“Do the best you can until you know better.
Then, when you know better, do better.” – Maya Angelou
Nearly 80% of women experience perimenopausal and menopausal vasomotor symptoms, with about one-half reporting daily symptoms that can persist for 7 to 10 years.1 Other common manifestations, including genitourinary syndrome of menopause, sleep disturbances, and mood changes, affect 45% to 80% of women.2 These symptoms profoundly impact quality of life, productivity, and health in many women. Given the prevalence of menopausal symptoms, clinicians must be familiar with evidence-based options and engage in shared decision-making to ensure individualized care.
Guidelines from the Menopause Society and National Institute for Health and Care Excellence plainly state that the benefits of systemic hormone therapy for the treatment of vasomotor symptoms outweigh the risks in women younger than 60 years or within 10 years of menopause onset, provided they have no contraindications.2,3 Evaluation of data from the Women's Health Initiative and other studies show that hormone therapy resulted in significant improvements in vasomotor symptoms, genitourinary syndrome of menopause, and fracture risk in appropriately selected patients.4
An article answering common questions about treating menopausal symptoms was published previously in American Family Physician.5 Table 1 summarizes the effects of systemic hormones on chronic disease,5 and Table 2 is a recommended individualized approach to prescribing.4–6 [corrected]
TABLE 2. Recommended Individualized Approach for Menopause Management
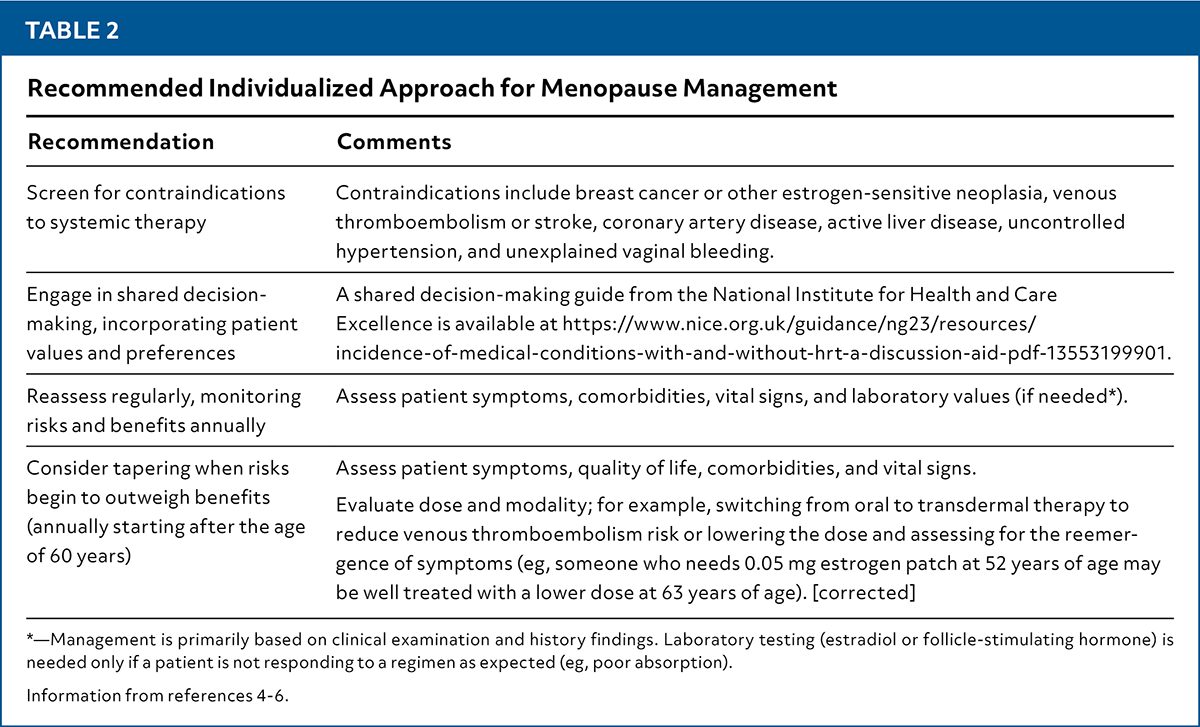
| Recommendation | Comments |
|---|---|
| Screen for contraindications to systemic therapy | Contraindications include breast cancer or other estrogen-sensitive neoplasia, venous thromboembolism or stroke, coronary artery disease, active liver disease, uncontrolled hypertension, and unexplained vaginal bleeding. |
| Engage in shared decision-making, incorporating patient values and preferences | A shared decision-making guide from the National Institute for Health and Care Excellence is available at https://www.nice.org.uk/guidance/ng23/resources/incidence-of-medical-conditions-with-and-without-hrt-a-discussion-aid-pdf-13553199901. |
| Reassess regularly, monitoring risks and benefits annually | Assess patient symptoms, comorbidities, vital signs, and laboratory values (if needed*). |
| Consider tapering when risks begin to outweigh benefits (annually starting after the age of 60 years) | Assess patient symptoms, quality of life, comorbidities, and vital signs. Evaluate dose and modality; for example, switching from oral to transdermal therapy to reduce venous thromboembolism risk or lowering the dose and assessing for the reemergence of symptoms (eg, someone who needs 0.05 mg estrogen patch at 52 years of age may be well treated with a lower dose at 63 years of age). [corrected] |
*—Management is primarily based on clinical examination and history findings. Laboratory testing (estradiol or follicle-stimulating hormone) is needed only if a patient is not responding to a regimen as expected (eg, poor absorption).
For patients with an intact uterus, a progestogen must accompany systemic estrogen to prevent an increased risk of endometrial hyperplasia and uterine carcinoma.2 Contraindications to systemic hormone therapy include history of breast cancer or other estrogen-sensitive neoplasia, venous thromboembolism, stroke, coronary artery disease, active liver disease, uncontrolled hypertension, and unexplained vaginal bleeding. Relative contraindications include migraine with aura, controlled hypertension, hypertriglyceridemia, strong family history of hormone-dependent cancers, and high cardiovascular or venous thromboembolism risk.2 As with everything we do in medicine, a personalized risk-benefit discussion should occur with each patient (eTable A).
eTABLE A Tips for Discussing the Risks and Benefits of Hormone Therapy With Patients
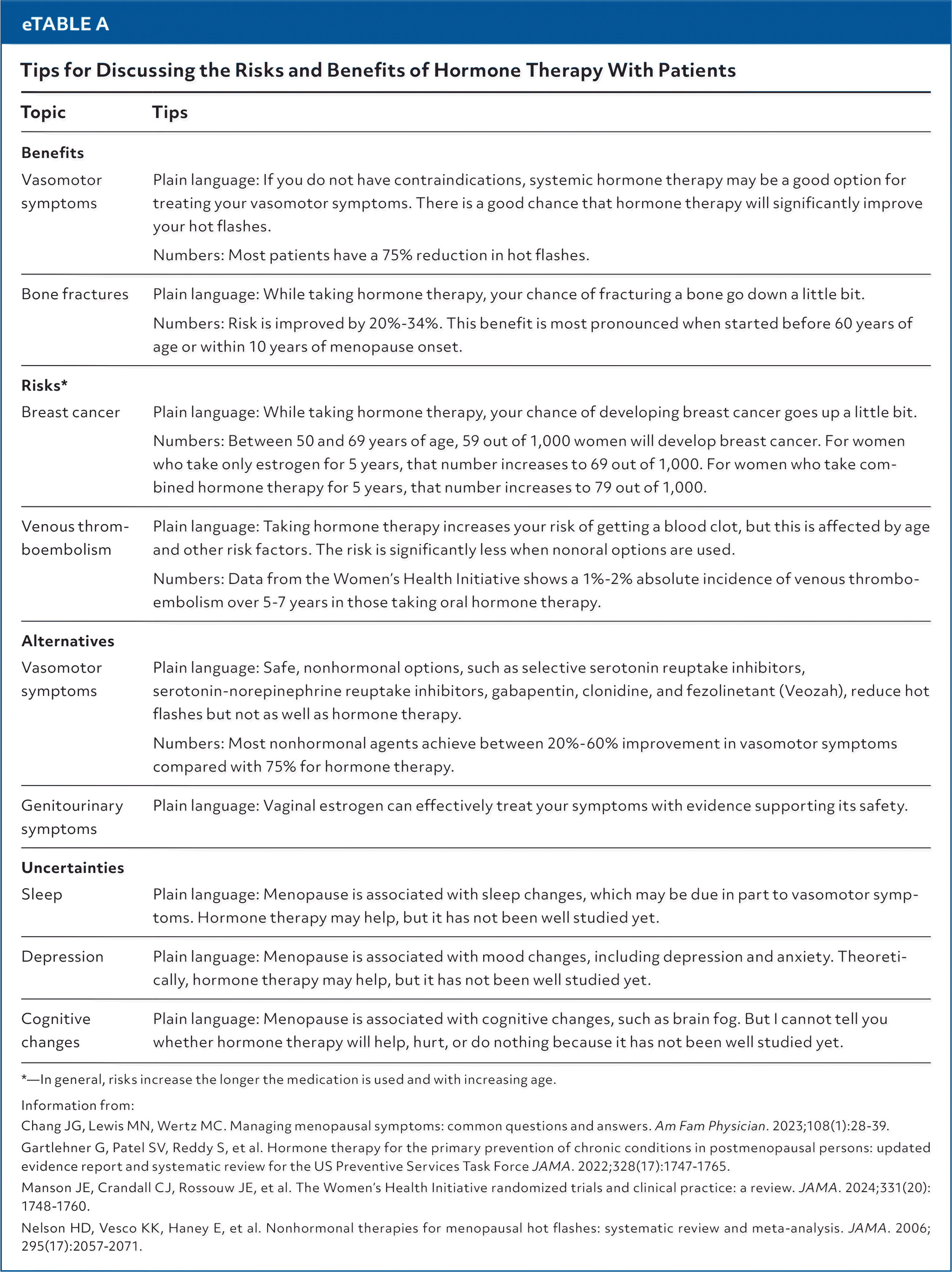
| Topic | Tips |
|---|---|
| Benefits | |
| Vasomotor symptoms | Plain language: If you do not have contraindications, systemic hormone therapy may be a good option for treating your vasomotor symptoms. There is a good chance that hormone therapy will significantly improve your hot flashes. Numbers: Most patients have a 75% reduction in hot flashes. |
| Bone fractures | Plain language: While taking hormone therapy, your chance of fracturing a bone go down a little bit. Numbers: Risk is improved by 20%–34%. This benefit is most pronounced when started before 60 years of age or within 10 years of menopause onset. |
| Risks* | |
| Breast cancer | Plain language: While taking hormone therapy, your chance of developing breast cancer goes up a little bit. Numbers: Between 50 and 69 years of age, 59 out of 1,000 women will develop breast cancer. For women who take only estrogen for 5 years, that number increases to 69 out of 1,000. For women who take combined hormone therapy for 5 years, that number increases to 79 out of 1,000. |
| Venous thromboembolism | Plain language: Taking hormone therapy increases your risk of getting a blood clot, but this is affected by age and other risk factors. The risk is significantly less when nonoral options are used. Numbers: Data from the Women's Health Initiative shows a 1%–2% absolute incidence of venous thromboembolism over 5–7 years in those taking oral hormone therapy. |
| Alternatives | |
| Vasomotor symptoms | Plain language: Safe, nonhormonal options, such as selective serotonin reuptake inhibitors, serotonin-norepinephrine reuptake inhibitors, gabapentin, clonidine, and fezolinetant (Veozah), reduce hot flashes but not as well as hormone therapy. Numbers: Most nonhormonal agents achieve between 20%–60% improvement in vasomotor symptoms compared with 75% for hormone therapy. |
| Genitourinary symptoms | Plain language: Vaginal estrogen can effectively treat your symptoms with evidence supporting its safety. |
| Uncertainties | |
| Sleep | Plain language: Menopause is associated with sleep changes, which may be due in part to vasomotor symptoms. Hormone therapy may help, but it has not been well studied yet. |
| Depression | Plain language: Menopause is associated with mood changes, including depression and anxiety. Theoretically, hormone therapy may help, but it has not been well studied yet. |
| Cognitive changes | Plain language: Menopause is associated with cognitive changes, such as brain fog. But I cannot tell you whether hormone therapy will help, hurt, or do nothing because it has not been well studied yet. |
*—In general, risks increase the longer the medication is used and with increasing age.
Information from:
Chang JG, Lewis MN, Wertz MC. Managing menopausal symptoms: common questions and answers. Am Fam Physician. 2023;108(1):28–39.
Gartlehner G, Patel SV, Reddy S, et al. Hormone therapy for the primary prevention of chronic conditions in postmenopausal persons: updated evidence report and systematic review for the US Preventive Services Task Force JAMA. 2022;328(17):1747–1765.
Manson JE, Crandall CJ, Rossouw JE, et al. The Women's Health Initiative randomized trials and clinical practice: a review. JAMA. 2024;331(20):1748–1760.
Nelson HD, Vesco KK, Haney E, et al. Nonhormonal therapies for menopausal hot flashes: systematic review and meta-analysis. JAMA. 2006;295(17):2057–2071.
It is important to know that since the original 2002 publication of the Women's Health Initiative, newer hormone therapy formulations and routes have demonstrated improved safety profiles. Micronized progesterone appears to be associated with a lower breast cancer risk compared with synthetic progestins.7 Transdermal estrogen has been associated with reduced venous thromboembolism risk compared with oral estrogen.8 Vaginal topical estrogen, effective for genitourinary syndrome of menopause, has minimal systemic absorption and does not increase the risk of breast cancer or cardiovascular disease.9
Family physicians should be prepared to offer patients the best, most effective, safe options. An estimated 1 million to 2.5 million women are using unregulated compounded bioidentical hormones and pellets annually.3 The North American Menopause Society advises against the use of these products because they are not approved by the US Food and Drug Administration and have known risks of inconsistent dosing and purity.10,11 Patients often pursue these products when they feel dismissed, underscoring the value of supportive, evidence-based counseling.
For patients who are not appropriate for or not interested in hormone therapy, well-studied, effective nonhormonal options include selective serotonin reuptake inhibitors, serotoninnorepinephrine reuptake inhibitors, gabapentin, clonidine, and the new class of KNDy (kisspeptin, neurokinin B, and dynorphin) neuron modulators (eg, fezolinetant [Veozah]). Although these medications are less effective than hormone therapy for managing vasomotor symptoms, it is important to know that evidence-based alternatives are available and can be used when appropriate.12
Hormone therapy is not recommended solely for the prevention of chronic conditions such as cardiovascular disease or dementia; however, it is shown to prevent osteoporosis.2,13 It also is not a weight loss product. Including menopause and perimenopause in the differential diagnosis is essential when evaluating new symptoms in women who are 40 to 70 years of age.
Hormone receptors cover the female body, creating diverse symptom experiences across this multidecade period. Although research is not yet robust, shared decision-making around the use of hormone therapy allows expansion of treatment options and could potentially improve outcomes.2,3,14
In November 2025, the US Food and Drug Administration removed the boxed warnings about increased risk of breast cancer, cardiovascular disease, and dementia from topical and systemic estrogen formulations.15 Controversies about the risks, benefits, and uncertainties of hormone therapy continue.
Current evidence demonstrates that when used to treat the common symptoms of menopause, systemic hormone therapy is safe for women younger than 60 years or within 10 years of menopause onset, provided they have no contraindications. Continued funding for women's health research, particularly in perimenopause, is necessary to add to the knowledge base. We also must be clear with patients about what we know and do not know as claims around hormone therapy circulate. As always we must remain curious, current, and compassionate. Doing so ensures that patients receive the best care science has to offer.


