Clinical Question
Does melatonin prevent hospital-acquired delirium?
Evidence-Based Answer
Melatonin should not be used to prevent hospital-acquired delirium because it does not reduce the incidence, duration, or complications of delirium, including increased length of stay or mortality. (Strength of Recommendation [SOR]: A, meta-analysis of randomized controlled trials [RCTs].) Melatonin does not decrease the incidence of intensive care unit (ICU) delirium (SOR: B, conflicting meta-analyses of RCTs) or postoperative delirium (SOR: A, meta-analysis of RCTs). Melatonin is not currently recommended for the prevention of delirium in patients admitted to the hospital. (SOR: C, expert opinion.) However, ramelteon (Rozerem), a melatonin receptor agonist, may lower the incidence of delirium in hospitalized patients. (SOR: A, meta-analysis of RCTs.)
Evidence Summary
A 2021 systematic review and meta-analysis evaluated 14 RCTs (n = 1,712) to determine whether melatonin and melatonin receptor agonists would prevent delirium in hospitalized adults.1 The trials randomized patients (mean age = 34 to 84 years) to melatonin (0.5 to 50 mg per kg from one to 14 days), ramelteon (8 mg per day from one day up to the length of stay), placebo, or usual care. The primary outcome was the incidence of delirium; secondary outcomes included duration of delirium, length of hospitalization, and mortality. Treatment with melatonergics decreased the risk of delirium (relative risk [RR] = 0.61; 95% CI, 0.42 to 0.89; P = .009; I2 = 66%; number needed to treat [NNT] = 13). However, subgroup analysis by treatment drug showed that ramelteon, but not melatonin, lowered the risk of delirium (Table 1).1 Additional subanalyses by care setting found that melatonergics reduced delirium in patients admitted to the ICU but not in surgical (postoperative) or medical patients. Neither of the melatonergics decreased the duration of delirium, length of hospital stay, or mortality. Results in the meta-analysis were limited by high heterogeneity in dose, duration, and formulation of medications; small sample size of included RCTs; and evidence of publication bias.
TABLE 1. Meta-analysis Results of the Effect of Melatonin Receptor Agonists on the Incidence of Delirium in Hospitalized Patients
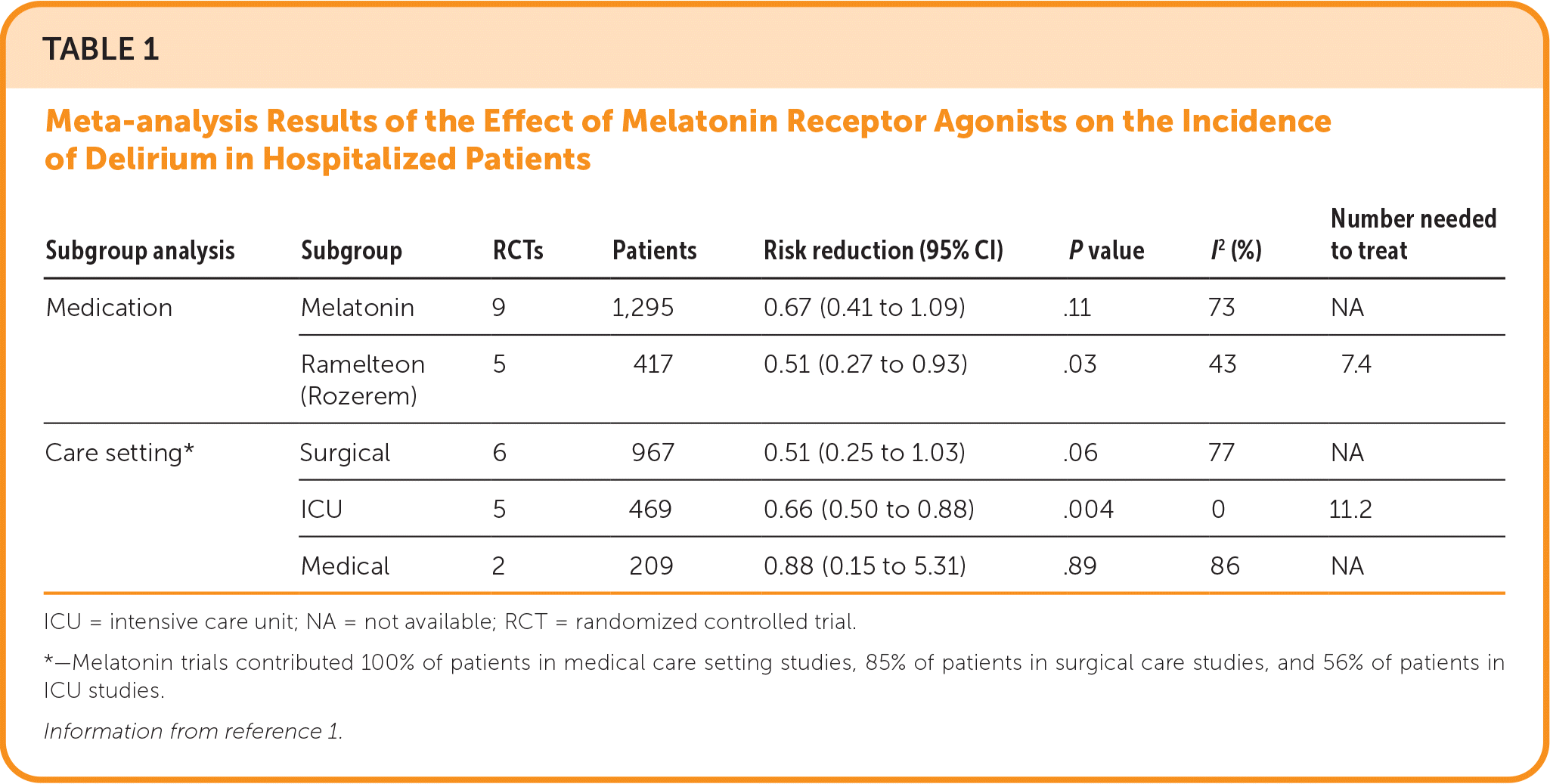
| Subgroup analysis | Subgroup | RCTs | Patients | Risk reduction (95% CI) | P value | I2 (%) | Number needed to treat |
|---|---|---|---|---|---|---|---|
| Medication | Melatonin | 9 | 1,295 | 0.67 (0.41 to 1.09) | .11 | 73 | NA |
| Ramelteon (Rozerem) | 5 | 417 | 0.51 (0.27 to 0.93) | .03 | 43 | 7.4 | |
| Care setting* | Surgical | 6 | 967 | 0.51 (0.25 to 1.03) | .06 | 77 | NA |
| ICU | 5 | 469 | 0.66 (0.50 to 0.88) | .004 | 0 | 11.2 | |
| Medical | 2 | 209 | 0.88 (0.15 to 5.31) | .89 | 86 | NA |
ICU = intensive care unit; NA = not available; RCT = randomized controlled trial.
*—Melatonin trials contributed 100% of patients in medical care setting studies, 85% of patients in surgical care studies, and 56% of patients in ICU studies.
Information from reference 1.
A 2023 systematic review and meta-analysis with nine RCTs (including six RCTs distinct from the previously discussed meta-analysis) evaluated whether melatonin or ramelteon decreased the incidence of delirium in patients in the ICU and found no benefit.2 The trials randomized patients (mean age = 34 to 72 years) to melatonin (1 to 30 mg per day), ramelteon (8 mg per day), or placebo. The primary outcome was the incidence of ICU delirium; secondary outcomes included ICU length of stay, days of mechanical ventilation, and mortality. Treatment with melatonergics did not decrease the incidence of delirium in ICU patients (six trials; n = 1,625; RR = 0.76; 95% CI, 0.54 to 1.07; P = .12; I2 = 64%). Subgroup analysis by treatment drug did not show any individual effect (melatonin: five trials; n = 1,537; RR = 0.82; 95% CI, 0.57 to 1.17; P = .27; I2 = 63%). Neither melatonin nor ramelteon decreased the number of days of mechanical ventilation, the length of stay in the ICU, or mortality. This meta-analysis was limited by the high risk of bias in the primary analysis. The population studied had a relatively younger mean age (seven out of nine RCTs, mean age = younger than 65 years). A sensitivity analysis indicated the results were not robust and the trial was underpowered.
A 2020 systematic review and meta-analysis with six RCTs (n = 913), two cohort studies (n = 582), and one case-control study (n = 65) specifically examined the effect of melatonergics on postoperative delirium.3 The trials compared melatonin (3 to 50 mg per kg) or ramelteon (8 mg per day) with placebo, oxazepam, or other medications (mainly zolpidem or suvorexant [Belsomra]) before surgery. Patients had a mean age of 56 to 84 years and underwent cardiac, orthopedic, pulmonary, or intrathoracic surgeries. The primary outcome was the incidence of delirium three to eight days after surgery. Melatonergics reduced postoperative delirium more than comparison therapies (nine studies; n = 1,536; odds ratio [OR] = 0.45; 95% CI, 0.24 to 0.84; P = .01; I2 = 68%; NNT = 16). Subgroup analysis by treatment drug found a reduction in postoperative delirium with melatonin (seven studies; n = 1,405; OR = 0.52; 95% CI, 0.28 to 0.98; P = .04; I2 = 71%; NNT = 16) and ramelteon (two studies; n = 131; OR = 0.11; 95% CI, 0.02 to 0.65; P = .01; I2 = 0%; NNT = 8). However, subgroup analysis using RCTs alone (i.e., all melatonin trials) found no effect (six trials; n = 905; OR = 0.59; 95% CI, 0.30 to 1.16; P = .13; I2 = 61%), suggesting the treatment effect for melatonin was determined by lower quality studies. Three of these RCTs appeared within the surgery subanalysis of the 2021 review.1 Key weaknesses of the review included high heterogeneity in the dosage of melatonin and timing of administration, types of surgical interventions, types of anesthesia modalities, and the tools used to screen for delirium. Many studies had small sample sizes, increasing the risk of small-study effect bias.
Recommendations From Others
The 2017 European Society of Anaesthesiology guidelines on postoperative delirium reported insufficient data on melatonin and did not provide a formal recommendation on its use as a prophylactic.4 A 2018 Society of Critical Care Medicine clinical practice guideline for the management of patients in the ICU analyzed the utility of sleep-promoting medications, including melatonin, to improve sleep in critically ill adults.5 There were no recommendations on the use of melatonin to improve sleep in critically ill adults due to the lack of high-quality evidence.
Copyright © Family Physicians Inquiries Network. Used with permission.

