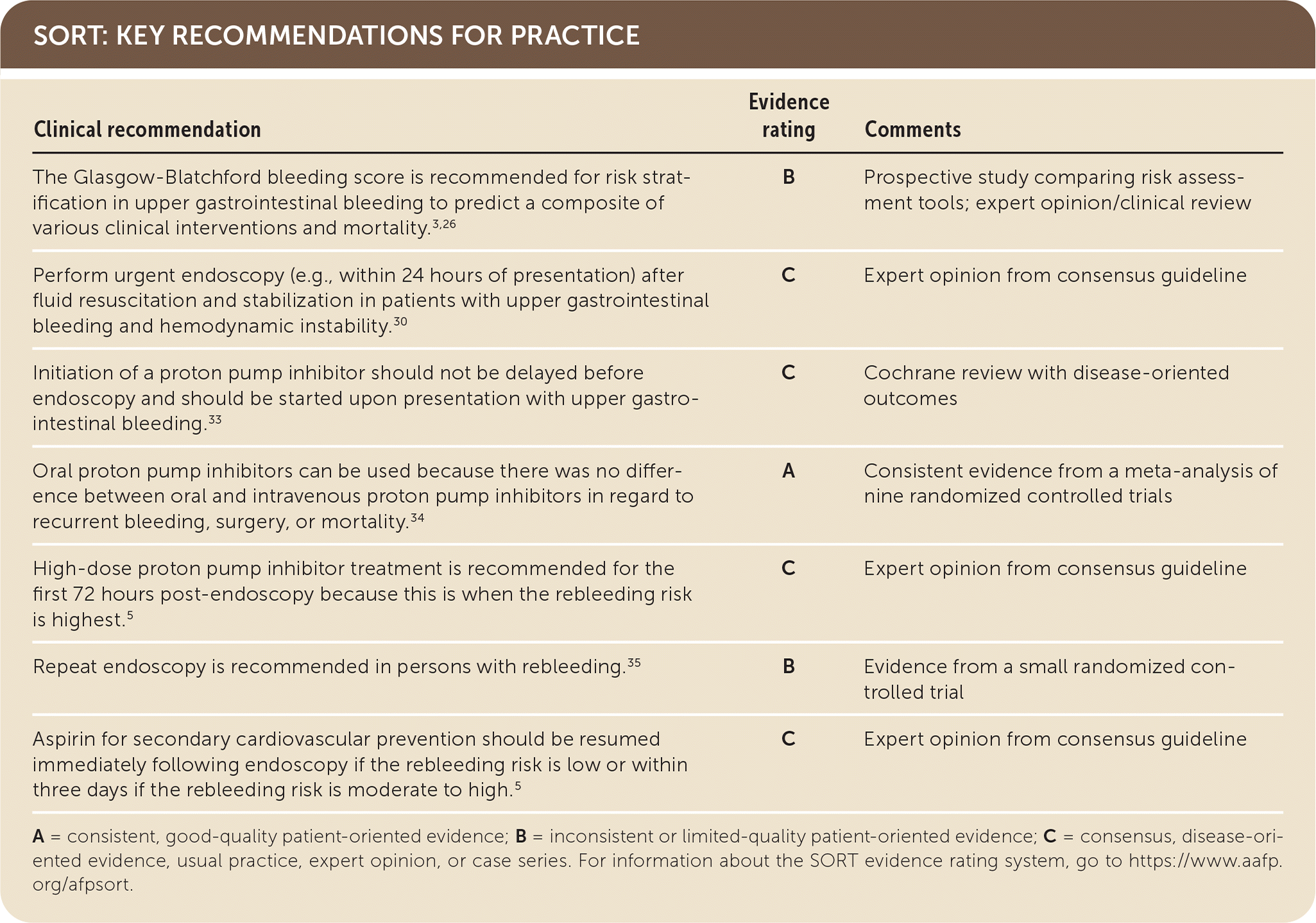
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Risk Factors
The most common risk factors for upper GI bleeding include prior upper GI bleeding (relative risk [RR] = 13.5), anticoagulant use (RR = 12.7), high-dose nonsteroidal anti-inflammatory drug (NSAID) use (RR = 5.8), and older age (RR = 5.6).1 The overall incidence of peptic ulcer disease is decreasing, likely because of an increase in the treatment of Helicobacter pylori and an increase in the use of proton pump inhibitors; however, the incidence of peptic ulcer disease not related to H. pylori or to NSAID use is increasing.1,3 Renal failure, though less common, is a risk factor for upper GI bleeding, particularly in the first year of dialysis1 because of platelet dysfunction, platelet-vessel wall interaction, and blood coagulation abnormalities.
Specific Risk Factors and Management Considerations
Causes of upper GI bleeding include peptic ulcer bleeding, gastritis, esophagitis, variceal bleeding, Mallory-Weiss syndrome, and cancer.1 Discussion of variceal bleeding and cancer is beyond the scope of this article.
PEPTIC ULCER DISEASE
NSAID use and H. pylori infection account for approximately 80% of peptic ulcer disease and peptic ulcer bleeding.1 Recent guidelines recommend testing for H. pylori in individuals with a history of or active peptic ulcer disease unless a previous cure has been documented. Treatment should be offered to all individuals testing positive for H. pylori infection.4
EROSIVE DISORDERS
Erosive disorders include esophagitis, gastritis, and duodenitis; they generally have a benign course and excellent prognosis. 5 Therapeutic endoscopy is typically not required, and many of these patients may be discharged early.5 A meta-analysis of 52 studies found that proton pump inhibitor therapy for erosive disorders was more effective than histamine-2 receptor antagonists for cumulative healing rates at eight weeks.6 Another meta-analysis of 10 studies including 15,316 persons found no difference in the healing rates of esophagitis for various proton pump inhibitors.7
MALLORY-WEISS SYNDROME
Mallory-Weiss syndrome, a mucosal tear at the distal esophagus, frequently heals spontaneously, but significant bleeding may occur. One prospective study including 1,811 individuals found that the incidence of Mallory-Weiss syndrome was 7.3 per 100,000 persons, with mortality similar to patients with peptic ulcer bleeding.8 Mortality was significantly higher in patients older than 65 or with multiple comorbid conditions.8
MEDICATIONS ASSOCIATED WITH UPPER GI BLEEDING
Antiplatelet Therapy. Aspirin carries a 37% increased risk of GI bleeding and is often prescribed as part of dual antiplatelet therapy for secondary prevention9 (Table 13,5,9–16 ). Aspirin combined with clopidogrel (Plavix) increases the risk of major GI bleeding by 93%.9 A 2018 consensus statement recommended proton pump inhibitor use in patients taking a single antiplatelet agent who have a history of upper GI bleeding or in patients taking dual antiplatelet therapy who have multiple risk factors for upper GI bleeding.17 A 2018 meta-analysis that involved four randomized controlled trials (RCTs) and included 33,492 individuals reported that the addition of a proton pump inhibitor to dual antiplatelet therapy decreased the risk of GI bleeding by 42%; however, the addition increased the occurrence of major adverse cardiovascular events by 17%.18 The role of low-dose aspirin for primary prevention is controversial; therefore, it is important to balance the risk of upper GI bleeding with the benefits of cardiovascular disease risk reduction.
TABLE 1. Review of Antithrombotics and Gastrointestinal Bleeding
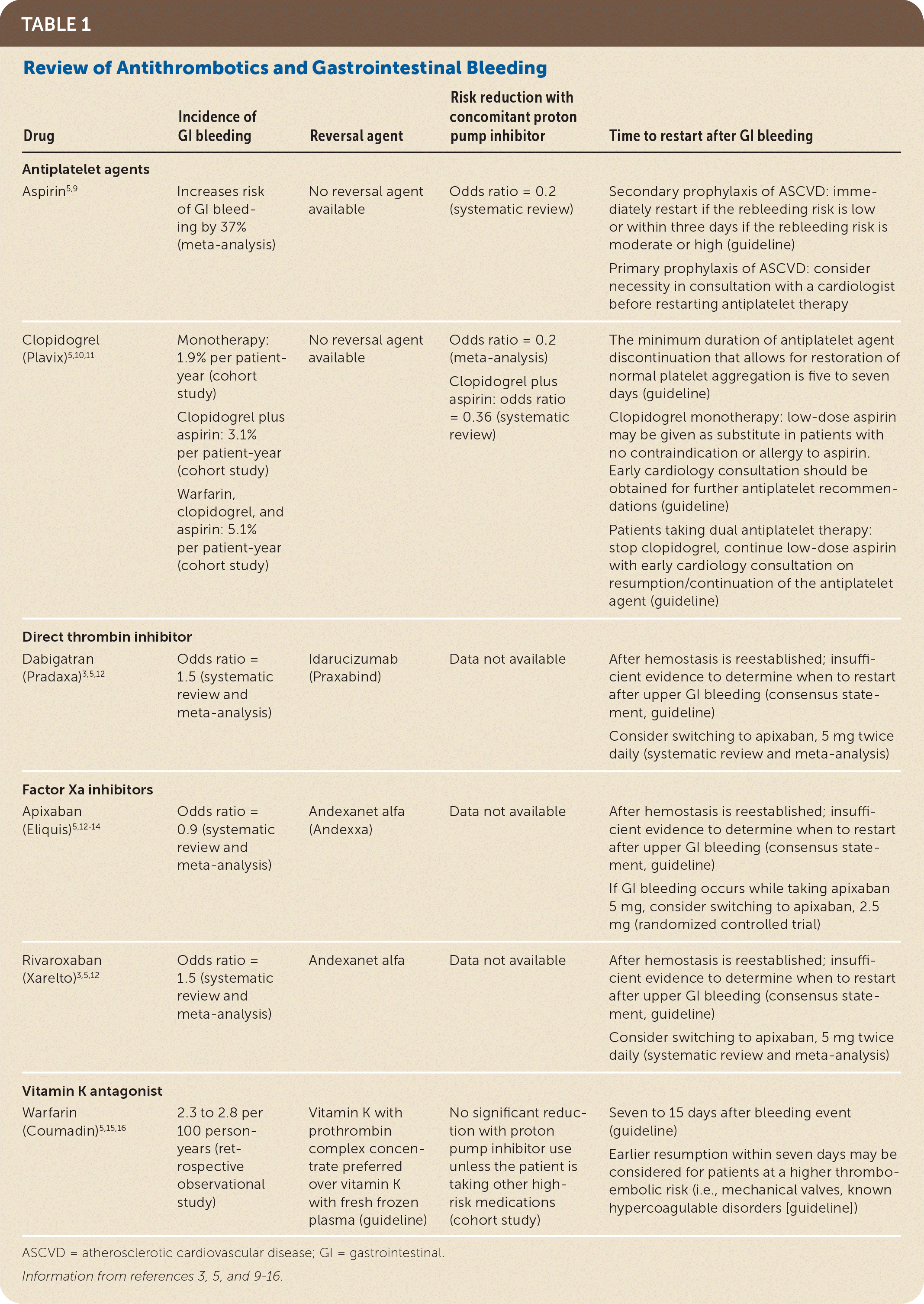
| Drug | Incidence of GI bleeding | Reversal agent | Risk reduction with concomitant proton pump inhibitor | Time to restart after GI bleeding |
|---|---|---|---|---|
| Antiplatelet agents | ||||
| Aspirin5,9 | Increases risk of GI bleeding by 37% (meta-analysis) | No reversal agent available | Odds ratio = 0.2 (systematic review) | Secondary prophylaxis of ASCVD: immediately restart if the rebleeding risk is low or within three days if the rebleeding risk is moderate or high (guideline) Primary prophylaxis of ASCVD: consider necessity in consultation with a cardiologist before restarting antiplatelet therapy |
| Clopidogrel (Plavix)5,10,11 | Monotherapy: 1.9% per patient-year (cohort study) Clopidogrel plus aspirin: 3.1% per patient-year (cohort study) Warfarin, clopidogrel, and aspirin: 5.1% per patient-year (cohort study) | No reversal agent available | Odds ratio = 0.2 (meta-analysis) Clopidogrel plus aspirin: odds ratio = 0.36 (systematic review) | The minimum duration of antiplatelet agent discontinuation that allows for restoration of normal platelet aggregation is five to seven days (guideline) Clopidogrel monotherapy: low-dose aspirin may be given as substitute in patients with no contraindication or allergy to aspirin. Early cardiology consultation should be obtained for further antiplatelet recommendations (guideline) Patients taking dual antiplatelet therapy: stop clopidogrel, continue low-dose aspirin with early cardiology consultation on resumption/continuation of the antiplatelet agent (guideline) |
| Direct thrombin inhibitor | ||||
| Dabigatran (Pradaxa)3,5,12 | Odds ratio = 1.5 (systematic review and meta-analysis) | Idarucizumab (Praxabind) | Data not available | After hemostasis is reestablished; insufficient evidence to determine when to restart after upper GI bleeding (consensus statement, guideline) Consider switching to apixaban, 5 mg twice daily (systematic review and meta-analysis) |
| Factor Xa inhibitors | ||||
| Apixaban (Eliquis)5,12–14 | Odds ratio = 0.9 (systematic review and meta-analysis) | Andexanet alfa (Andexxa) | Data not available | After hemostasis is reestablished; insufficient evidence to determine when to restart after upper GI bleeding (consensus statement, guideline) If GI bleeding occurs while taking apixaban 5 mg, consider switching to apixaban, 2.5 mg (randomized controlled trial) |
| Rivaroxaban (Xarelto)3,5,12 | Odds ratio = 1.5 (systematic review and meta-analysis) | Andexanet alfa | Data not available | After hemostasis is reestablished; insufficient evidence to determine when to restart after upper GI bleeding (consensus statement, guideline) Consider switching to apixaban, 5 mg twice daily (systematic review and meta-analysis) |
| Vitamin K antagonist | ||||
| Warfarin (Coumadin)5,15,16 | 2.3 to 2.8 per 100 person-years (retrospective observational study) | Vitamin K with prothrombin complex concentrate preferred over vitamin K with fresh frozen plasma (guideline) | No significant reduction with proton pump inhibitor use unless the patient is taking other high-risk medications (cohort study) | Seven to 15 days after bleeding event (guideline) Earlier resumption within seven days may be considered for patients at a higher thromboembolic risk (i.e., mechanical valves, known hypercoagulable disorders [guideline]) |
ASCVD = atherosclerotic cardiovascular disease; GI = gastrointestinal.
Anticoagulants. GI bleeding with warfarin (Coumadin) therapy has an incidence of 1% to 4% per year.5 Because non–vitamin K oral anticoagulants show similar or better stroke prevention than warfarin with a reduction in major bleeding, they are now recommended more than warfarin.12 A 2017 systematic review of 23 RCTs that included 94,656 individuals showed that apixaban (Eliquis), 5 mg twice daily, had lower rates of GI bleeding than any other non–vitamin K oral anticoagulant at any dose.12
NSAIDs. NSAIDs are associated with peptic ulcer disease and peptic ulcer bleeding. Celecoxib (Celebrex) has been associated with less peptic ulcer disease/peptic ulcer bleeding than other NSAIDs; however, this benefit is lost when it is combined with low-dose aspirin. If the use of systemic NSAID therapy is unavoidable and there is a high risk of upper GI bleeding, the lowest dose of celecoxib or naproxen with a gastroprotective agent (i.e., proton pump inhibitor, high-dose histamine-2 receptor antagonist, misoprostol [Cytotec]) should be considered.19 Patients should be tested for H. pylori before initiating long-term (at least four weeks) NSAID use.20
Selective Serotonin Reuptake Inhibitors (SSRIs). A meta-analysis of 22 studies that included more than 1 million participants found that SSRI use increased the risk of upper GI bleeding by 55%.21 The risk of upper GI bleeding increases when SSRIs are used concomitantly with NSAIDs (272%) or antiplatelets (148%).21 However, a 2017 cohort study including 14,343 participants demonstrated no association of SSRI treatment with an increased risk of endoscopy-refractory bleeding, rebleeding, or 30-day mortality.22 It is reasonable to continue SSRI use unless the patient has had a previous bleed and is on concomitant medications that also increase bleeding risk, in which case an alternative to SSRIs may be warranted.
Clinical Presentation
Signs and symptoms of upper GI bleeding may include abdominal pain, lightheadedness, dizziness, syncope, hematemesis, and melena. 23 Physicians should inquire about prior upper GI bleeding, history of coagulopathy, and use of antithrombotics, NSAIDs, or SSRIs. To assess the risk of cirrhosis and variceal bleeding, physicians should inquire about prior liver disease, viral hepatitis, and alcohol use. Physical examination includes assessment of hemodynamic stability, presence of abdominal pain or rebound tenderness, and examination of stool color. Predictive factors of upper GI bleeding include history of melena, melanotic stool on examination, tachycardia, and hemoglobin less than 8 g per dL (80 g per L).24
Initial Evaluation and Stabilization
The initial evaluation includes a complete history, examination, and laboratory evaluation. The goal of the initial evaluation is to determine the severity of the bleeding, to identify the possible source of bleeding, and to guide management decisions, such as hospitalization, transfusion, and diagnostic testing.3 Patients should maintain “nothing by mouth” (except medication) orders while being evaluated.
Laboratory tests should include a complete blood count, basic metabolic panel, coagulation panel, liver tests, and type and screen.23 [corrected] Measurement of hemoglobin and hematocrit levels should be repeated based on the severity of the upper GI bleeding, clinical judgment, and hemodynamic stability.23 A blood urea nitrogen/creatinine ratio greater than 36 is suggestive of upper GI bleeding when distinguishing it from lower GI bleeding (sensitivity 90%).23 In patients with significant upper GI bleeding, place two large-bore peripheral intravenous catheters and a bolus of normal saline or lactated Ringer solution to correct hypovolemia and to maintain blood pressure because tachycardia and hypotension may be signs of significant blood loss.23 Regular monitoring of heart rate, blood pressure, and pulse oximetry is necessary during resuscitation and stabilization. Endotracheal intubation may be necessary to protect the airway in patients with significant and ongoing bleeding.3 Management of patients with life-threatening bleeding warranting intensive monitoring is beyond the scope of this article.
There are no RCTs or guidelines to support the use of nasogastric lavage. A retrospective study of 632 individuals admitted with upper GI bleeding found that nasogastric lavage did not lower mortality, surgery, or transfusion requirements.23,25 The use of prokinetic agents before endoscopy is controversial; however, a meta-analysis of eight RCTs that included 598 individuals found that an erythromycin infusion before endoscopy improved visualization, reduced the need for second-look endoscopy, and reduced hospital stay.3 Clinical prediction guides are necessary for upper GI bleeding risk stratification and to determine therapy. The Glasgow-Blatchford bleeding score (https://www.mdcalc.com/glasgow-blatchford-bleeding-score-gbs) is used to predict a composite of various clinical interventions (such as endoscopic treatment or surgery) and mortality, and it appears to be superior to other prediction guides.3 A Glasgow-Blatchford bleeding score less than or equal to one predicts survival without the need for intervention (sensitivity 98.6%, specificity 34.6%),26 and these patients may be discharged from the emergency department for outpatient treatment and diagnostic testing.
TRANSFUSIONS AND COAGULOPATHY
Current guidelines recommend blood transfusion for patients with upper GI bleeding when hemoglobin is less than 8 g per dL (80 g per L), including patients with coronary artery disease, recent cardiac surgery, or hematologic malignancies.27 [updated] Patients with coronary artery disease, recent cardiac surgery, or hematologic malignancies require more liberal transfusion strategies. 23 Expert consensus recommends platelet transfusion with platelet counts less than 50 × 103 per uL (50 × 109 per L).28 Patients with upper GI bleeding and an international normalized ratio (INR) greater than 1.5 have increased mortality compared with patients who have an INR less than 1.5.29 Expert consensus has concluded that correction of moderate coagulopathy (i.e., INR of 1.5 to 2.5) should not delay endoscopy, but INR greater than 2.5 should be corrected before endoscopy 27 (Table 13,5,9–16 ).
Treatments
ENDOSCOPY
Patients with hemodynamic instability and signs of upper GI bleeding should be offered urgent endoscopy (within 24 hours of presentation or onset of bleeding) after fluid resuscitation and stabilization.30 The majority of efficacy data on endoscopic therapy is from trials including individuals who have peptic ulcer bleeding. The recommended endoscopic methods for peptic ulcer bleeding include injection of epinephrine, injection of other sclerosants, bipolar electrocoagulation probes, heater probes, or endoclips.3 Endoscopic injection of epinephrine should not be used as monotherapy in patients with peptic ulcer bleeding because it has higher rates of failure than epinephrine injection combined with another modality (i.e., endoclips or heater probe).31
ARTERIAL EMBOLIZATION AND SURGERY
Transcatheter arterial embolization is typically performed after endoscopy if endoscopy has failed to control bleeding. A common strategy in patients with failed endoscopic hemostasis is to attempt transcatheter arterial embolization, then proceed to surgery if hemostasis is not obtained. However, a systematic review of nine studies including 711 individuals found that surgery was associated with a 59% lower risk of rebleeding compared with transcatheter arterial embolization with no difference in mortality.32
PROTON PUMP INHIBITORS
Proton pump inhibitors should be started upon presentation with upper GI bleeding.33 Initiation of a proton pump inhibitor should not be delayed before endoscopy.___ANCHOR_START___/afp20200301p294-sort3___ANCHOR_END___Oral proton pump inhibitors may be preferred over intravenous ones because of cost savings when used in conjunction with endoscopic treatment.34 A 2017 meta-analysis of nine RCTs showed no difference between oral and intravenous proton pump inhibitors in recurrent bleeding, surgery, or mortality.34 Guidelines recommend high-dose proton pump inhibitor treatment (i.e., esomeprazole [Nexium], 80 mg per day) for the first 72 hours post-endoscopy because this is when rebleeding risk is highest.5 After 72 hours of high-dose therapy, patients with high-risk endoscopic lesions should receive twice-daily proton pump inhibitors through day 14, whereas those with low-risk lesions require only once-daily proton pump inhibitor therapy. Once-daily proton pump inhibitor therapy should be continued for four to eight weeks in patients with peptic ulcer bleeding. In patients with peptic ulcer bleeding, the benefits of proton pump inhibitor therapy outweigh the risks.3
Rebleeding and Follow-up Strategies
REBLEEDING
Repeat endoscopy is recommended in persons with rebleeding because hemostasis is achieved in 73% of these patients.35 If additional bleeding occurs after the second endoscopy, transcatheter arterial embolization or surgery may be attempted.3
WHEN TO RESTART ANTITHROMBOTIC THERAPY
Deciding when to restart antithrombotic therapy after upper GI bleeding is difficult because of a lack of sufficient data. A 2018 meta-analysis of seven observational studies including 5,685 individuals who had atrial fibrillation showed the resumption of anticoagulant therapy after GI bleeding vs. not restarting therapy was associated with a 10.8% absolute risk reduction for all-cause mortality (number needed to treat = 9).36 Guidelines recommend warfarin be resumed seven to 15 days after a bleeding event.5,13 Resumption of warfarin should be considered within seven days for patients at a higher thromboembolic risk (i.e., patients with mechanical valves or known hypercoagulable disorders).5 Patients who experience GI bleeding on a non–vitamin K oral anticoagulant should be switched to apixaban.13 If bleeding occurs while taking apixaban 5 mg, dose reduction is recommended.13
Guidelines recommend resumption of aspirin for secondary prevention of cardiovascular disease immediately following endoscopy if the rebleeding risk is low or within three days if the rebleeding risk is moderate or high.5 One RCT of 156 individuals analyzed immediate reintroduction of aspirin with proton pump inhibitors post-endoscopy and found that patients who did not resume aspirin had an increase in all-cause mortality at eight weeks.3 When to resume dual antiplatelet therapy is challenging because these patients often have acute coronary syndrome or recent coronary stent placement. In individuals requiring dual antiplatelet therapy, aspirin should be resumed first after hemostasis is reestablished because of reduced bleeding risk compared with clopidogrel.37 Early resumption of clopidogrel within five to seven days should be considered in high-risk patients (i.e., those who have had a coronary stent within 30 days).37
H. PYLORI ERADICATION
First-line therapy choices for H. pylori are numerous, and the choice of therapy should be individualized. When choosing a treatment regimen, physicians should evaluate previous antibiotic exposure, allergies, and local resistance patterns. A test of cure is recommended at least four weeks after the completion of antibiotics and one to two weeks after completion of proton pump inhibitor therapy.4
This article updates a previous article on this topic by Wilkins, et al.38
Data Sources: PubMed, Cochrane Library, TRIP databases, Essential Evidence Plus, and guidelines.gov were searched for relevant articles using variations of the keywords upper gastrointestinal, peptic ulcer, Helicobacter pylori, esophagitis, or Mallory-Weiss in combination with bleeding. Additional search terms such as risk stratification, treatment, or drug names were added in additional searches to focus on a particular topic. The search included guidelines, meta-analyses, and randomized controlled trials. Search dates: May and December 2018; November 2019.

