The use of diagnostic radiography has doubled in the past two decades. Image Gently (children) and Image Wisely (adults) are multidisciplinary initiatives that seek to reduce radiation exposure by eliminating unnecessary procedures and offering best practices. Patients with an estimated glomerular filtration rate less than 30 mL per minute per 1.73 m2 may have increased risk of nephropathy when exposed to iodinated contrast media and increased risk of nephrogenic systemic fibrosis when exposed to gadolinium-based contrast agents. American College of Radiology Appropriateness Criteria can help guide specific diagnostic imaging choices. Noncontrast head computed tomography is the first-line modality when a stroke is suspected. Magnetic resonance imaging stroke protocols and computed tomography perfusion scans can augment evaluation and potentially expand pharmacologic and endovascular therapy timeframes. Imaging should be avoided in patients with uncomplicated headache syndromes unless the history or physical examination reveals red flag features. Cardiac computed tomography angiography, stress echocardiography, and myocardial perfusion scintigraphy (nuclear stress test) are appropriate for patients with chest pain and low to intermediate cardiovascular risk and have comparable sensitivity and specificity. Computed tomography pulmonary angiography is the preferred test for high-risk patients or those with a positive d-dimer test result, and ventilation-perfusion scintigraphy is reserved for patients with an estimated glomerular filtration rate less than 30 mL per minute per 1.73 m2 or a known contrast allergy. Computed tomography with intravenous contrast is preferred for evaluating adults with suspected appendicitis; however, ultrasonography should precede computed tomography in children, and definitive treatment should be initiated if positive. Ultrasonography is the first-line modality for assessing right upper quadrant pain suggestive of biliary disease. Mass size and patient age dictate surveillance recommendations for adnexal masses. Imaging should not be performed for acute (less than six weeks) low back pain unless red flag features are found on patient history. Ultrasonography should be used for the evaluation of suspicious thyroid nodules identified incidentally on computed tomography.
Estimates suggest that 30% of all U.S. health expenditures are a result of waste, with approximately $100 billion lost on overtreatment and low-value care; inappropriate radiography is a major component.1 The use of advanced imaging, including computed tomography (CT), magnetic resonance imaging (MRI), ultrasonography, and nuclear medicine, has doubled in a 16-year period, accounting for 11% of allowed Medicare charges in 2018.2,3 Awareness of risks, benefits, and recommendations related to radiography enhances shared decision-making and reduces unnecessary testing.2,4
WHAT'S NEW ON THIS TOPIC
Diagnostic Imaging
The use of advanced imaging, including computed tomography, magnetic resonance imaging, ultrasonography, and nuclear medicine, has doubled in the past 16 years, accounting for 11% of 2018 allowed Medicare charges.
The Protecting Access to Medicare Act, which will be fully implemented as of January 1, 2022, requires clinicians to consult appropriate use criteria via a clinical decision support mechanism before ordering advanced diagnostic imaging services for Medicare Part B patients.
The proportion of personal radiation exposure attributed to medical sources rose from 5% in the 1980s to more than 50% by 2009, with computed tomography scans constituting 25% of all radiation exposure despite a decrease in radiation per scan.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
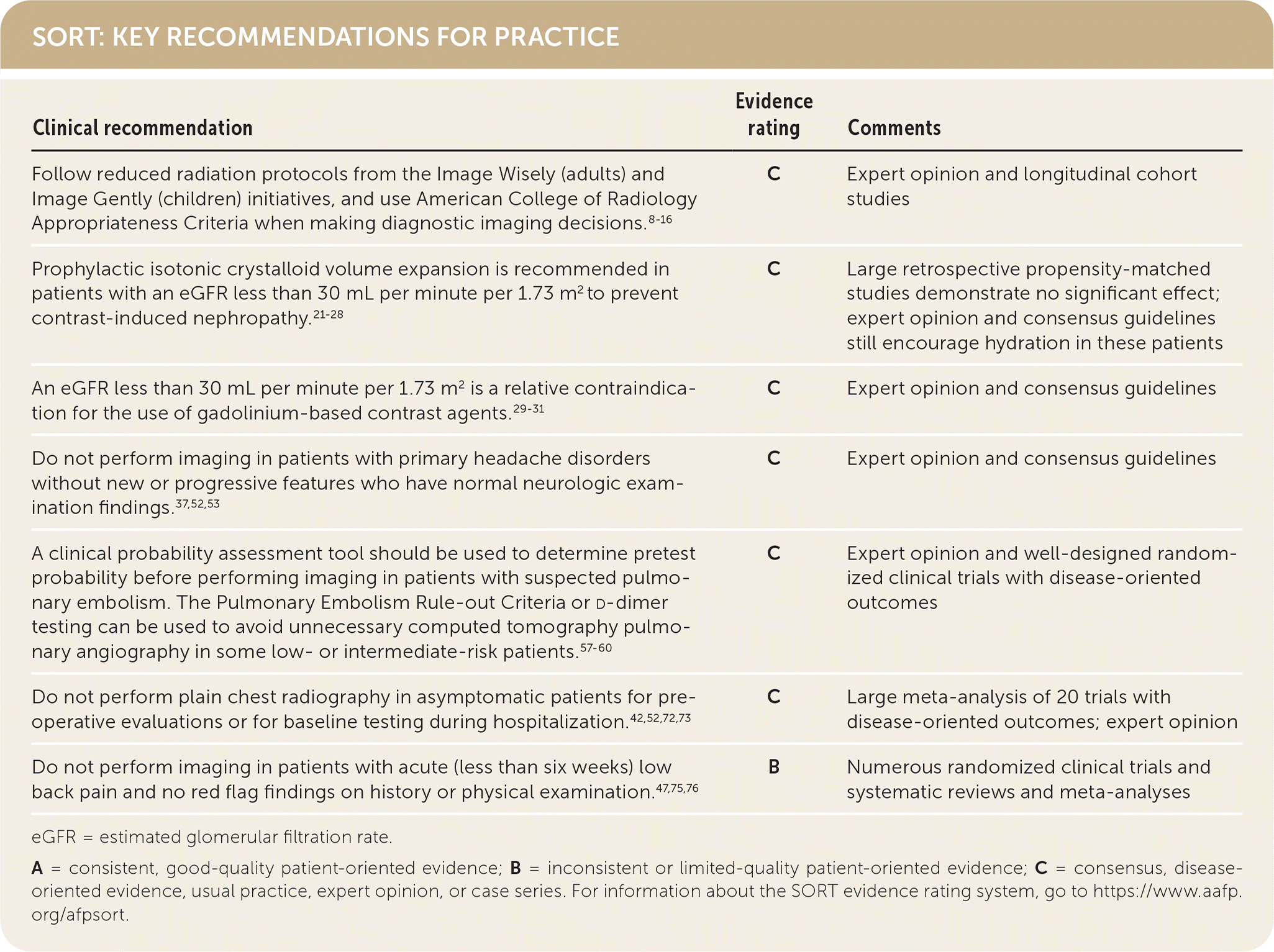
eGFR = estimated glomerular filtration rate.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
BEST PRACTICES IN DIAGNOSTIC IMAGING

| Recommendation | Sponsoring organization |
|---|---|
| Avoid using CT as the first-line imaging modality in the evaluation of suspected appendicitis in children. Ultrasonography should be performed first, with CT or magnetic resonance imaging considered in equivocal cases. | American Academy of Pediatrics |
| Do not perform CT for evaluation of suspected appendicitis in children until after ultrasonography has been considered as an option. | American College of Radiology |
| CT scans are not necessary in the routine evaluation of abdominal pain. | American Academy of Pediatrics |
| Avoid CT scans of the head in emergency department patients with minor head injury who are at low risk based on validated decision rules. | American College of Emergency Physicians |
| CT scans are not necessary in the evaluation of minor head injuries. | American Academy of Pediatrics |
| Do not routinely obtain CT scans in children with mild head injuries. | American Association of Neurological Surgeons and Congress of Neurological Surgeons |
CT = computed tomography.
Source: For more information on the Choosing Wisely Campaign, see https://www.choosingwisely.org. For supporting citations and to search Choosing Wisely recommendations relevant to primary care, see https://www.aafp.org/afp/recommendations/search.htm.
The 2014 Protecting Access to Medicare Act (PAMA) requires clinicians to consult appropriate use criteria via a clinical decision support tool before ordering advanced diagnostic imaging services for Medicare Part B patients.5 Appropriate use criteria have been employed since 2018 as part of PAMA, but on January 1, 2020, the year-long education and operations testing period began. During this period, imaging orders that do not include use of a clinician decision support will still be performed and covered without payment consequences, although consultation with a clinical decision support mechanism is recommended. The Centers for Medicare and Medicaid Services extended this trial period for another year, until December 2021.6 The list of qualifying clinical decision support mechanisms was recently updated.6
PAMA will initially focus on eight areas: suspected or diagnosed coronary artery disease; suspected pulmonary embolism; traumatic and nontraumatic headache; hip pain; low back pain; shoulder pain, including suspected rotator cuff injury; suspected or diagnosed lung cancer (primary or metastatic); and neck pain. The program will be fully implemented as of January 1, 2022, after which payment consequences could result from failure to employ appropriate use criteria.5,7
Reduced radiation protocols from the Image Wisely (adults) and Image Gently (children) initiatives and American College of Radiology (ACR) Appropriateness Criteria should be used when making diagnostic imaging decisions.8–16 ACR Appropriateness Criteria cover 193 diagnostic imaging and interventional radiology topics, with 942 clinical variants and more than 1,680 clinical scenarios.17
Radiation Safety and Risk
Most data on cumulative ionizing radiation exposure and cancer incidence come from survivors of atomic tragedies and those working in the nuclear industry, reinforcing a dose response relative to cancer risk.8–10
Radiation dose can be measured in grays (Gy; a measure of absorbed dose) or sieverts (Sv; a measure of equivalent dose). Ionizing radiation exposure varies by type of imaging study (Table 1).18,19 The proportion of radiation exposures in the United States that is attributed to medical sources has risen from 5% in the 1980s to more than 50% in 2009, with CT scans constituting 25% of all exposures despite a decrease in radiation per scan.20 The estimated lifetime relative risk of developing cancer is 5% per Sv of radiation.12 Based on this, the U.S. Food and Drug Administration estimates that the risk of developing a fatal malignancy associated with one CT scan is approximately one out of 2,000.13 This risk is reliant on the patient's age at exposure, the organs exposed, the patient's sex, and other variables.
TABLE 1. Approximate Ionizing Radiation Exposure from Common Imaging
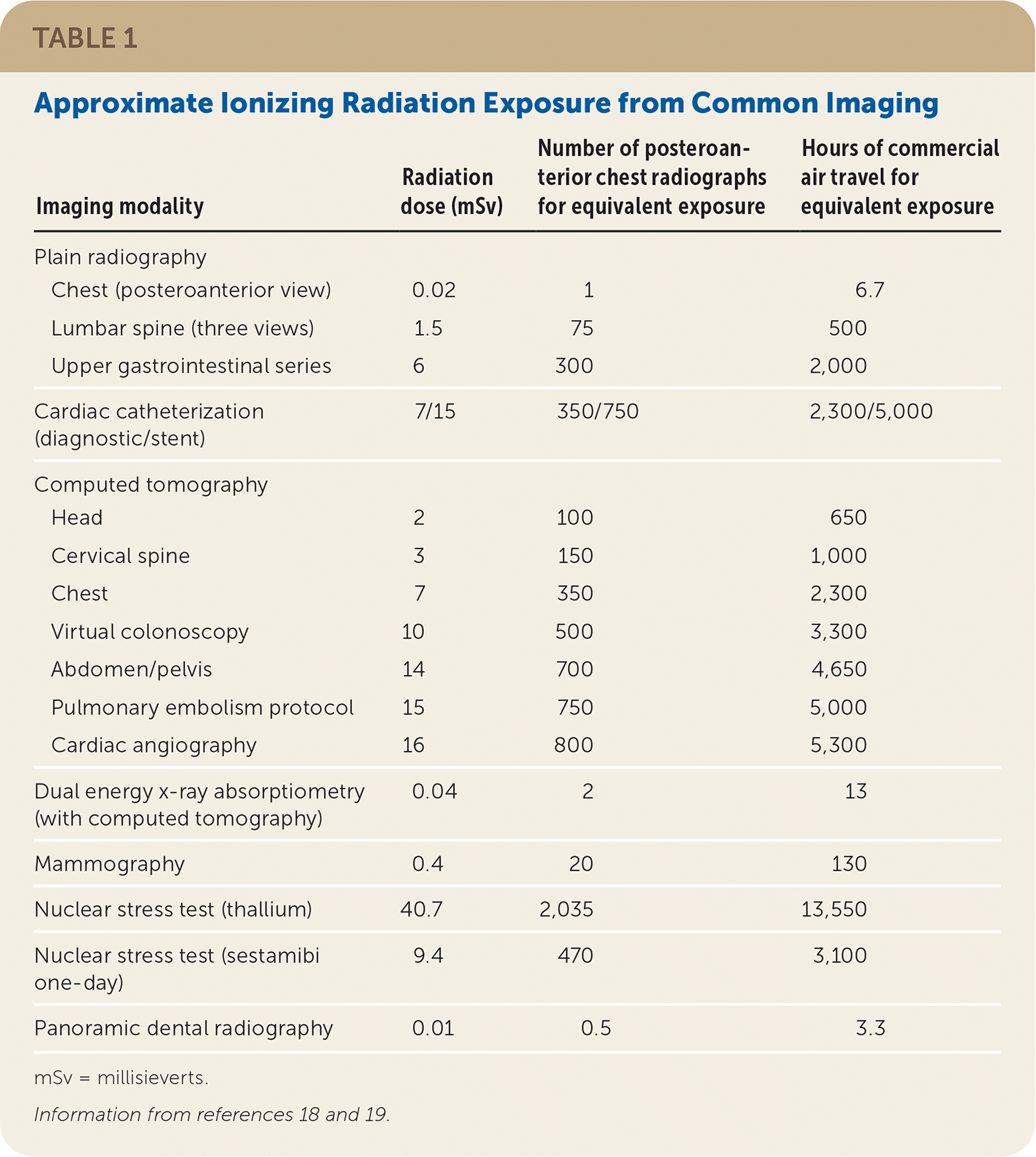
| Imaging modality | Radiation dose (mSv) | Number of posteroanterior chest radiographs for equivalent exposure | Hours of commercial air travel for equivalent exposure |
|---|---|---|---|
| Plain radiography | |||
| Chest (posteroanterior view) | 0.02 | 1 | 6.7 |
| Lumbar spine (three views) | 1.5 | 75 | 500 |
| Upper gastrointestinal series | 6 | 300 | 2,000 |
| Cardiac catheterization (diagnostic/stent) | 7/15 | 350/750 | 2,300/5,000 |
| Computed tomography | |||
| Head | 2 | 100 | 650 |
| Cervical spine | 3 | 150 | 1,000 |
| Chest | 7 | 350 | 2,300 |
| Virtual colonoscopy | 10 | 500 | 3,300 |
| Abdomen/pelvis | 14 | 700 | 4,650 |
| Pulmonary embolism protocol | 15 | 750 | 5,000 |
| Cardiac angiography | 16 | 800 | 5,300 |
| Dual energy x-ray absorptiometry (with computed tomography) | 0.04 | 2 | 13 |
| Mammography | 0.4 | 20 | 130 |
| Nuclear stress test (thallium) | 40.7 | 2,035 | 13,550 |
| Nuclear stress test (sestamibi one-day) | 9.4 | 470 | 3,100 |
| Panoramic dental radiography | 0.01 | 0.5 | 3.3 |
mSv = millisieverts.
According to the Biological Effects of Ionizing Radiations (BEIR) VII report, there is no single radiation dose at which harm begins to occur. Instead, there are small linear increases in risk as exposure increases.16 The risk seems to be most significant for children, in which estimates suggest 1.9 new cases of leukemia for every 10,000 head CT scans.14 Within the radiographic community, there have been concerns about the overestimation of cancer risk based on flawed analyses in BEIR VII, and several population-based studies do not corroborate the anticipated incidence of malignancy attributable to medical imaging.15
Assuming a sex and age distribution similar to that of the United States population, the BEIR VII lifetime risk model predicts that approximately one in 100 individuals would develop cancer (solid cancer or leukemia) from a single 100-mSv dose of radiation (roughly that of a contrast-enhanced CT scan of the chest, abdomen, and pelvis), whereas 42 of these 100 individuals would be expected to develop malignancy from other causes. Risk is dose-dependent in this model.16
The risks of imaging are balanced against a potential inability to formulate definitive diagnoses and treatment plans without imaging. Table 2 includes resources for radiation reduction, including cumulative radiation calculators.
TABLE 2. Resources for Managing Radiation Exposure from Imaging
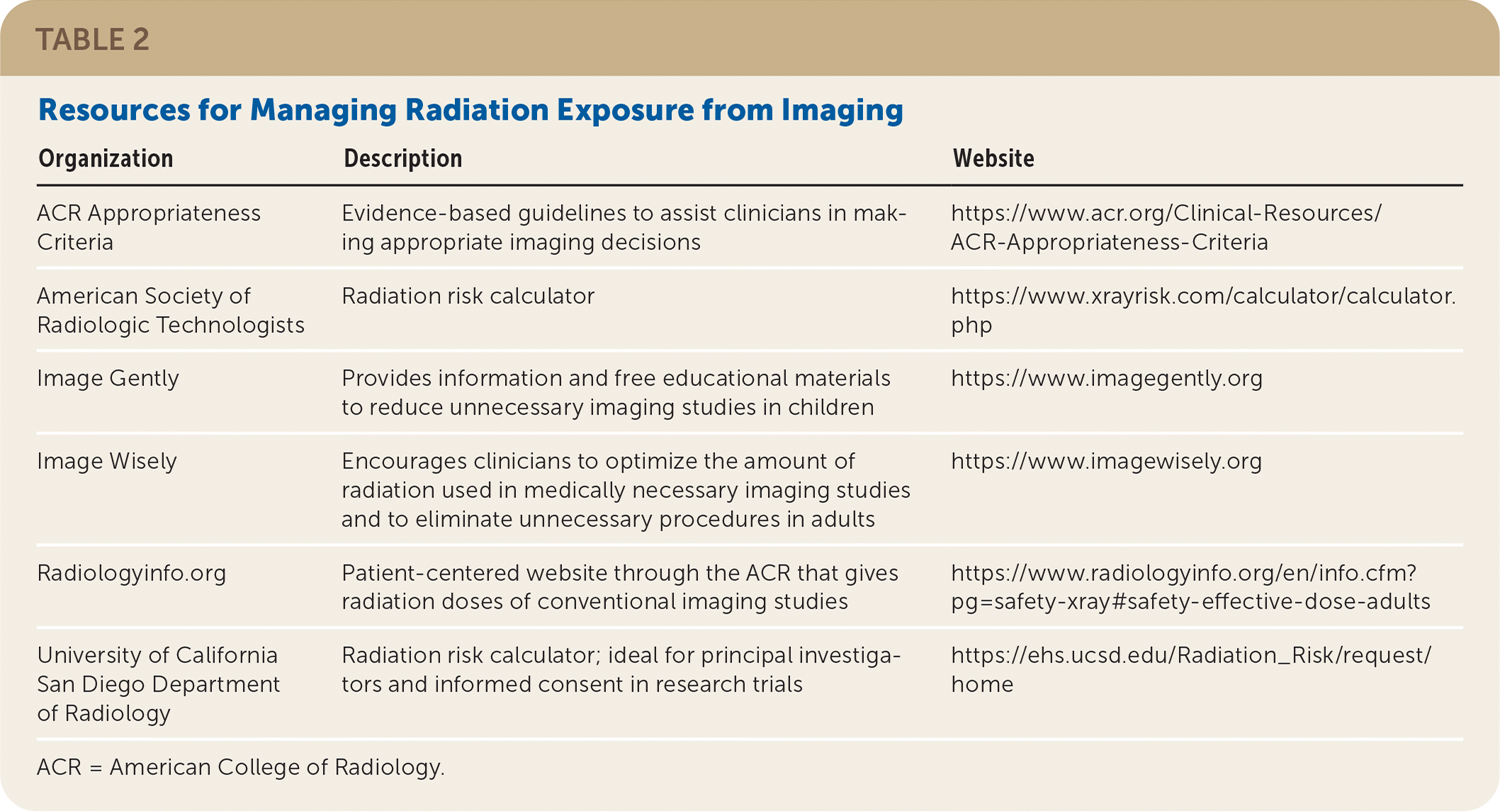
| Organization | Description | Website |
|---|---|---|
| ACR Appropriateness Criteria | Evidence-based guidelines to assist clinicians in making appropriate imaging decisions | https://www.acr.org/Clinical-Resources/ACR-Appropriateness-Criteria |
| American Society of Radiologic Technologists | Radiation risk calculator | https://www.xrayrisk.com/calculator/calculator.php |
| Image Gently | Provides information and free educational materials to reduce unnecessary imaging studies in children | https://www.imagegently.org |
| Image Wisely | Encourages clinicians to optimize the amount of radiation used in medically necessary imaging studies and to eliminate unnecessary procedures in adults | https://www.imagewisely.org |
| Radiologyinfo.org | Patient-centered website through the ACR that gives radiation doses of conventional imaging studies | https://www.radiologyinfo.org/en/info.cfm?pg=safety-xray#safety-effective-dose-adults |
| University of California San Diego Department of Radiology | Radiation risk calculator; ideal for principal investigators and informed consent in research trials | https://ehs.ucsd.edu/Radiation_Risk/request/home |
ACR = American College of Radiology.
CONTRAST-INDUCED NEPHROPATHY
Contrast-induced nephropathy is a proposed mechanism of postcontrast acute kidney injury, presuming a direct nephrotoxic effect of iodinated CT contrast media.21,22 The data support only an association between contrast media and acute kidney injury, although the diagnostic criteria for postcontrast acute kidney injury is well-defined as at least one of the following occurring within 48 hours of contrast administration: (1) absolute increase in serum creatinine level of 0.3 mg per dL (26.52 μmol per L) or more from baseline, (2) a relative increase in serum creatinine level of 50% or more from baseline, and (3) a urine output reduced to 0.5 mL per kg per hour or less for at least six hours.23
A meta-analysis of nonrandomized studies including 25,000 patients showed no increased risk of acute kidney injury with contrast (6.4%) vs. no contrast (6.5%).24 Subsequently, two propensity-matched studies of 21,000 patients with normal renal function and 12,500 patients with preexisting renal dysfunction demonstrated no increased risk of acute kidney injury following the use of contrast media.25,26 A similar propensity-matched study showed no increased risk of contrast-induced nephropathy in patients with normal renal function, but it did show an increased risk in patients with a serum creatinine level greater than 1.2 mg per dL (106.08 μmol per L) or an estimated glomerular filtration rate (eGFR) less than 30 mL per minute per 1.73 m2.27
Based on the evidence, and in accordance with a recent consensus statement, intravenous contrast is not independently nephrotoxic in patients with a stable baseline eGFR of 45 mL per minute per 1.73 m2 or greater, and it is not nephrotoxic (or rarely so) in patients with an eGFR of 30 to 44 mL per minute per 1.73 m2.23,28 Studies are mixed for patients with an eGFR less than 30 mL per minute per 1.73 m2; therefore, the ACR continues to recommend prophylactic isotonic crystalloid volume expansion (numerous volume and time-based protocols exist, with no one preferred regimen).21–28
NEPHROGENIC SYSTEMIC FIBROSIS
Nephrogenic systemic fibrosis involves thickening of the skin and subcutaneous tissue and can be rapidly progressive, leading to contractures, joint immobility, and rarely death.29 It is presumed to occur after exposure to gadolinium-based contrast agents (e.g., with contrast-enhanced MRI). It typically occurs within days to months but has been reported years after contrast exposure.30 In patients with an eGFR less than 30 mL per minute per 1.73 m2, the risk of nephrogenic systemic fibrosis is 1% to 7% per exposure to older gadolinium-based contrast agents.31 This risk is inversely proportional to eGFR level and increases with higher doses of contrast or repeated exposures. There are no reported cases of nephrogenic systemic fibrosis in patients with normal renal function (eGFR greater than 60 mL per minute per 1.73 m2), and the risk is low with standard doses of modern gadolinium-based contrast.23
MRI CONTRAINDICATIONS
Many contraindications to MRI are dependent on the machine, patient history, and presence of indwelling devices.32 Absolute contraindications include the presence of metallic foreign bodies in the eye, contact lenses that monitor intraocular pressure and include portions of metal, some insulin pumps (internal and external), and temporary external transvenous pacemaker leads. The presence of an implantable loop recorder, an implantable cardiac defibrillator, a reflux management system, or a pacemaker requires someone experienced in managing these devices and the potential complications if a device fails.32,33 Although manufacturers label many devices as compatible with MRI, imaging requires careful consideration and coordination with the implanting specialty and radiology team. Information about the safety of these devices during imaging is available from manufacturers and from other sources, such as http://www.MRISafety.com/List.html.
Medical Decision Support and Appropriate Use of Imaging
The American Academy of Family Physicians (AAFP) position paper on radiology encourages judicious use of diagnostic radiography to reduce adverse effects and avoid overuse.34 The AAFP recommends using evidence-based criteria, such as ACR Appropriateness Criteria, to assist in clinical decision-making.34 The ACR Appropriateness Criteria rank imaging tests for the evaluation of various conditions from 1 (least recommended) to nine (most recommended), with a relative radiation score.35 ACR Appropriateness Criteria scores for selected clinical scenarios are presented in Table 3.36–47
TABLE 3. American College of Radiology Appropriateness Criteria Ratings for Selected Diagnostic Imaging Studies
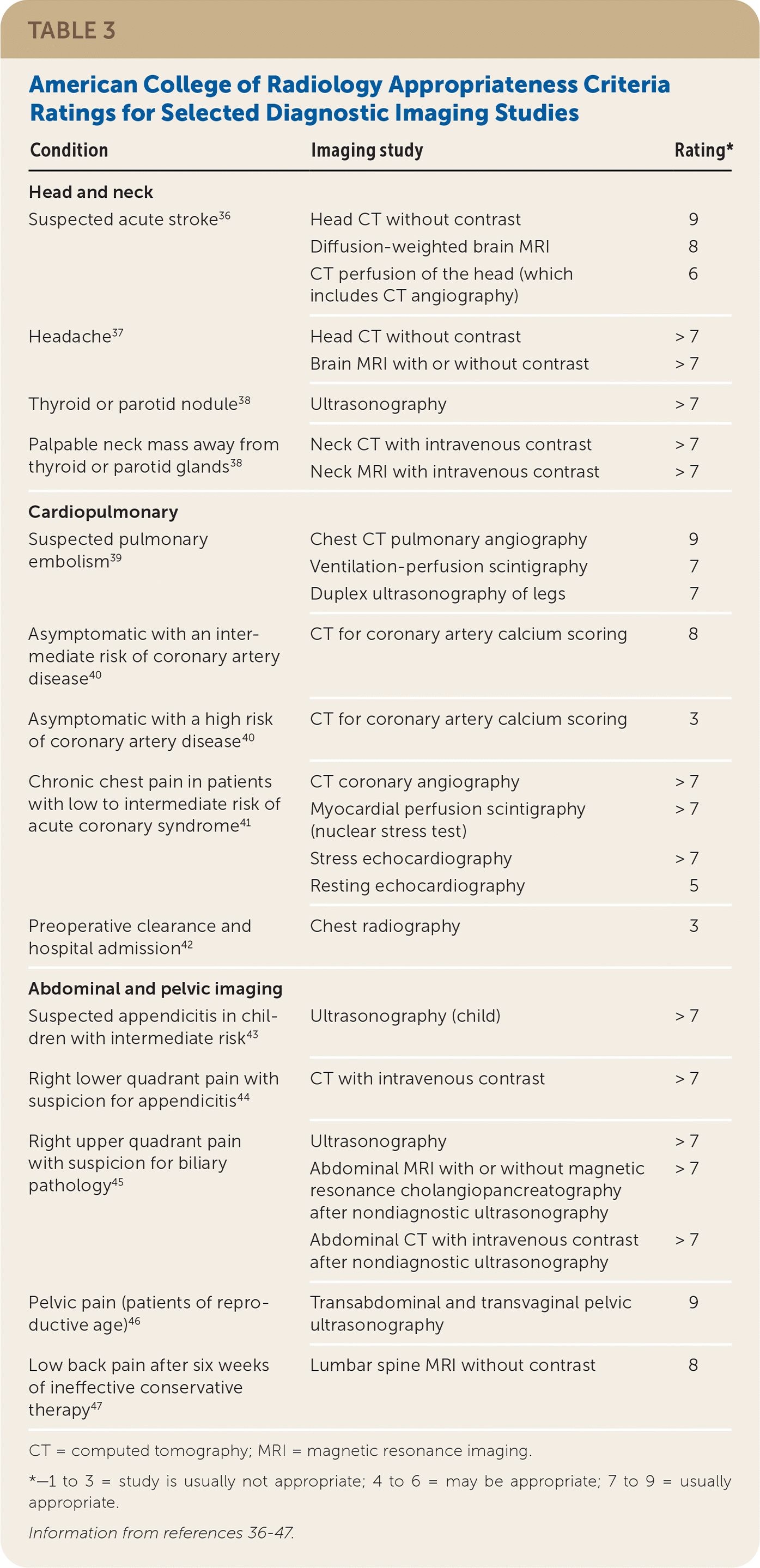
| Condition | Imaging study | Rating* |
|---|---|---|
| Head and neck | ||
| Suspected acute stroke36 | Head CT without contrast | 9 |
| Diffusion-weighted brain MRI | 8 | |
| CT perfusion of the head (which includes CT angiography) | 6 | |
| Headache37 | Head CT without contrast | > 7 |
| Brain MRI with or without contrast | > 7 | |
| Thyroid or parotid nodule38 | Ultrasonography | > 7 |
| Palpable neck mass away from thyroid or parotid glands38 | Neck CT with intravenous contrast | > 7 |
| Neck MRI with intravenous contrast | > 7 | |
| Cardiopulmonary | ||
| Suspected pulmonary embolism39 | Chest CT pulmonary angiography | 9 |
| Ventilation-perfusion scintigraphy | 7 | |
| Duplex ultrasonography of legs | 7 | |
| Asymptomatic with an intermediate risk of coronary artery disease40 | CT for coronary artery calcium scoring | 8 |
| Asymptomatic with a high risk of coronary artery disease40 | CT for coronary artery calcium scoring | 3 |
| Chronic chest pain in patients with low to intermediate risk of acute coronary syndrome41 | CT coronary angiography | > 7 |
| Myocardial perfusion scintigraphy (nuclear stress test) | > 7 | |
| Stress echocardiography | > 7 | |
| Resting echocardiography | 5 | |
| Preoperative clearance and hospital admission42 | Chest radiography | 3 |
| Abdominal and pelvic imaging | ||
| Suspected appendicitis in children with intermediate risk43 | Ultrasonography (child) | > 7 |
| Right lower quadrant pain with suspicion for appendicitis44 | CT with intravenous contrast | > 7 |
| Right upper quadrant pain with suspicion for biliary pathology 45 | Ultrasonography | > 7 |
| Abdominal MRI with or without magnetic resonance cholangiopancreatography after nondiagnostic ultrasonography | > 7 | |
| Abdominal CT with intravenous contrast after nondiagnostic ultrasonography | > 7 | |
| Pelvic pain (patients of reproductive age)46 | Transabdominal and transvaginal pelvic ultrasonography | 9 |
| Low back pain after six weeks of ineffective conservative therapy 47 | Lumbar spine MRI without contrast | 8 |
CT = computed tomography; MRI = magnetic resonance imaging.
*—1 to 3 = study is usually not appropriate; 4 to 6 = may be appropriate; 7 to 9 = usually appropriate.
HEAD AND NECK
Suspected Acute Stroke. Noncontrast head CT should be performed in any patient with suspected stroke because it is required to determine the need for thrombolysis.48 The choice of additional imaging modalities varies depending on patient presentation, medical institution, and previous management decisions. Through advanced imaging protocols, typical symptomatic treatment windows can be expanded based on imaging characteristics of ischemic lesions.49
Diffusion-weighted MRI increases the sensitivity of stroke detection to greater than 95% in the first three hours.36 Fluid-attenuated inversion recovery is an MRI protocol that peaks in intensity six to 24 hours after ischemia. When fluid-attenuated inversion recovery is contrasted with concomitant diffusion-weighted imaging, it helps to determine stroke duration, which can guide treatment decisions such as initiating pharmacologic thrombolysis or endovascular therapy. CT perfusion is another modality to help guide consideration for endovascular therapy based on the ratio of infarcted tissue to penumbral tissue in specific vascular distributions.50
Headache. Imaging should be performed in patients with headache and abnormal findings on neurologic examination or new symptoms, or progressive symptoms in those with chronic headaches. Red flag features also require consideration for imaging and include recent head trauma, exertional exacerbation of pain, known or suspected cancer, immunocompromised state, pregnancy, thunderclap onset (i.e., severe and acute, reaches peak intensity in less than one minute, and lasts for more than five minutes), and age older than 50 years.51 Imaging is not required in patients with primary headache disorders (i.e., migraine, tension-type, and trigeminal autonomic cephalalgias) that do not meet the previously mentioned criteria.37,52,53
In emergencies, a noncontrast head CT is sensitive enough to rule out new intracranial hemorrhage, mass effect, or normal-pressure hydrocephalus.37 An MRI with or without contrast is more sensitive for pathologies involving a neoplasm, vascular disease, the posterior fossa, or cervicomedullary lesions and for high and low intracranial pressure disorders. MRI is therefore preferable in evaluating headaches in a nonemergency setting.53
Palpable Neck Anomalies. Imaging assessment of palpable thyroid or parotid lesions (e.g., thyroid nodules) begins with ultrasonography.38,54 For other neck masses, contrast-enhanced studies can demonstrate vascular associations and more associated local anatomy. CT or MRI can also be used in the evaluation of neck masses. A CT scan of the neck with intravenous contrast is usually the preferred first study in adults with neck masses outside of the thyroid or parotid glands because of its increased availability and the risk of malignancy.55 Ultrasonography is the preferred imaging modality in children, regardless of mass location.38
CARDIOPULMONARY IMAGING
Suspected Pulmonary Embolism. A clinical probability assessment tool, such as the Geneva score or Wells score, should be used to determine pretest probability before performing imaging in patients with suspected pulmonary embolism. These tools help stratify patient risk as low (approximate 10% chance of pulmonary embolism), intermediate (30% chance), or high (65% chance).56 The Pulmonary Embolism Rule-out Criteria (https://www.mdcalc.com/perc-rule-pulmonary-embolism) have a 97% sensitivity and greater than 99% negative predictive value for ruling out pulmonary embolism in patients in the emergency department who have a low pretest probability.57 For patients who have low to intermediate risk using the Wells criteria (https://www.mdcalc.com/wells-criteria-pulmonary-embolism) or the Geneva score (https://www.mdcalc.com/geneva-score-revised-pulmonary-embolism [original]; https://www.mdapp.co/geneva-score-for-pulmonary-embolism-calculator-115/ [simplified]), a d-dimer test with an age-adjusted cutoff is recommended.58 CT pulmonary angiography is the preferred test for high-risk patients or those with a positive d-dimer test result. This test has a sensitivity of 83%, specificity of 96%, and positive predictive value of 96% when used with clinical probability algorithms.59,60 A 2019 large prospective trial showed potential for changing d-dimer cutoffs used for proceeding with imaging based on clinical pretest probability, although no updated guidelines have advocated this position.61
Ventilation-perfusion scintigraphy (V/Q scan) is preferable for patients with an eGFR less than 30 mL per minute per 1.73 m2 or a known contrast allergy.56,62,63 A V/Q scan produces a 65 to 250 times lower radiation dose to the breasts/chest than CT angiography and can be considered in young or pregnant patients, although institutional protocols and availability may limit its use.60 Studies suggest V/Q scans have a sensitivity of 77% and specificity of 97% but a high percentage (25%) of inconclusive results.62 In patients who are pregnant, documentation of a deep venous thrombosis on duplex ultrasonography can obviate the need for ionizing radiation.39
Evaluation of Coronary Arteries. For asymptomatic patients at intermediate risk of coronary artery disease (as determined by the Framingham Risk Score or American College of Cardiology/American Heart Association Pooled Cohort Equation), a Multi-Ethnic Study of Atherosclerosis score can be calculated using information from a coronary artery calcium test and routine laboratory tests to further stratify patient risk.64 Despite the U.S. Preventive Services Task Force finding insufficient evidence to recommend coronary artery calcium testing and concerns about harms from the test, nonzero coronary artery calcium scores appear to enhance patient adherence to therapeutic plans for cardiovascular prevention.65–67
For patients with acute or chronic chest pain, the appropriateness of imaging depends on patient risk calculated using one of many risk scores (e.g., HEART [history, electrocardiogram, age, risk factors, troponin], TIMI [thrombolysis in myocardial infarction]). CT coronary angiography, stress echocardiography, and myocardial perfusion scintigraphy (nuclear stress test) are appropriate tests for patients with chest pain who have low to intermediate cardiovascular risk.41,68–70
CT coronary angiography has a more than 95% negative predictive value, which is comparable with myocardial perfusion scintigraphy and stress echocardiography.71 However, CT coronary angiography offers no physiologic data because it involves administration of iodinated contrast only to highlight marked stenosis. Conversely, in myocardial perfusion scintigraphy, a radiotracer is taken up avidly in healthy tissue after stress is induced (i.e., from exercise or a vasodilator). Ischemic tissue takes up only a fixed amount of radiotracer, therefore demonstrating anatomic areas of reduced cardiac function and impaired blood flow. Transthoracic echocardiography demonstrates wall motion abnormalities and potential ischemic complications during episodes of chest pain.41
Plain Chest Radiography. The Choosing Wisely campaign discourages performing plain chest radiography in asymptomatic patients for preoperative evaluations or for baseline testing during hospitalization.42,52,72,73
ABDOMINAL AND PELVIC IMAGING
Right Lower Quadrant Pain. For adults, an abdominal CT scan with intravenous contrast is the preferred imaging technique for acute right lower quadrant pain when appendicitis is suspected.44 Oral contrast does not increase sensitivity or specificity in the evaluation of suspected acute appendicitis.74 Ultrasonography should precede CT in children, and definitive treatment should be initiated if results are positive.52
Right Upper Quadrant Pain. Ultrasonography is the initial imaging choice for suspected biliary disease in patients with right upper quadrant pain. For nondiagnostic ultrasound findings, an abdominal MRI with magnetic resonance cholangiopancreatography is a helpful next step. Magnetic resonance cholangiopancreatography can reveal signs of ductal dilatation and acute and chronic cholecystitis. CT with intravenous contrast does not outperform MRI in the diagnostic assessment of right upper quadrant pain and lacks sensitivity in assessing for stones in the common bile duct, although it can effectively identify other pathologies. Cholescintigraphy (e.g., hepatobiliary iminodiacetic acid scan) is a superior study for acalculous cholecystitis if initial ultrasonography is inconclusive.45
Adnexal Imaging. Transvaginal pelvic ultrasonography is the preferred imaging modality for patients with abnormal vaginal bleeding and pelvic pain (acute and chronic). Follow-up is not needed in premenopausal patients with simple cysts or corpus luteum cysts less than 5 cm in size or in postmenopausal patients with simple cysts or corpus luteum cysts less than 1 cm in size.52
LOW BACK PAIN
Imaging should be avoided in patients with acute (less than six weeks) low back pain and no red flag findings on history or physical examination.47,75,76 If red flags are present or six weeks of conservative therapy (e.g., physical therapy) is ineffective, lumbar spine MRI without contrast should be performed in patients with low back pain.47 Findings on low back imaging are common and are of questionable clinical significance. In one study of asymptomatic adults, MRI showed a herniated disk at one spinal level in 64% of patients and on multiple levels in 38% of patients.76
INCIDENTALOMAS
Abnormal radiographic findings discovered in studies performed for unrelated reasons (e.g., an adrenal mass identified during a CT scan to rule out appendicitis) occur in 20% to 50% of imaging studies.77 These incidentalomas can present a significant challenge in balancing perceived low-risk findings with potential frequent radiographic follow-up.78 The ACR provides clinical guidance for the follow-up of patients with incidentalomas.79
Follow-up is indicated for all suspicious thyroid nodules (evidence of localized invasion or pathologic adenopathy) found incidentally on CT. Nonsuspicious nodules should be further characterized with ultrasonography in patients younger than 35 years if the nodule is 1 cm or larger and in patients 35 years and older if the nodule is 1.5 cm or larger.52 Comprehensive standards for follow-up are organ-based and beyond the scope of this article.
This article updates a previous article on this topic by Crownover and Bepko.80
Data Sources: The following evidence-based medicine resources were searched: Essential Evidence Plus, the Agency for Healthcare Research and Quality evidence reports, Clinical Evidence, the Cochrane database, PubMed, and the Trip database. Key words were diagnostic imaging, radiation risk, ionizing radiation, nephrogenic systemic fibrosis, diffusion-weighted imaging, headache, chest pain, right upper quadrant abdominal pain, right lower quadrant abdominal pain, adnexal masses, suspected stroke, and back pain. Search date: January 15, 2020, and August 28, 2020.
The views expressed in this article are those of the authors and do not reflect the official policy of the U.S. Navy, the U.S. Department of Defense, or the U.S. government.

