Phosphatidylethanol (PEth) is a whole blood alcohol biomarker. It is detectable for up to four weeks after consuming alcohol.1 A PEth level of 20 ng per mL or higher is often used as the threshold to identify moderate to heavy alcohol consumption.2 The U.S. Preventive Services Task Force recommends using validated questionnaires to screen for unhealthy alcohol use but does not recommend PEth.3 The American Psychiatric Association suggests testing for PEth or other alcohol biomarkers for ongoing monitoring or as part of the initial evaluation in patients with alcohol use disorder.4

| Test | Indication | Population | Cost |
|---|---|---|---|
| PEth blood testing | Identifies moderate to heavy alcohol consumption in the past four weeks* | Age ≥ 18 years | $150 (may vary by region)11 |
PEth = phosphatidylethanol.
*—Moderate consumption is considered 2 to 4 drinks per day (28 to 56 g per day) several times per week or up to 60 g per day for men and 40 g per day for women. Heavy consumption is more than 4 drinks per day several times per week.2
Accuracy
In a small prospective randomized study (n = 44), healthy adults were assigned to either three months of no alcohol use or moderate consumption of red wine.5 Women in the moderate consumption group were allowed 16 g of red wine per day, and men were allowed 33 g per day. In the abstinence group, average PEth levels were 14.1 ng per mL at baseline and less than 3.5 ng per mL after three months (P = .001). PEth values in the moderate consumption group were 12.7 ng at baseline and 15.5 ng after three months (P = .91). The area under the receiver operating characteristic (AUROC) curve for PEth was 0.92 (95% CI, 0.82 to 1.0), demonstrating reliable accuracy for distinguishing between abstention and moderate consumption of alcohol.
Studies have shown good correlation between the Alcohol Use Disorders Identification Test-Consumption (AUDIT-C) and PEth. A large cross-sectional study of adult trauma patients (n = 238) compared AUDIT-C scores with whole blood PEth levels that were drawn within six hours of the trauma incident.6 The AUROC curve was 0.791 (95% CI, 0.722 to 0.860; n = 64) for alcohol misuse and 0.885 (95% CI, 0.830 to 0.939; n = 33) for severe misuse.
Another meta-analysis identified 21 studies (n = 3,085) with positive AUDIT-C scores in women and men (3 or greater and 4 or greater, respectively) and found that 82% had detectable PEth levels (8 ng per mL or higher).7 Patients with a body mass index of 30 kg per m2 or greater had lower odds of detectable PEth levels (odds ratio [OR] = 0.44; 95% CI, 0.28 to 0.69; P < .001; n = 427). Factors that led to a higher probability of detectable PEth levels included normal or high hemoglobin (OR = 2.19; 95% CI, 1.34 to 3.59; P < .002; n = 2,412) and advanced hepatic fibrosis (OR = 1.83; 95% CI, 1.24 to 2.71; P < .002; n = 183). PEth sensitivity was not affected when adjusted for age, ethnicity, HIV status, and method of blood collection (e.g., finger prick or venipuncture). Study findings are summarized in Table 1.5–7
TABLE 1. Summary of Studies Assessing PEth Levels in the Identification of Alcohol Misuse
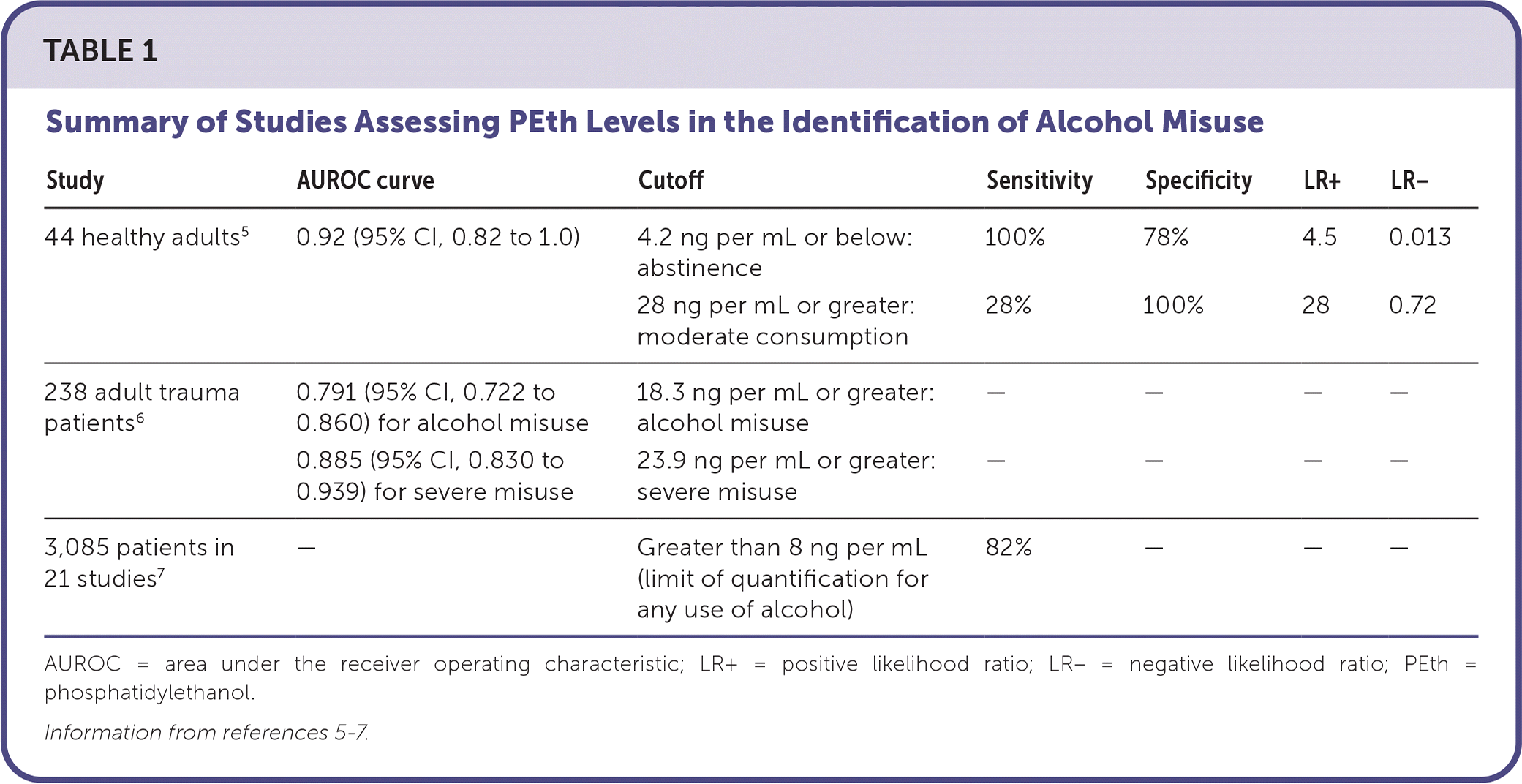
| Study | AUROC curve | Cutoff | Sensitivity | Specificity | LR+ | LR− |
|---|---|---|---|---|---|---|
| 44 healthy adults5 | 0.92 (95% CI, 0.82 to 1.0) | 4.2 ng per mL or below: abstinence 28 ng per mL or greater: moderate consumption | 100% 28% | 78% 100% | 4.5 28 | 0.013 0.72 |
| 238 adult trauma patients6 | 0.791 (95% CI, 0.722 to 0.860) for alcohol misuse 0.885 (95% CI, 0.830 to 0.939) for severe misuse | 18.3 ng per mL or greater: alcohol misuse 23.9 ng per mL or greater: severe misuse | — — | — — | — — | — — |
| 3,085 patients in 21 studies7 | — | Greater than 8 ng per mL (limit of quantification for any use of alcohol) | 82% | — | — | — |
AUROC = area under the receiver operating characteristic; LR+ = positive likelihood ratio; LR− = negative likelihood ratio; PEth = phosphatidyl ethanol.
Benefit
PEth can indicate recent alcohol consumption within the past month.6 Self-reporting tools, such as the AUDIT-C, may be less accurate if impacted by recall bias; PEth testing may improve detection of harmful alcohol use in settings where patients underreport their alcohol consumption.6,8
Harms
PEth levels cannot determine the specific amount or pattern of alcohol consumed.9 PEth testing is best used to detect moderate or heavy alcohol use over a four-week period.1,6,7 Although the correlation between AUDIT-C scores and PEth is moderately strong, PEth cannot distinguish abstinence from light drinking.9 It should not be used to confirm abstinence.
Cost
PEth is measured through whole blood sampling via venipuncture or capillary dried blood spot under Current Procedural Terminology (CPT) code 80321.10 PEth testing costs approximately $150.11 An Advance Beneficiary Notice of Noncoverage may be required to cover the cost of PEth tests for patients with Medicare.12
Bottom Line
PEth testing should be used only when clinical uncertainty remains after employing other standard assessments to detect harmful alcohol consumption over the previous four weeks. Although it is sensitive and specific, the sensitivity may be increased in patients with normal or elevated hemoglobin levels and advanced hepatic fibrosis and decreased in patients with a body mass index of 30 kg per m2 or greater. There are no consensus guidelines for use of PEth testing in clinical settings.13
The views and opinions expressed herein are solely those of the authors and do not necessarily represent the position of the U.S. Air Force, the U.S. Department of Defense, or the U.S. government at large.

