| Drug | Dosage | Dose form | Cost* |
|---|---|---|---|
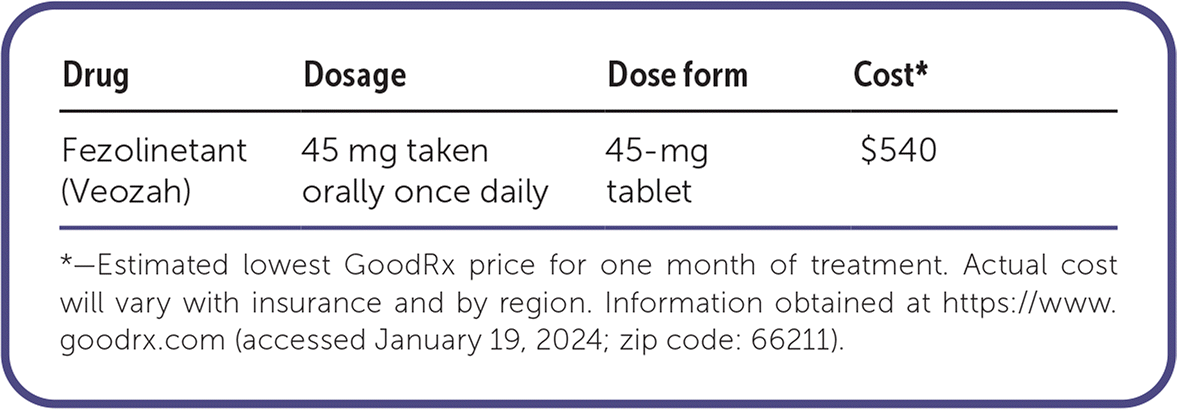
| Fezolinetant (Veozah) | 45 mg taken orally once daily | 45-mg tablet | $540 |
*—Estimated lowest GoodRx price for one month of treatment. Actual cost will vary with insurance and by region. Information obtained at https://www.goodrx.com (accessed January 19, 2024; zip code: 66211).
Fezolinetant (Veozah) is a nonhormonal medication labeled for the treatment of moderate to severe vasomotor symptoms of menopause.1 It is a small-molecule neurokinin 3 receptor antagonist, blocking neurokinin B from binding to thermoregulatory neurons. This prevents a cascade of heat dysregulation.
Safety
The main safety concern with fezolinetant is its effect on liver function. It is contraindicated in people with cirrhosis, severe renal impairment, or end-stage renal disease.1 Fezolinetant may cause early transient liver function test elevations greater than three times the upper limit of normal in about 2% of patients; these elevations resolve with discontinuation. It is unknown whether these liver function test elevations are clinically important. Patients should have baseline liver function testing when starting treatment with fezolinetant, and again at three, six, and nine months. Testing should be repeated if any symptoms of drug-induced liver injury develop. Fezolinetant should be discontinued if transaminase levels are twice the upper limit of normal at baseline or later rise to three times the upper limit of normal.1
Fezolinetant is contraindicated with concomitant use of cytochrome P450 1A2 inhibitors, such as fluvoxamine and cimetidine, because these agents may increase the plasma concentration of fezolinetant.
Malignancy rates in various organs increased with the use of fezolinetant compared with placebo, but there was no correlation to duration of exposure, cancer type, or body system. These increased rates were attributed to a higher number of undiagnosed preexisting malignancies in the treatment group. To date, fezolinetant has not been associated with cardiovascular or breast cancer risk or thromboembolic disease. Fezolinetant has only been studied in women (predominantly White) younger than 65 years, for a maximum period of 52 weeks.
Tolerability
Fezolinetant is well tolerated. Adverse effects include abdominal pain, diarrhea, back pain, hot flashes, and insomnia. Most of these are 1% to 2% more likely in patients taking fezolinetant than those given placebo. In premarketing trials, about 3 out of 4 patients starting treatment with fezolinetant were still taking it one year later.2–4
Effectiveness
Fezolinetant has been studied in three clinical trials involving a total of 1,100 people.2–4 Participants experienced an average of seven vasomotor symptoms per day, ranging from moderate intensity (noticeable) to severe intensity (interrupting an activity).
After 12 weeks of treatment with fezolinetant, symptoms were reduced by 2.3 to 2.6 episodes per day compared with placebo. On a scale of 0 to 6, symptom severity was reduced by 0.2 to 0.3 points compared with placebo.
Fezolinetant has not been compared directly with hormone therapy or nonhormonal treatments such as low-dose paroxetine or venlafaxine. Treatment with estrogen or venlafaxine typically produces a 90% reduction in symptom intensity and frequency within one month of use.5 Fezolinetant does not affect bone density or treat vaginal dryness associated with menopause.
Price
The cost of fezolinetant is about $540 per month, in addition to the cost of liver function monitoring. In contrast, oral estradiol costs about $10 for a 30-day supply.
Simplicity
Fezolinetant should be taken orally once daily, with or without food.
Bottom Line
Fezolinetant is well tolerated; however, it is more expensive and less effective than hormone therapy or available nonhormonal treatments for the relief of vasomotor symptoms associated with menopause.

