Breast cancer is the most common nonskin cancer in women and accounts for 30% of all new cancers in the United States. The highest incidence of breast cancer is in women 70 to 74 years of age. Numerous risk factors are associated with the development of breast cancer. A risk assessment tool can be used to determine individual risk and help guide screening decisions. The U.S. Preventive Services Task Force (USPSTF) and American Academy of Family Physicians (AAFP) recommend against teaching average-risk women to perform breast self-examinations. The USPSTF and AAFP recommend biennial screening mammography for average-risk women 50 to 74 years of age. However, there is no strong evidence supporting a net benefit of mammography screening in average-risk women 40 to 49 years of age; therefore, the USPSTF and AAFP recommend individualized decision-making in these women. For average-risk women 75 years and older, the USPSTF and AAFP conclude that there is insufficient evidence to recommend screening, but the American College of Obstetricians and Gynecologists and the American Cancer Society state that screening may continue depending on the woman's health status and life expectancy. Women at high risk of breast cancer may benefit from mammography starting at 30 years of age or earlier, with supplemental screening such as magnetic resonance imaging. Supplemental ultrasonography in women with dense breasts increases cancer detection but also false-positive results.
Breast cancer is the most common nonskin cancer in women and accounts for 30% of all new cancers in the United States.1 From 2001 to 2016, more than 2.3 million women in the United States were diagnosed with breast cancer.2 The incidence of breast cancer increases after 25 years of age, peaking between 70 and 74 years.2 Approximately one in eight women will develop invasive breast cancer (12.8% lifetime risk).1
WHAT'S NEW ON THIS TOPIC
Breast Cancer Screening
A 2016 meta-analysis calculated that per 10,000 women screened with mammography, three breast cancer deaths are avoided over 10 years in women 40 to 49 years of age, eight deaths are avoided in women 50 to 59 years, 21 deaths are avoided in women 60 to 69 years, and 13 deaths are avoided in women 70 to 74 years. [corrected]
One out of every eight women 40 to 49 years of age who has a screening mammogram will subsequently undergo additional imaging, and for every case of invasive breast cancer detected by screening mammography in this age group, 10 women will have had a biopsy.
In a large, multicenter trial, women with dense breasts and a negative standard mammogram result had two-year screening with MRI or standard mammography. The interval cancer rate was lower in the MRI group than in the mammography group; however, MRI had a high false-positive rate with hundreds of negative breast biopsy results among the 4,738 women who underwent MRI screening.
MRI = magnetic resonance imaging.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
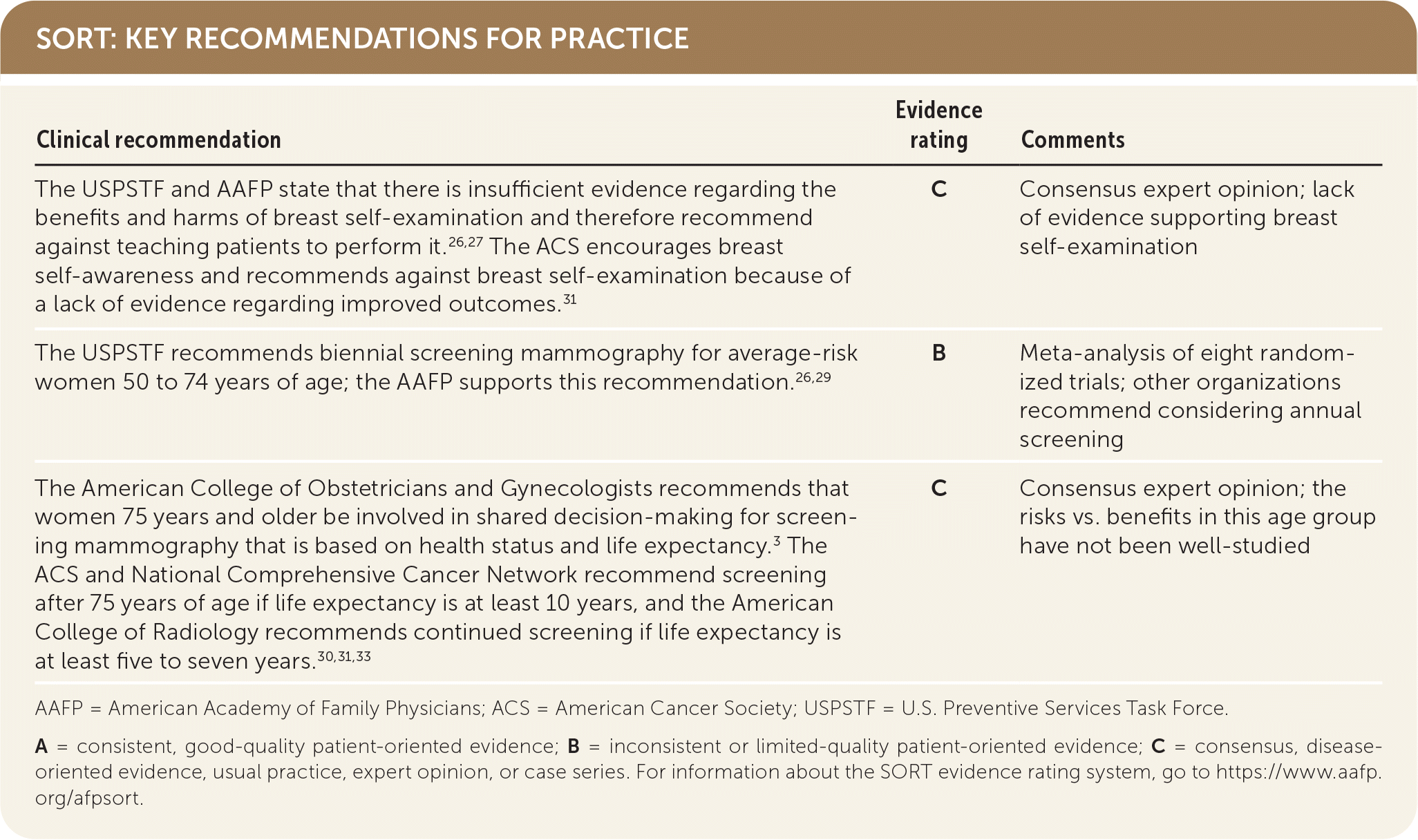
AAFP = American Academy of Family Physicians; ACS = American Cancer Society; USPSTF = U.S. Preventive Services Task Force.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
BEST PRACTICES IN PREVENTIVE MEDICINE

| Recommendation | Sponsoring organization |
|---|---|
| Do not routinely use breast magnetic resonance imaging for breast cancer screening in average-risk women. | Society of Surgical Oncology |
| Do not perform screening mammography in asymptomatic patients with normal examination findings who have less than a five-year life expectancy. | American Society of Breast Surgeons – Benign Breast Disease |
| Do not recommend screening for breast cancer if life expectancy is estimated to be less than 10 years. | Society for Post-Acute and Long-Term Care Medicine |
| Do not recommend screening for breast, colorectal, prostate, or lung cancers without considering life expectancy and the risks of testing, overdiagnosis, and overtreatment. | American Geriatrics Society |
Source: For more information on the Choosing Wisely Campaign, see https://www.choosingwisely.org. For supporting citations and to search Choosing Wisely recommendations relevant to primary care, see https://www.aafp.org/afp/recommendations/search.htm.
The overall mortality rate in U.S. women with breast cancer is about 20 per 100,000. Mortality rates are highest in women 85 years and older (170 per 100,000).2 White women have the highest rate of breast cancer diagnosis, whereas Black women have the highest rate of breast cancer–related death.2 Breast cancer is also the most common cause of cancer-related death in Hispanic women and the second leading cause of cancer-related death behind lung cancer among all women.2
Cancer screening recommendations are determined by the patient's current anatomy. Transgender females with breast tissue and transgender males who have not undergone complete mastectomy should receive screening mammography based on guidelines for cisgender persons (see https://www.aafp.org/afp/2018/1201/p645.html#sec-4).
What Are the Risk Factors for Breast Cancer?
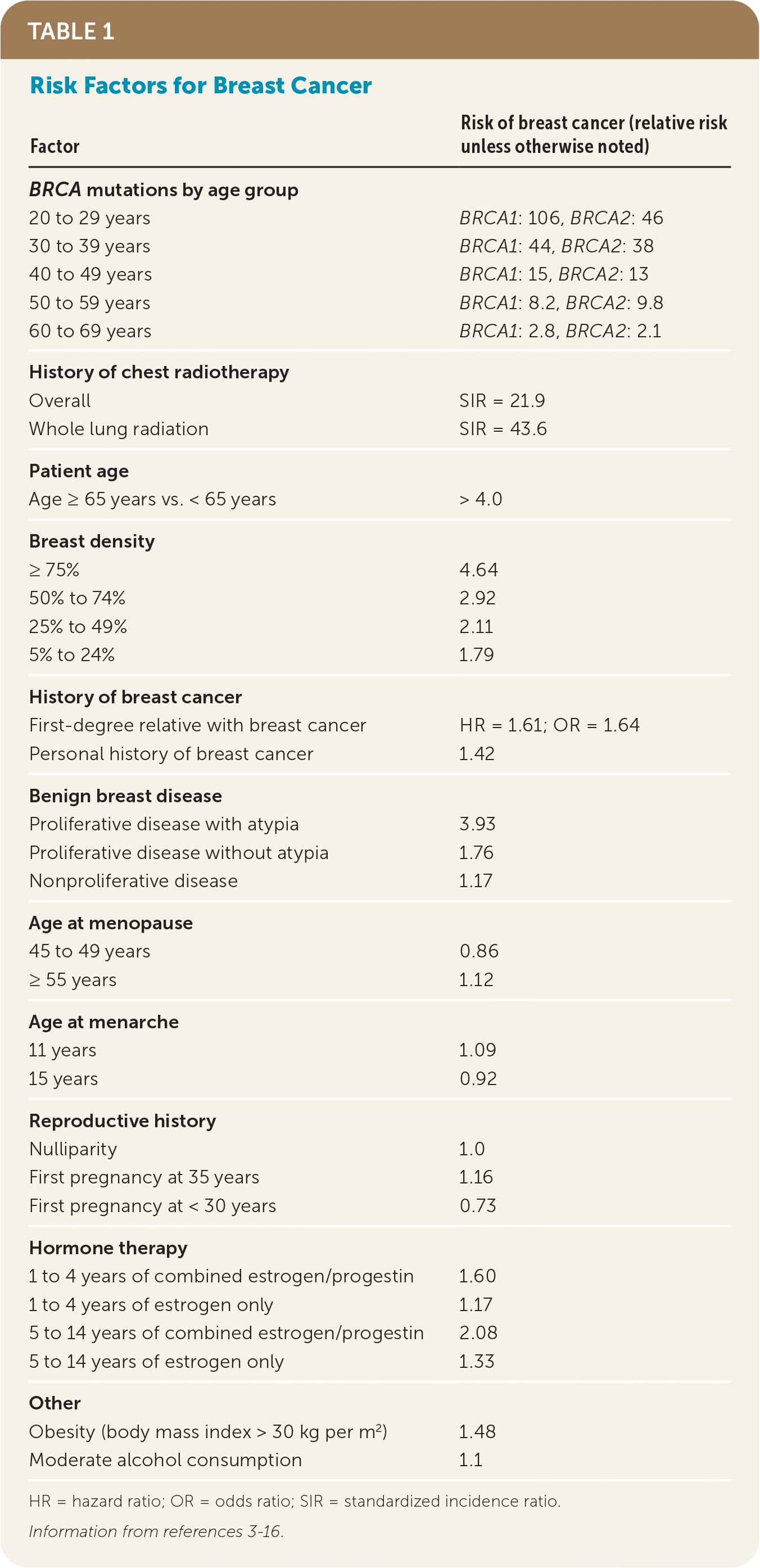
The strongest risk factors are a history of childhood chest radiation, older age, increased breast density, family history of breast cancer, and certain genetic mutations (Table 1).3–16 However, most women who develop invasive breast cancer do not have any of these risk factors.3
TABLE 1. Risk Factors for Breast Cancer
| Factor | Risk of breast cancer (relative risk unless otherwise noted) |
|---|---|
| BRCA mutations by age group | |
| 20 to 29 years | BRCA1: 106, BRCA2: 46 |
| 30 to 39 years | BRCA1: 44, BRCA2: 38 |
| 40 to 49 years | BRCA1: 15, BRCA2: 13 |
| 50 to 59 years | BRCA1: 8.2, BRCA2: 9.8 |
| 60 to 69 years | BRCA1: 2.8, BRCA2: 2.1 |
| History of chest radiotherapy | |
| Overall | SIR = 21.9 |
| Whole lung radiation | SIR = 43.6 |
| Patient age | |
| Age ≥ 65 years vs. < 65 years | > 4.0 |
| Breast density | |
| ≥ 75% | 4.64 |
| 50% to 74% | 2.92 |
| 25% to 49% | 2.11 |
| 5% to 24% | 1.79 |
| < 5% | 1.0 |
| History of breast cancer | |
| First-degree relative with breast cancer | HR = 1.61; OR = 1.64 |
| Personal history of breast cancer | 1.42 |
| Benign breast disease | |
| Proliferative disease with atypia | 3.93 |
| Proliferative disease without atypia | 1.76 |
| Nonproliferative disease | 1.17 |
| Age at menopause | |
| 45 to 49 years | 0.86 |
| ≥ 55 years | 1.12 |
| Age at menarche | |
| 11 years | 1.09 |
| 15 years | 0.92 |
| Reproductive history | |
| Nulliparity | 1.0 |
| First pregnancy at 35 years | 1.16 |
| First pregnancy at < 30 years | 0.73 |
| Hormone therapy | |
| 1 to 4 years of combined estrogen/progestin | 1.60 |
| 1 to 4 years of estrogen only | 1.17 |
| 5 to 14 years of combined estrogen/progestin | 2.08 |
| 5 to 14 years of estrogen only | 1.33 |
| Other | |
| Obesity (body mass index > 30 kg per m2) | 1.48 |
| Moderate alcohol consumption | 1.1 |
HR = hazard ratio; OR = odds ratio; SIR = standardized incidence ratio.
EVIDENCE SUMMARY
A retrospective cohort study demonstrated a standardized incidence ratio (i.e., the ratio of observed to expected number of cases) of 21.9 for breast cancer in women who received chest radiation during childhood.4 Higher doses of radiation were associated with higher risk, and the highest risk was in those who received whole lung radiation (standardized incidence ratio = 43.6). The overall cumulative risk of developing breast cancer by 50 years of age was 30%.4
Increasing age is another strong risk factor. Invasive breast cancer will be diagnosed in one out of 42 women 50 to 59 years of age, and this rate increases to one out of 14 in women 70 years and older.5
Breast density is the amount of glandular and stromal tissue compared with adipose tissue shown on a mammogram. A systematic review and meta-analysis found that compared with women who do not have dense breasts, the relative risk of developing breast cancer is 1.79 for women with breast density between 5% and 24% and 4.64 for those with breast density of 75% or higher.6
Data from the Breast Cancer Surveillance Consortium and the Collaborative Breast Cancer Study showed that having a first-degree relative with breast cancer increases a woman's personal risk by a hazard ratio of 1.61 and odds ratio of 1.64.7 For patients with BRCA mutations, the risk of developing breast cancer by 80 years of age is 60% to 63%, regardless of family history.8
How Can Physicians Estimate the Risk of Developing Breast Cancer?
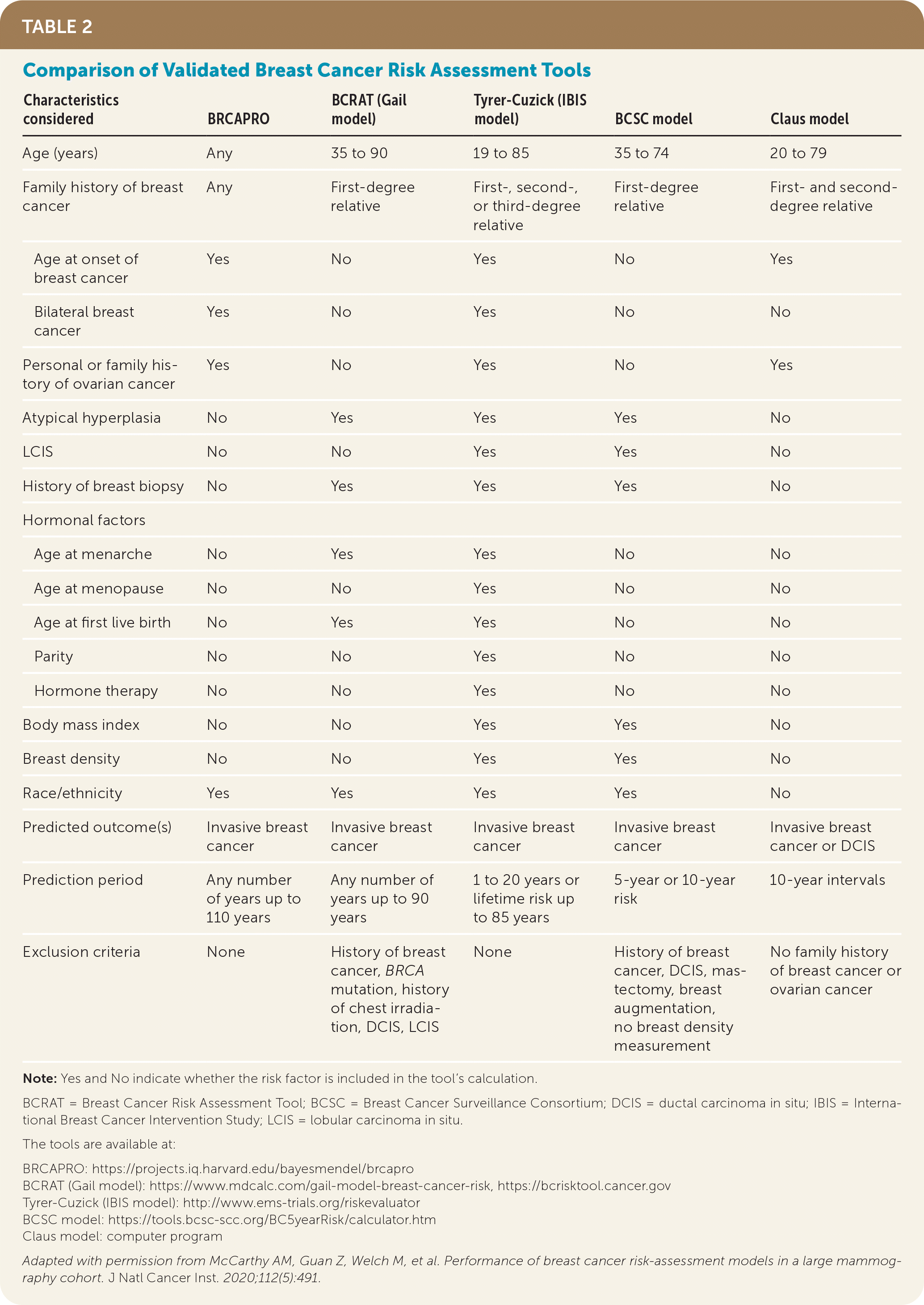
Several validated risk assessment tools are available to stratify breast cancer risk (Table 2).17 These tools can assist physicians and patients in developing individualized plans regarding screening, genetic testing, or chemoprevention.
TABLE 2. Comparison of Validated Breast Cancer Risk Assessment Tools
| Characteristics considered | BRCAPRO | BCRAT (Gail model) | Tyrer-Cuzick (IBIS model) | BCSC model | Claus model |
|---|---|---|---|---|---|
| Age (years) | Any | 35 to 90 | 19 to 85 | 35 to 74 | 20 to 79 |
| Family history of breast cancer | Any | First-degree relative | First-, second-, or third-degree relative | First-degree relative | First- and second-degree relative |
| Age at onset of breast cancer | Yes | No | Yes | No | Yes |
| Bilateral breast cancer | Yes | No | Yes | No | No |
| Personal or family history of ovarian cancer | Yes | No | Yes | No | Yes |
| Atypical hyperplasia | No | Yes | Yes | Yes | No |
| LCIS | No | No | Yes | Yes | No |
| History of breast biopsy | No | Yes | Yes | Yes | No |
| Hormonal factors | |||||
| Age at menarche | No | Yes | Yes | No | No |
| Age at menopause | No | No | Yes | No | No |
| Age at first live birth | No | Yes | Yes | No | No |
| Parity | No | No | Yes | No | No |
| Hormone therapy | No | No | Yes | No | No |
| Body mass index | No | No | Yes | Yes | No |
| Breast density | No | No | Yes | Yes | No |
| Race/ethnicity | Yes | Yes | Yes | Yes | No |
| Predicted outcome(s) | Invasive breast cancer | Invasive breast cancer | Invasive breast cancer | Invasive breast cancer | Invasive breast cancer or DCIS |
| Prediction period | Any number of years up to 110 years | Any number of years up to 90 years | 1 to 20 years or lifetime risk up to 85 years | 5-year or 10-year risk | 10-year intervals |
| Exclusion criteria | None | History of breast cancer, BRCA mutation, history of chest irradiation, DCIS, LCIS | None | History of breast cancer, DCIS, mastectomy, breast augmentation, no breast density measurement | No family history of breast cancer or ovarian cancer |
Note: Yes and No indicate whether the risk factor is included in the tool's calculation.
BCRAT = Breast Cancer Risk Assessment Tool; BCSC = Breast Cancer Surveillance Consortium; DCIS = ductal carcinoma in situ; IBIS = International Breast Cancer Intervention Study; LCIS = lobular carcinoma in situ.
The tools are available at:
BRCAPRO: https://projects.iq.harvard.edu/bayesmendel/brcapro
BCRAT (Gail model): https://www.mdcalc.com/gail-model-breast-cancer-risk, https://bcrisktool.cancer.gov
Tyrer-Cuzick (IBIS model): http://www.ems-trials.org/riskevaluator
BCSC model: https://tools.bcsc-scc.org/BC5yearRisk/calculator.htm
Claus model: computer program
Adapted with permission from McCarthy AM, Guan Z, Welch M, et al. Performance of breast cancer risk-assessment models in a large mammography cohort. J Natl Cancer Inst. 2020;112(5):491.
EVIDENCE SUMMARY
A large retrospective cohort study compared the six-year accuracy of five validated risk assessment tools among 35,921 women 40 to 84 years of age who underwent screening mammography in the United States from 2007 to 2009.17 The models were BRCAPRO (https://projects.iq.harvard.edu/bayesmendel/bayesmendel-r-package); Breast Cancer Risk Assessment Tool, or Gail model (https://bcrisktool.cancer.gov, https://www.mdcalc.com/gail-model-breast-cancer-risk); Tyrer-Cuzick model, or International Breast Cancer Intervention Study model (http://www.ems-trials.org/riskevaluator); Breast Cancer Surveillance Consortium model (https://tools.bcsc-scc.org/BC5yearRisk/calculator.htm); and Claus model (computer program).
Based on overall performance, the positive predictive values were 2.6% for BRCAPRO and the Tyrer-Cuzick model, 2.9% for the Breast Cancer Risk Assessment Tool and Breast Cancer Surveillance Consortium model, and 3.9% for the Claus model. The negative predictive values were high at 97% or more for all of the models.17
Screening mammography reduces breast cancer–related mortality, with larger reductions as women get older.
EVIDENCE SUMMARY
Modeling studies estimate that in women 40 to 49 years of age, the number needed to screen (NNS) with annual mammography to prevent one breast cancer death is 746. The NNS decreases to 351 in women 50 to 59 years and to 233 in women 60 to 69 years. The NNS is 377 in women 70 to 79 years of age.18 However, randomized controlled trials have demonstrated a substantially higher NNS. A meta-analysis performed for the U.S. Preventive Services Task Force (USPSTF) calculated that per 10,000 women screened with mammography, only three breast cancer deaths are avoided over 10 years in women 40 to 49 years of age, eight deaths are avoided in women 50 to 59 years, 21 deaths are avoided in women 60 to 69 years, and 13 deaths are avoided in women 70 to 74 years.19 [corrected]
Between 2008 and 2017, yearly rates of newly diagnosed breast cancer increased by 0.3%, and rates of breast cancer death fell by 1.5%.20 This may be partly attributable to early detection of small, curable breast cancers that have a five-year relative survival rate of 98.8% posttreatment.20 Studies have shown a reduction in the incidence of large tumors, which is also likely because of early detection of smaller tumors by mammography.21
Lower death rates, however, may also reflect improved treatments. With older treatments, the reduction in mortality after screening mammography was approximately 12 deaths per 100,000 women. With improved treatments, the reduction in mortality after screening mammography is now about eight deaths per 100,000 women.21
What Are the Potential Harms of Breast Cancer Screening?
False-positive results are common with screening mammography, especially in younger women, leading to further imaging and radiation exposure and subsequent breast biopsies that can be painful, can cause anxiety, and usually yield benign results. Furthermore, screening can lead to overdiagnosis and overtreatment of cancers that may never have become symptomatic or life-threatening.
EVIDENCE SUMMARY
According to the USPSTF, the false-positive rate of mammography is highest in women 40 to 49 years of age at 121 per 1,000 and decreases with age to 69.6 per 1,000 women 70 to 79 years of age.22 About one of every eight women 40 to 49 years of age who has a screening mammogram will subsequently undergo additional imaging, and for every case of invasive breast cancer detected by screening mammography in this age group, 10 women will have had a biopsy, compared with only three women in their 70s.22
False-positive results are associated with increased antidepressant and anxiolytic prescriptions, with a relative risk of 1.13 to 1.19.23 Women at highest risk of needing antidepressant and anxiolytic therapy are those 40 to 49 years of age who underwent multiple tests, including a biopsy, and who had to wait more than one week to be told the results were false-positive.23
Systematic reviews have found that screening mammography leads to an overdiagnosis rate of 10% to 30%.24,26 [corrected] Overdiagnosis can lead to unnecessary treatments for screening-detected breast cancers. Sometimes this involves treating ductal carcinoma in situ that would have been inconsequential over a woman's lifetime.3 A study based on a large U.S. cancer registry reported that out of 297,000 women 40 years and older who had a mastectomy in 2013, 18% may not have needed one.25 Thus, the USPSTF concludes that there is no strong evidence supporting mammography screening of average-risk women in their 40s.26
What Are the Screening Recommendations for Patients at Average Risk?
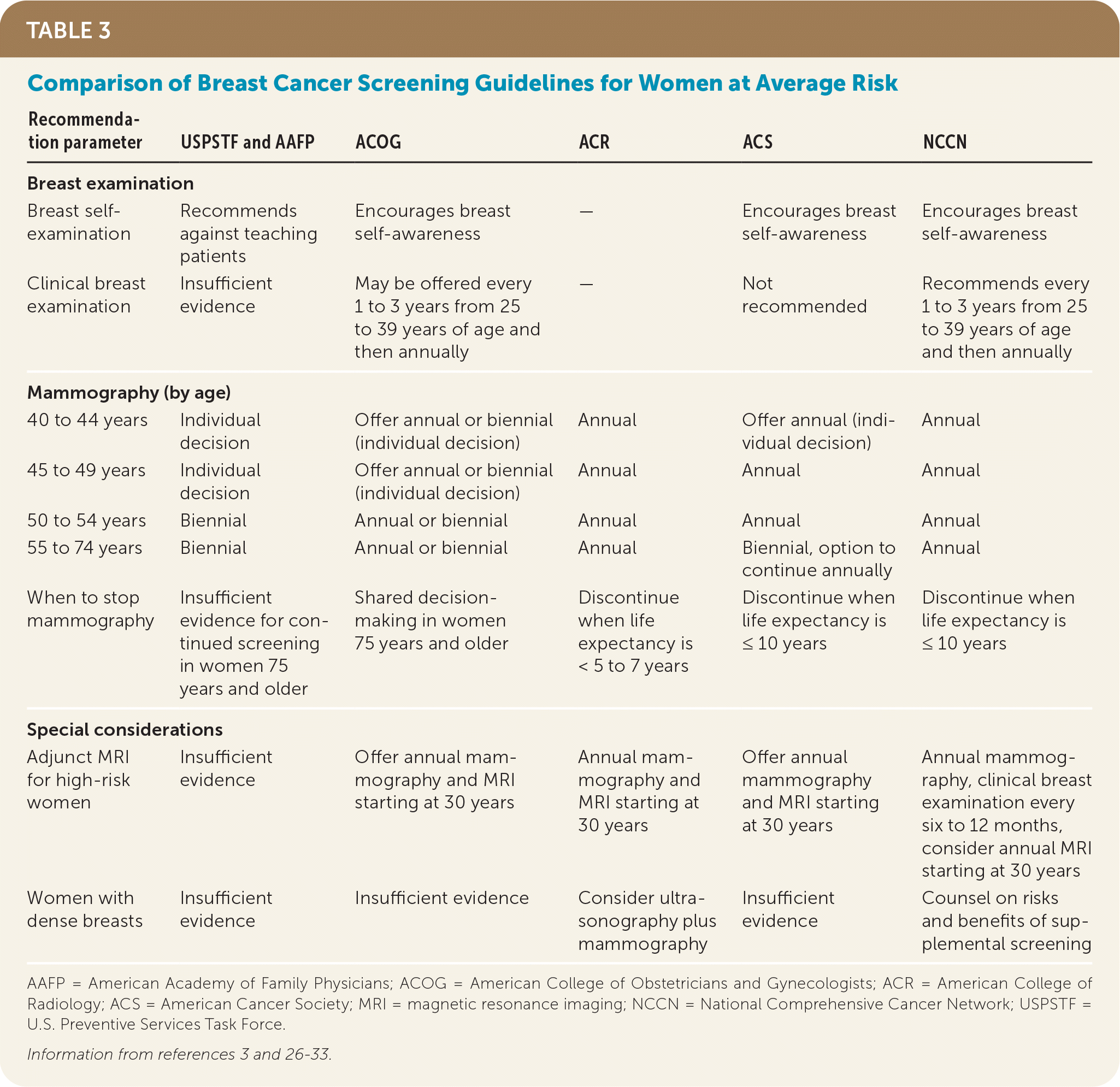
Recommendations for breast self-examinations, clinical breast examinations, and mammography vary among organizations. Table 3 summarizes recommendations from the USPSTF, the American Academy of Family Physicians (AAFP), the American College of Obstetricians and Gynecologists (ACOG), the American College of Radiology (ACR), the American Cancer Society (ACS), and the National Comprehensive Cancer Network (NCCN).3,26–33
TABLE 3. Comparison of Breast Cancer Screening Guidelines for Women at Average Risk
| Recommendation parameter | USPSTF and AAFP | ACOG | ACR | ACS | NCCN |
|---|---|---|---|---|---|
| Breast examination | |||||
| Breast self-examination | Recommends against teaching patients | Encourages breast self-awareness | — | Encourages breast self-awareness | Encourages breast self-awareness |
| Clinical breast examination | Insufficient evidence | May be offered every 1 to 3 years from 25 to 39 years of age and then annually | — | Not recommended | Recommends every 1 to 3 years from 25 to 39 years of age and then annually |
| Mammography (by age) | |||||
| 40 to 44 years | Individual decision | Offer annual or biennial (individual decision) | Annual | Offer annual (individual decision) | Annual |
| 45 to 49 years | Individual decision | Offer annual or biennial (individual decision) | Annual | Annual | Annual |
| 50 to 54 years | Biennial | Annual or biennial | Annual | Annual | Annual |
| 55 to 74 years | Biennial | Annual or biennial | Annual | Biennial, option to continue annually | Annual |
| When to stop mammography | Insufficient evidence for continued screening in women 75 years and older | Shared decision-making in women 75 years and older | Discontinue when life expectancy is < 5 to 7 years | Discontinue when life expectancy is ≤ 10 years | Discontinue when life expectancy is ≤ 10 years |
| Special considerations | |||||
| Adjunct MRI for high-risk women | Insufficient evidence | Offer annual mammography and MRI starting at 30 years | Annual mammography and MRI starting at 30 years | Offer annual mammography and MRI starting at 30 years | Annual mammography, clinical breast examination every six to 12 months, consider annual MRI starting at 30 years |
| Women with dense breasts | Insufficient evidence | Insufficient evidence | Consider ultrasonography plus mammography | Insufficient evidence | Counsel on risks and benefits of supplemental screening |
AAFP = American Academy of Family Physicians; ACOG = American College of Obstetricians and Gynecologists; ACR = American College of Radiology; ACS = American Cancer Society; MRI = magnetic resonance imaging; NCCN = National Comprehensive Cancer Network; USPSTF = U.S. Preventive Services Task Force.
EVIDENCE SUMMARY
Breast Self-Examination. The USPSTF and AAFP recommend against teaching patients to perform breast self-examinations because of a lack of supporting evidence.26,27 ACOG, the NCCN, and the ACS encourage breast self-awareness (i.e., patient familiarity with how her breasts usually feel and look) and advise women to seek medical attention if they notice breast changes.3,31,33 There may be some rationale for breast self-awareness based on the frequency of self-detection cited in some studies. For example, out of 361 breast cancer survivors who participated in the 2003 National Health Interview Survey, 43% reported detecting their own cancers.34
Clinical Breast Examination. The USPSTF and AAFP state that there is insufficient evidence to assess the benefits and harms of clinical breast examinations.26,28 The ACS recommends against these examinations because of insufficient evidence of benefit and a high rate of false-positive results (55 false-positives for every breast cancer detected).31,35 For average-risk women 40 years and older, ACOG says that annual clinical breast examinations may be offered, and the NCCN recommends annual clinical breast examinations.3,33
Mammography. Evidence of benefit varies with a woman's age. The USPSTF found lower mortality rates and a reduced risk of advanced breast cancer in women 50 years and older who had mammography screening (relative risk = 0.62; 95% CI, 0.46 to 0.83) but not in women 39 to 49 years of age (relative risk = 0.98; 95% CI, 0.74 to 1.37).19 The number of breast cancer deaths prevented with screening over 10 years was 12.5 per 10,000 women 50 years and older but only 2.9 per 10,000 women in their 40s.19 Overall, women 50 to 59 years of age have the best balance of risks and benefits from mammography.3,19
ACS data, however, showed improved mortality benefit across all age groups, although the benefit was lower in younger women. The NNS to reduce mortality rates by 20% was 1,770 for women in their 40s, 1,087 for women in their 50s, and 835 for women in their 60s.31
The USPSTF recommends biennial screening mammography for women 50 to 74 years of age.26 This recommendation excludes women 40 to 49 years of age because the number needed to invite (NNI) of 1,904 and the NNS of 1,034 to detect one case of breast cancer with screening mammography were considered too high. The NNI of 1,339 and NNS of 455 in women 50 to 59 years of age and the NNI of 377 and NNS of 233 for women 60 to 69 years of age were considered acceptable.18 The AAFP supports the USPSTF recommendation.29
The ACS recommends annual screening mammography starting at 45 years of age and transitioning to biennial screening at 55 years of age.31 This recommendation is based on multivariable analyses suggesting that women in the younger age group are more likely than older women to have advanced stage cancer when screened biennially rather than annually.31
The NCCN recommends annual screening mammography.33,36 ACOG recommends shared decision-making based on a discussion of benefits and harms when deciding between annual and biennial screening intervals.3
At What Age Should Breast Cancer Screening Be Discontinued?
Women at average risk should continue screening mammography through 74 years of age.3,26,29–31,33 Starting at 75 years of age, women should be involved in shared decision-making based on overall health status and life expectancy according to ACOG recommendations.3 The ACS and NCCN recommend continued screening after 75 years of age if life expectancy is at least 10 years, and the ACR recommends continued screening if life expectancy is at least five to seven years.30,31,33 The USPSTF states that there is insufficient evidence to assess the benefits and harms of screening past 74 years of age, and the AAFP supports this finding.26,29
EVIDENCE SUMMARY
Randomized controlled trials have shown that when mammography screening prevents a death, the death would have occurred within five to seven years after screening; thus, screening women with limited life expectancy is not warranted.36 In addition, the number of life-years gained from screening decreases from 7.8 to 11.4 per 1,000 mammograms at 74 years of age to 4.8 to 7.8 per 1,000 at 80 years and to 1.4 to 2.4 per 1,000 at 90 years.37 When adjusted for quality of life, the number of life-years gained decreases even further, and by 90 to 92 years of age, all life-years gained are counter-balanced by a loss in quality of life, presumably because of treatment adverse effects.37 Yet, despite these data and the corresponding recommendations, 62% of women 75 to 79 years of age and 50% of women 80 years or older get mammograms, and 70% to 86% of physicians recommend mammography for 80-year-old women.38,39
What Are the Screening Recommendations for Patients at Increased Risk?
ACS recommends that women with a 20% or higher lifetime risk of breast cancer (assessed using a risk assessment tool [Table 217]) be offered annual mammography and magnetic resonance imaging (MRI), typically starting at 30 years of age.32 For high-risk women 25 to 29 years of age, ACOG recommends a clinical breast examination every six to 12 months and annual breast MRI with contrast. For patients 30 years and older, ACOG recommends annual mammography and MRI with contrast.40 The NCCN recommends that women with a lifetime risk of more than 20% have breast self-awareness and receive a clinical breast examination every six to 12 months starting at 21 years of age. Annual breast MRI is recommended starting at 25 years of age with annual screening mammography starting at 30 years.33 Women younger than 25 years with a history of chest radiation should have breast self-awareness and receive a clinical breast examination every six to 12 months starting 10 years after radiation therapy. Once these women are 25 years old, annual breast MRI is recommended, then screening mammography starting at 30 years of age.33 The USPSTF states that there is insufficient evidence to assess the benefits and harms of using MRI for breast cancer screening, and the AAFP supports this finding.26,29
EVIDENCE SUMMARY
The evidence for adding annual MRI screening to mammography and clinical breast examinations in women with more than a 20% lifetime risk of breast cancer is based on nonrandomized screening trials and observational studies from the 1990s.32 These studies showed that MRI has a sensitivity of 71% to 100% for detecting breast cancer in high-risk women vs. mammography's sensitivity of 16% to 40% in the same population. However, MRI is less specific (81% to 99%) compared with mammography (93% to more than 99%), resulting in higher rates of false-positives, subsequent medical appointments, and biopsies, with a positive predictive value of 20% to 40%. No data were collected on survival rates with MRI screening or on the optimal MRI screening interval.32
Does Supplemental Imaging Have a Role in Evaluating Dense Breasts?
Almost 50% of women 40 to 74 years of age have dense breasts, which is a risk factor for breast cancer and for false-negative results on standard mammography.41 Ultrasonography, MRI, and digital breast tomosynthesis (also known as 3D mammography) have been proposed as methods to detect breast cancers that might be missed on mammography in women with dense breasts.
EVIDENCE SUMMARY
The ACR recommends considering ultrasonography in addition to screening mammography based on a randomized multicenter trial showing improved cancer detection rates compared with mammography alone (1.9 vs. 4.2 per 1,000).30,42 Ultrasonography may be particularly useful for women who have a 15% to 20% lifetime risk of breast cancer and dense breasts but no additional risk factors.43
Data from the Connecticut Experiments showed an additional 2.3 cancers detected per 1,000 women with dense breasts who were screened with ultrasonography in addition to mammography.43 By the fourth year of the study, the positive predictive value had increased from 7.3% to 20.1%, indicating an improved learning curve for the radiologists regarding which lesions to biopsy. Another study, involving 2,662 women with dense breasts plus one other risk factor for breast cancer, showed that adding ultrasonography to mammography increased the sensitivity of breast cancer detection compared with mammography alone (52% vs. 76%).42
It is important to note, however, that the increased sensitivity comes at the cost of increasing false-positives. An observational cohort study of 6,081 women with varying risk of breast cancer showed that the false-positive rate was 22.2 per 1,000 screens for mammography alone vs. 52 per 1,000 screens for mammography plus ultrasonography (relative risk = 2.23).44
MRI has also been studied as a screening option in women with dense breasts. A large multicenter trial randomized women with dense breasts and a negative result on standard mammography to two-year screening with either MRI or standard mammography.45 The cancer detection rate during the two years was lower in the MRI group than in the mammography group (2.5 vs. 5 per 1,000 screens). More than 90% of MRI-detected cancers, however, were stage 0 or 1, and MRI screening resulted in a high false-positive rate (79.8 per 1,000 screens) with hundreds of negative breast biopsy results among the 4,738 women who underwent MRI screening.
MRI has also been compared with digital breast tomosynthesis. There were higher rates of cancer detection with MRI (11.8 per 1,000 screens) than with digital breast tomosynthesis (4.8 per 1,000 screens), but no data are available on long-term outcomes.46 A study comparing standard mammography with digital breast tomosynthesis is underway.47
The long-term survival of women whose breast cancers were detected with supplemental imaging modalities has not been studied.
This article updates previous articles on this topic by Tirona,48 Knutson and Steiner,49 and Apantaku.50
Data Sources: A PubMed search was completed in Clinical Queries using the key terms breast cancer, breast cancer screening, risk factors for breast cancer, breast cancer risk assessment tools, breast cancer screening recommendations, breast density, mammography, supplemental screening. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Also searched were the Agency for Healthcare Research and Quality Effective Healthcare Reports, the Cochrane database, and Essential Evidence Plus. Search date: April 2020.

