Targeted therapies, which include monoclonal antibodies and small molecule inhibitors, have significantly changed the treatment of cancer over the past 10 years. These drugs are now a component of therapy for many common malignancies, including breast, colorectal, lung, and pancreatic cancers, as well as lymphoma, leukemia, and multiple myeloma. The mechanisms of action and toxicities of targeted therapies differ from those of traditional cytotoxic chemotherapy. Targeted therapies are generally better tolerated than traditional chemotherapy, but they are associated with several adverse effects, such as acneiform rash, cardiac dysfunction, thrombosis, hypertension, and proteinuria. Small molecule inhibitors are metabolized by cytochrome P450 enzymes and are subject to multiple drug interactions. Targeted therapy has raised new questions about the tailoring of cancer treatment to an individual patient's tumor, the assessment of drug effectiveness and toxicity, and the economics of cancer care. As more persons are diagnosed with cancer and as these patients live longer, primary care physicians will increasingly provide care for patients who have received targeted cancer therapy.
For decades, the hallmark of medical treatment for cancer has been intravenous cytotoxic chemotherapy. These drugs target rapidly dividing cells, including cancer cells and certain normal tissues. As a result, many patients experience the classic toxicities of alopecia, gastrointestinal symptoms, and myelosuppression. In the past decade, however, a dramatic shift in cancer therapy has occurred. Although traditional cytotoxic chemotherapy remains the treatment of choice for many malignancies, targeted therapies are now a component of treatment for many types of cancer, including breast, colorectal, lung, and pancreatic cancers, as well as lymphoma, leukemia, and multiple myeloma. Of the new anticancer drugs approved by the U.S. Food and Drug Administration (FDA) since 2000, 15 have been targeted therapies, compared with only five traditional chemotherapeutic agents.1
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| The acneiform rash caused by EGFR inhibitors may be treated with topical or systemic antibiotics, short-term topical steroids, and topical retinoids. | C | 11 |
| Diarrhea caused by EGFR inhibitors is usually self-limited and responds to symptomatic treatment (e.g., loperamide [Imodium]). | C | 12 |
| Antiangiogenic therapy should be interrupted around the time of surgical procedures because of an increased risk of bleeding and wound complications. | C | 20 |
| Patients taking small molecule inhibitors should undergo careful medication review and may require dosage modification if they are taking other medications metabolized by cytochrome P450 enzymes. | C | 32,33 |
EGFR = epidermal growth factor receptor.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see https://www.aafp.org/afpsort.xml.
The two main types of targeted therapy are monoclonal antibodies and small molecule inhibitors. With their distinct mechanisms of action and toxicities, these agents have changed many aspects of the practice of oncology. Targeted therapies have expanded the concept of individually tailored cancer treatment because some of these drugs may be effective in patients whose cancers have a specific molecular target, but they may not be effective in the absence of such a target. This distinction may be influenced by patient ethnicity and sex, as well as by tumor histology.2 In addition, targeted therapies require new approaches to determine optimal dosing, to assess patient adherence to therapy, and to evaluate treatment effectiveness. The cost of these agents, which can exceed several thousand dollars per month,3,4 may become an important issue in health care economics.
As more persons are diagnosed with cancer and as these patients live longer, primary care physicians will increasingly provide care for patients who have received these drugs. Therefore, an understanding of the toxicities and potential drug interactions associated with targeted cancer therapies is important. For instance, new-onset acne in a 70-year-old woman with lung cancer could be a side effect of erlotinib (Tarceva), an oral small molecule inhibitor, but it could also be a surrogate marker of the drug's effectiveness.5,6 If the patient is also taking warfarin (Coumadin), erlotinib could increase her degree of anticoagulation. Given these and other complexities, the purpose of this review is to provide non-oncologists with a basic understanding of the biology, clinical uses, toxicities, and impact of these new cancer therapies. A glossary of oncology terms is provided in Table 1.
Table 1 Glossary of Oncology Terms
| Angiogenesis. The growth of new blood vessels from preexisting vasculature |
| Epidermal growth factor receptor (EGFR, also known as HER1). A tyrosine kinase that, when activated by binding of specific ligands, triggers intracellular signaling that ultimately leads to cell proliferation, invasion, and migration; it is a target of treatment (with the monoclonal antibodies cetuximab [Erbitux] and panitumumab [Vectibix], and the small molecule inhibitors erlotinib [Tarceva], gefitinib [Iressa], and lapatinib [Tykerb]) in multiple tumor types |
| Fragment antigen binding (Fab). The region of an antibody responsible for recognizing and binding to antigens |
| Fragment crystallizable (Fc). The region of an antibody responsible for interacting with immune system components such as natural killer cells and the complement cascade; in some instances, it may be conjugated to a lethal payload such as a radioisotope or toxin |
| HER2/neu. A tyrosine kinase related to epidermal growth factor receptor; it has a role in the pathogenesis of breast cancer and is a target of treatment (with the monoclonal antibody trastuzumab [Herceptin] and the small molecule inhibitor lapatinib [Tykerb]) in the 25 percent of persons with breast cancer in which HER2/neu is overexpressed. Overexpression of HER2/neu is associated with disease recurrence and worse prognosis. HER2 is named because it has similar structure to human epidermal growth factor receptor (HER1); neu is so named because it was derived from a neuroglioblastoma cell line |
| Ligand. A molecule that binds to a specific receptor |
| Monoclonal antibodies. Identical antibodies produced by a single type of immune cell; in targeted cancer therapy, they are directed against molecules unique to, overexpressed in, or mutated in cancer cells |
| Small molecule inhibitors. Drugs that interfere with the function of molecules involved in the development and progression of cancer; most commonly, they interfere with tyrosine kinases |
| Tyrosine kinase. Enzyme that transfers a phosphate group from adenosine triphosphate to a tyrosine amino acid residue in a protein, which may then trigger downstream molecular signaling |
| Vascular endothelial growth factor (VEGF). A signaling protein involved in angiogenesis; it binds to tyrosine kinases (VEGF receptors) to initiate and promote angiogenesis. It is a target of treatment with the monoclonal antibody bevacizumab (Avastin) |
Biology of Targeted Therapy
Traditional cytotoxic chemotherapy works primarily through the inhibition of cell division (Figure 1). In addition to cancer cells, other rapidly dividing cells (e.g., hair, gastrointestinal epithelium, bone marrow) are affected by these drugs. In contrast, targeted therapy blocks the proliferation of cancer cells by interfering with specific molecules required for tumor development and growth (Figure 2). Some of these molecules may be present in normal tissues, but they are often mutated or overexpressed in tumors. Among the earliest targeted therapies were antibodies directed against the cell surface markers cluster of differentiation 20 (CD20), CD33, and CD52, which are present on lymphoma and leukemia cells. Because CD20 is also present on normal lymphoid cells, targeting of this molecule affects overall immune function. This observation has led to the use of the anti-CD20 monoclonal antibody rituximab (Rituxan) for the treatment of autoimmune diseases such as rheumatoid arthritis,7,8 in addition to non-Hodgkin's lymphoma.9
Figure 1.
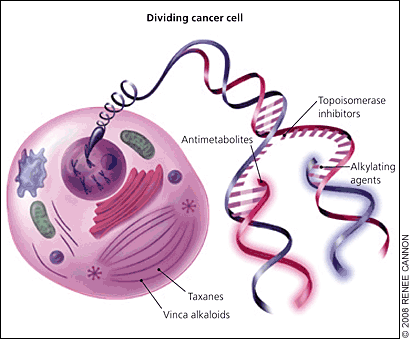
Mechanisms of traditional chemotherapy. These drugs act on rapidly dividing cells, which include normal tissues (e.g., hair, gastrointestinal epithelium, bone marrow) in addition to cancer cells. Alkylating agents interfere with DNA base pairing, leading to strand breaks and arresting DNA replication. Topoisomerase inhibitors prevent DNA uncoiling. Taxanes and vinca alkaloids interfere with micro-tubule function required for cell mitosis. Antimetabolites block the formation and use of nucleic acids essential for DNA replication.
The molecular pathways most often targeted in the treatment of solid tumors (e.g., breast, lung, and colorectal cancers) are those of the epidermal growth factor receptor (EGFR, also known as HER1), vascular endothelial growth factor (VEGF), and HER2/neu (Figure 2). Such pathways can be inhibited at multiple levels: by binding and neutralizing ligands (i.e., molecules that bind to specific receptor sites on cells); by occupying receptor-binding sites (thereby preventing ligand binding); by blocking receptor signaling within the cancer cell; or by interfering with downstream intra-cellular molecules. Monoclonal antibodies, which are usually water soluble and large (typical molecular weight of approximately 150,000 Da), target extracellular components of these pathways, such as ligands and receptor-binding domains. In contrast, small molecule inhibitors (typical molecular weight of approximately 500 Da) can enter cells, thereby blocking receptor signaling and interfering with downstream intracellular molecules.
Figure 2.
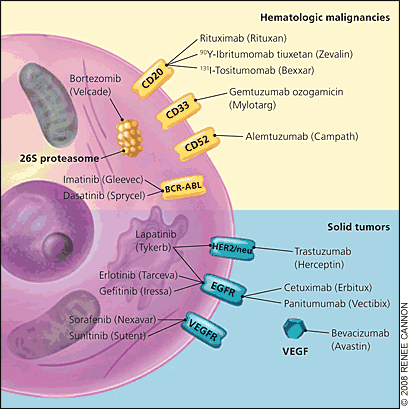
Mechanisms of targeted therapies. The molecular targets in this figure are not overexpressed in a single cell type, but rather on various malignant and normal tissues. For example, CD20 is present on lymphoma and normal lymphoid cells, HER2/neu is present on 25 percent of breast cancer cells, and VEGFR is present on normal and tumor-associated vasculature. Downstream intracellular signaling molecules, some of which are targeted by small molecule inhibitors, are not depicted. Some drugs (e.g., sorafenib [Nexavar], sunitinib [Sutent], imatinib [Gleevec], dasatinib [Sprycel]) have multiple targets, most of which are not depicted. (CD = cluster of differentiation; BCR-ABL = breakpoint cluster region-Abelson; EGFR = epithelial growth factor receptor; VEGFR = vascular endothelial growth factor receptor; VEGF = vascular endothelial growth factor.)
EGFR, which is present in multiple tumor types, contributes to cancer cell proliferation, invasion, and migration.10 Because EGFR is also present in normal epithelial tissue (i.e., skin and mucosa), EGFR inhibition can lead to significant dermatologic (Figure 3) and gastrointestinal toxicities. Of note, in many cases the development of a rash seems to indicate that the treatment may be working.5,6,11 In severe cases, dermatologic toxicity may require discontinuation of the EGFR inhibitor and implementation of measures such as topical or systemic antibiotics, topical retinoids, or topical steroids.11 Additionally, up to 50 percent of patients taking EGFR inhibitors develop diarrhea. For most patients, this toxicity is self-limited and responds to symptomatic treatment, such as loperamide (Imodium).12 Occasionally, severe diarrhea may result in significant volume loss and may require administration of parenteral fluids.
Figure 3.
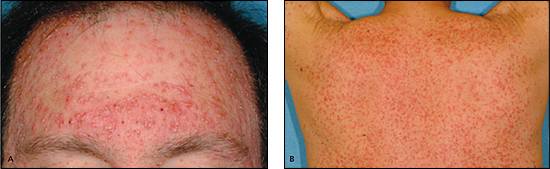
Acneiform rash on (A) the face and (B) back of patients treated with cetuximab (Erbitux), a monoclonal antibody targeting epidermal growth factor receptor.
Targeting of VEGF limits cancer growth by preventing angiogenesis (i.e., the formation of new blood vessels from pre-existing vasculature), a key process in cancer development and progression.13,14 Without new blood vessel formation, tumors cannot grow more than 2 to 3 mm beyond the existing vasculature.15 Targeting of VEGF may also normalize the vasculature within a tumor, thereby improving delivery of other chemotherapeutic agents.16 However, effects on normal blood vessels can also occur, leading to such toxicities as bleeding, thrombosis, hypertension, and—through alterations to glomerular capillaries17—proteinuria. For example, the anti-VEGF monoclonal antibody bevacizumab (Avastin) is approved for treatment of non-small cell lung cancer in patients with adenocarcinoma histology, but not in those with squamous cell tumors. In clinical trials, patients with squamous cell histology had unacceptably high rates of life-threatening hemoptysis.18,19 Likewise, bevacizumab therapy in patients with colorectal cancer should be discontinued for up to eight weeks after surgery because it has been associated with increased rates of postoperative bleeding and wound complications.20
In some instances, targeted therapy has led to truly tailored therapy. Trastuzumab (Herceptin) is a monoclonal antibody directed against HER2/neu, a molecular target related to EGFR that is overexpressed in approximately 25 percent of patients with breast cancer.21 Because trastuzumab is ineffective in the 75 percent of patients with breast cancers that do not overexpress HER2/neu, it is used only if HER2/neu overexpression is documented in tumor tissue.22–24 Similarly, targeting of EGFR in patients with non-small cell lung cancer is most effective against cancers that are highly dependent on the EGFR signaling pathway.25 This trait is most likely to occur in nonsmoking Asian females with bronchioloalveolar-type tumors.2
Such molecular profiling is not new to the field of oncology. For decades, the use of the hormone receptor modulator tamoxifen (Nolvadex, brand no longer available in the United States) has been limited to the two thirds of patients with breast cancer whose tumors express estrogen or progesterone receptors.26 However, treatment effectiveness does not always correlate with molecular biology. The effect of cetuximab (Erbitux), an anti-EGFR monoclonal antibody used in the treatment of colorectal cancer, is independent of the degree of EGFR expression in the tumor.27
Monoclonal Antibodies
In 1986, the FDA approved the first monoclonal antibody, muromonab-CD3 (Orthoclone OKT3), which prevents acute organ rejection after transplantation by blocking T-cell function. Since then, almost 20 other monoclonal antibodies have been approved, about one half of them for the treatment of cancer (Table 2). The fragment antigen binding (Fab) of a monoclonal antibody, which recognizes and binds to antigens, is responsible for the highly specific targeting that is possible with such therapies. Monoclonal antibodies exert their anticancer effects through a variety of mechanisms: by recruiting host immune functions (including natural killer cells and the complement cascade) to attack the target cell; by binding to ligands or receptors, thereby interrupting essential cancer cell processes; or by carrying a lethal payload, such as a radioisotope or toxin, to the target cell (i.e., conjugated monoclonal antibodies).28 Because their protein structure is denatured in the gastrointestinal tract, monoclonal antibodies are administered intravenously. They do not undergo hepatic metabolism, so they are not subject to significant drug interactions.
Table 2 Monoclonal Antibodies for Cancer Treatment
| Drug | Target | Antibody type | FDA-approved indications | Toxicities, side effects, and precautions | Monitoring |
|---|---|---|---|---|---|
| Alemtuzumab (Campath) | CD52 | Humanized, unconjugated | Chronic lymphocytic leukemia | Hematologic toxicity; opportunistic infections; rash | CBC; CD4 counts |
| Live vaccines should be avoided | |||||
| Herpes and Pneumocystis prophylaxis recommended | |||||
| Bevacizumab (Avastin) | VEGF | Humanized, unconjugated | Colorectal cancer, non-small cell lung cancer (nonsquamous) | Gastrointestinal perforation; wound healing complications; hemorrhage; arterial and venous thromboembolism; proteinuria; hypertension | Urinalysis; blood pressure |
| Discontinue use several weeks before elective surgery; do not restart until surgical incision has healed | |||||
| Cetuximab (Erbitux) | EGFR | Chimeric, unconjugated | Colorectal cancer, head and neck cancers | Acneiform rash; diarrhea; hypomagnesemia; nausea and vomiting; interstitial lung disease (rare) | Electrolyte levels; signs of inflammatory and infectious sequelae in patients with dermatologic toxicity; signs of pulmonary toxicity |
| Gemtuzumab Ozogamicin (Mylotarg) | CD33 | Humanized, toxin conjugate (calicheamicin) | Acute myeloid leukemia | Severe myelosuppression; hepatotoxicity | CBC; electrolyte levels; liver chemistries |
| 90Y-Ibritumomab Tiuxetan (Zevalin) | CD20 | Murine, radioisotope conjugate (yttrium-90) | Non-Hodgkin's lymphoma | Severe, prolonged myelosuppression; severe mucocutaneous reactions (e.g., Stevens-Johnson syndrome); risk of secondary malignancies (e.g., acute myeloid leukemia) | CBC; pretreatment antibody titers in patients who have received other murine-based radioimmunotherapy regimens |
| Radiation safety precautions required for one week after administration* | |||||
| Panitumumab (Vectibix) | EGFR | Human, unconjugated | Colorectal cancer | Acneiform rash; diarrhea; hypomagnesemia; hypocalcemia; nausea and vomiting; interstitial lung disease (rare) | Electrolyte levels; signs of inflammatory and infectious sequelae in patients with dermatologic toxicity; signs of ocular toxicity (e.g., conjunctivitis, ocular hyperemia, increased lacrimation, eye or eyelid irritation) |
| Rituximab (Rituxan) | CD20 | Chimeric, unconjugated | Non-Hodgkin's lymphoma, rheumatoid arthritis | Lymphocytopenia; HBV reactivation; severe mucocutaneous reactions (e.g., Stevens-Johnson syndrome) | CBC; signs of active HBV infection or hepatitis in patients who are HBV carriers |
| Live vaccines should be avoided | |||||
| 131I-Tositumomab (Bexxar) | CD20 | Murine, radioisotope conjugate (iodine-131) | Non-Hodgkin's lymphoma | Hypothyroidism; severe, prolonged myelosuppression; nausea and vomiting; secondary malignancies (e.g., acute myeloid leukemia) | CBC; thyroid function tests; pretreatment antibody titers in patients who have received other murine-based radioimmunotherapy regimens |
| Radiation safety precautions required for one week after administration* | |||||
| Trastuzumab (Herceptin) | HER2/neu | Humanized, unconjugated | Breast cancer with HER2/neu overexpression | Cardiomyopathy (especially if coadministered with anthracycline chemotherapy); cytopenias; rash | Electrocardiography; left ventricular ejection fraction |
note:All monoclonal antibodies are administered intravenously. Infusion reactions may occur with all monoclonal antibodies (more often with murine and chimeric antibodies) and are not listed as toxicities.
FDA = U.S. Food and Drug Administration; CD = cluster of differentiation; CBC = complete blood count; VEGF = vascular endothelial growth factor; EGFR = epidermal growth factor receptor; HBV = hepatitis B virus.
*— Radiation precautions include careful disposal of body fluid-contaminated material, condom use for sexual relations, and hand washing.
The design of monoclonal antibodies has changed over the past 20 years as biotechnology has improved. Early drugs in this class were created by immunizing mice with the target antigen. The resulting monoclonal antibodies were composed entirely of mouse proteins, which were potentially highly antigenic to humans, carrying a risk of hypersensitivity reaction during infusion. Patients treated with these early drugs often formed anti-mouse protein antibodies, which could neutralize the effect of the therapeutic antibody. To limit these undesirable effects, recently developed monoclonal antibodies contain an increased proportion of human components and a decreased proportion of murine components; chimeric antibodies are 65 percent human, humanized antibodies are 95 percent human, and human antibodies are 100 percent human.29 The type of antibody can often be identified by the suffix of the drug name: -momab (murine), -ximab (chimeric), -zumab (humanized), or -mumab (human).
Small Molecule Inhibitors
Small molecule inhibitors typically interrupt cellular processes by interfering with the intracellular signaling of tyrosine kinases (i.e., enzymes that transfer phosphate groups from adenosine triphosphate to tyrosine amino acid residues in proteins). Tyrosine kinase signaling initiates a molecular cascade that can lead to cell growth, proliferation, migration, and angiogenesis in normal and malignant tissues. EGFR, HER2/neu, and VEGF receptors are tyrosine kinases.
Small molecule inhibitors (Table 3) differ from monoclonal antibodies in several ways. They are usually administered orally rather than intravenously. They are chemically manufactured, a process that is often much less expensive than the bioengineering required for monoclonal antibodies.30 They achieve less specific targeting than do monoclonal antibodies,31 as evident in the multitargeting nature of the kinase inhibitors imatinib (Gleevec), dasatinib (Sprycel), sorafenib (Nexavar), and sunitinib (Sutent). Unlike monoclonal antibodies, most small molecule inhibitors are metabolized by cytochrome P450 enzymes, which may result in interactions with such medications as macrolide antibiotics, azole antifungals, certain anticonvulsants, protease inhibitors, warfarin, and St. John's wort.32,33 Whereas monoclonal antibodies have half-lives ranging from days to weeks (and are therefore usually administered once every one to four weeks), most small molecule inhibitors have half-lives of only hours and require daily dosing.
Table 3 Small Molecule Inhibitors for Cancer Treatment
| Drug | Target | FDA-approved indications | Toxicities, side effects, and precautions | Monitoring |
|---|---|---|---|---|
| Bortezomib(Velcade) | 26S proteasome | Multiple myeloma, mantle cell lymphoma (a subtype of non-Hodgkin's lymphoma) | Peripheral neuropathy; myelosuppression; rash; constipation; diarrhea; edema; nausea and vomiting | Signs and symptoms of peripheral neuropathy; CBC |
| Dasatinib (Sprycel) | BCR-ABL, SRC family, c-KIT, PDGFR | Chronic myeloid leukemia, acute lymphocytic leukemia | Rash; diarrhea; pleural effusion; fluid retention; mucositis; myelosuppression; QT interval prolongation | CBC; ECG; liver chemistries; weight; signs and symptoms of fluid retention |
| Erlotinib (Tarceva) | EGFR | Non-small cell lung cancer, pancreatic cancer | Acneiform rash; diarrhea; loss of appetite; nausea and vomiting; fatigue; conjunctivitis; elevated liver chemistries | Liver chemistries; signs of inflammatory or infectious sequelae in patients with dermatologic toxicity |
| Gefitinib (Iressa) | EGFR | Non-small cell lung cancer | Acneiform rash; diarrhea; loss of appetite; interstitial lung disease (rare); elevated liver chemistries | Liver chemistries; signs of inflammatory or infectious sequelae in patients with dermatologic toxicity |
| Imatinib (Gleevec) | BCR-ABL, c-KIT, PDGFR | Acute lymphocytic leukemia, chronic myeloid leukemia, gastrointestinal stromal tumor, hypereosinophilic syndrome, systemic mastocytosis | Rash; weight gain; edema; pleural effusion; cardiac toxicity (depression of LVEF); nausea and vomiting; arthralgias and myalgias; myelosuppression | CBC; liver chemistries; weight; signs and symptoms of fluid retention |
| Lapatinib (Tykerb) | HER2/neu, EGFR | Breast cancer with HER2/neu overexpression | Cardiac toxicity (depression of LVEF; QT prolongation); acneiform rash; palmar-plantar erythrodysesthesia (hand-foot syndrome); diarrhea; nausea and vomiting; elevated liver chemistries | LVEF; ECG; electrolyte levels; liver chemistries |
| Sorafenib (Nexavar) | BRAF, VEGFR, EGFR, PDGFR | Renal cell cancer, hepatocellular carcinoma | Hypertension; alopecia; bleeding; rash; palmar-plantar erythrodysesthesia (hand-foot syndrome); hypophosphatemia; diarrhea; nausea and vomiting; elevated amylase and lipase levels; myelosuppression; wound-healing complications | Blood pressure; dermatologic toxicity (including palmar-plantar erythrodysesthesia [hand-foot syndrome]); amylase, lipase, and phosphate levels; CBC |
| Discontinue treatment temporarily for surgical procedures | ||||
| Sunitinib (Sutent) | VEGFR, PDGFR, c-KIT, FLT3 | Renal cell cancer, gastrointestinal stromal tumor | Nausea and vomiting; yellow discoloration of skin; hypothyroidism; depression of LVEF; adrenal function abnormalities; diarrhea; myelosuppression; mucositis; elevated lipase and creatinine levels; elevated liver chemistries; increased uric acid levels | Adrenal function in patients with trauma or severe infection, or in those undergoing surgery; blood pressure; ECG; LVEF; CBC; electrolyte levels (magnesium and potassium); phosphate levels; signs and symptoms of pancreatitis; thyroid function tests |
note:All small molecule inhibitors are administered orally except bortezomib, which is administered intravenously. Most small molecule inhibitors undergo metabolism by cytochrome P450 enzymes and are therefore subject to multiple potential interactions (e.g., with anticonvulsants, azole anti-fungals, dexamethasone, isoniazid [Nydrazid], macrolide antibiotics, nefazodone [Serzone, brand no longer available in the United States], protease inhibitors, rifampin [Rifadin], St. John's wort, verapamil [Calan], and warfarin [Coumadin]).
FDA = U.S. Food and Drug Administration; CBC = complete blood count; BCR-ABL = breakpoint cluster region-Abelson; PDGFR = platelet-derived growth factor receptor; ECG = electrocardiography; EGFR = epidermal growth factor receptor; LVEF = left ventricular ejection fraction; VEGFR = vascular endothelial growth factor receptor.
Imatinib, one of the first small molecule inhibitors, is also one of the most effective. Approved in 2002 for the treatment of chronic myeloid leukemia, imatinib inhibits a continuously active tyrosine kinase that results from the translocation of chromosomes 9 and 22 (the Philadelphia chromosome). Because this molecular abnormality occurs in essentially all patients with chronic myeloid leukemia, imatinib therapy results in a complete hematologic response in 98 percent of patients.34,35 More recently, small molecule inhibitors targeting the EGFR pathway have been used in the treatment of solid tumors, such as non-small cell lung cancer (Figure 4).
Figure 4.
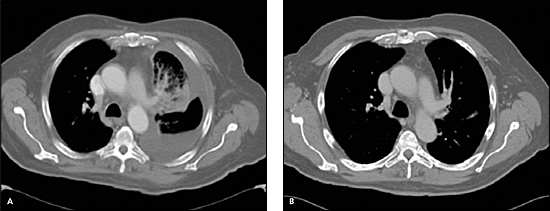
Chest computed tomography scan showing a dramatic response to targeted therapy in a patient with non-small cell lung cancer. (A) Baseline scan showing a left hilar tumor, lymphangitic spread, and a large pleural effusion. (B) Follow-up scan after two months of treatment with erlotinib (Tarceva), an oral small molecule inhibitor targeting epidermal growth factor receptor.
Implications of Targeted Therapy
The use of targeted therapy has markedly changed outcomes for some diseases. Imatinib has had a dramatic effect on chronic myeloid leukemia, and rituximab, sunitinib, and trastuzumab have revolutionized the treatment of non-Hodgkin's lymphoma, renal cell carcinoma, and breast cancer, respectively.23,36,37 In other instances, the degree of clinical benefit is more modest. In patients with advanced pancreatic cancer, the addition of erlotinib to standard chemotherapy increases the one-year survival rate from 17 to 24 percent, which correlates to an increase in median survival from 24 to 27 weeks.38
In addition to prolonging survival in patients with certain cancers, targeted therapies provide treatment options for some patients who may not otherwise be candidates for anticancer therapy. For instance, non-small cell lung cancer and non-Hodgkin's lymphoma primarily affect elderly patients, many of whom have medical comorbidities that limit the use of standard chemotherapy. Targeted therapies such as erlotinib and rituximab are often less toxic and better tolerated than traditional chemotherapy, offering these patients additional treatment options.
DOSING AND EFFECTIVENESS
Targeted therapy has introduced several new issues for oncologists. Determining optimal dosing is one challenge. Clinical trials of traditional chemotherapeutic drugs generally determine toxicity through the degree of myelosuppression. Targeted therapies, however, often do not cause significant hematologic toxicity. Assessment of treatment effectiveness also may require a paradigm shift. When traditional chemotherapy is effective, reduction in tumor volume is anticipated on serial radiographic studies. In contrast, some targeted therapies may impart a clinical benefit by stabilizing tumors, rather than shrinking them.
To determine the dosing and effectiveness of targeted therapies, cancer researchers increasingly are turning to pharmacodynamic end points, such as tumor metabolic activity on positron emission tomography scans, levels of circulating tumor and endothelial cells, and serial levels of target molecules in tumor tissue.39–41 These studies add complexity and cost to clinical research. In addition, repeat biopsies of tumor tissue may be inconvenient for patients and unacceptable to institutional review boards. Although these studies may initially increase the time and expense of therapy, they may improve its long-term cost-effectiveness by identifying the subset of patients most likely to benefit from specific drugs.
ADHERENCE
Because traditional chemotherapy is usually administered intravenously in an observed infusion area, patient adherence to treatment regimens is readily assessed. Delays and omissions in chemotherapy doses, whether they are the result of patient preference or treatment-related toxicities, are immediately recognized and documented. In contrast, most small molecule inhibitors are taken at home on a long-term daily basis. Thus, the task of assessing patient adherence more closely resembles that encountered with therapies for chronic diseases such as diabetes or hypertension. The few studies performed to date show that patient adherence to oral cancer treatment regimens is highly variable and somewhat unpredictable.42
COST
Targeted therapy also introduces new economic considerations. Substituting oral small molecule inhibitors for traditional chemotherapy eliminates some treatment costs, including those associated with vascular access and intravenous infusions. However, targeted therapy is often used in addition to, rather than in place of, traditional chemotherapy. If targeted therapy includes monoclonal antibodies, costs can escalate exponentially. For example, multidrug colorectal cancer treatment regimens containing bevacizumab or cetuximab cost up to $30,790 for eight weeks of treatment, compared with $63 for an eight-week regimen of fluorouracil (Adrucil) and leucovorin, the standard treatment until the mid-1990s.3,43,44

