Am Fam Physician. 2024;109(3):277-278
Author disclosure: No relevant financial relationships.

| Test | Indication | Population | Cost* |
|---|---|---|---|
| DermTech Pigmented Lesion Assay | A noninvasive test to guide biopsy decisions for atypical, pigmented lesions meeting at least one ABCDE criteria for melanoma | Adults 18 years and older with Fitzpatrick skin types I through III | $760 |
The DermTech Pigmented Lesion Assay (PLA) is a noninvasive test used for the detection of melanoma. Adhesive stickers are used to collect skin cells from the entire surface of suspicious melanocytic lesions that have one or more ABCDE criteria for melanoma. These stickers are analyzed for genomic markers associated with melanoma: RNA (LINC00518 and PRAME) and optional add-on DNA (TERT). When results are positive for one or more genomic markers, a biopsy is recommended. If results are negative for genomic markers, biopsy deferral and continued monitoring of the lesion are recommended.1 The PLA has not been approved by the U.S. Food and Drug Administration.1
Accuracy
An initial validation study including 398 samples compared results from the PLA with a consensus histopathologic assessment from three dermatopathologists. Among the 398 samples (87 melanomas and 311 nonmelanomas), the PLA demonstrated a sensitivity of 91%, specificity of 69%, positive likelihood ratio of 2.94, and negative likelihood ratio of 0.13.2
For diagnostic accuracy, a pooled analysis from a systematic review of five studies (n = 2,043) comparing PLA with histopathologic examination found a sensitivity of 86.9%, specificity of 82.4%, positive likelihood ratio of 4.94, and negative likelihood ratio of 0.16.3 However, the quality of the included studies was low due to the high risk of bias and inconsistencies in study design, possibly decreasing the reliability of sensitivity and specificity and overestimating accuracy.3
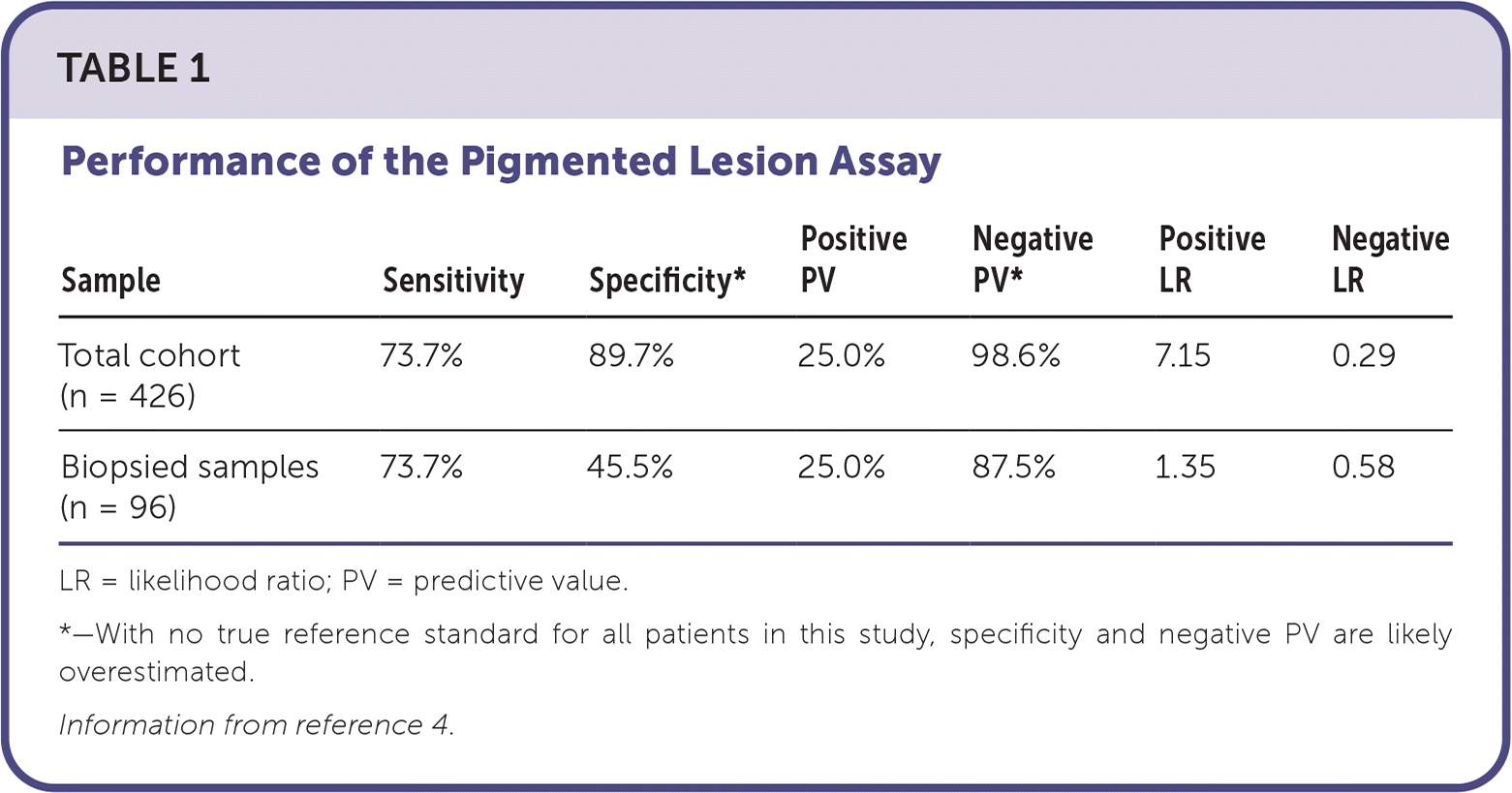
A retrospective cohort study assessed the performance of PLA testing during routine clinical use in dermatology clinics. There was no reference standard for all patients. The study included 426 pigmented lesions, 96 of which were biopsied based on clinical concern. Of the total lesions, 370 were negative on PLA testing, and 56 were positive. Of the 40 PLA-negative lesions biopsied due to high clinical concern, five (12.5%) were diagnosed as melanoma, and 16 (40%) were diagnosed as atypical melanocytic nevi. The remaining PLA-negative lesions were managed with surveillance, and none exhibited concerning clinical changes after four to six months. Of the 56 PLA-positive lesions, 14 (25%) were diagnosed as melanoma. The number needed to biopsy to detect one melanoma was 5 (19 out of 96), with a biopsy ratio of 4.1 benign lesions for each melanoma detected. For the total cohort (n = 426), PLA-negative results without a biopsy were treated as true negatives (due to low clinical concern). For the biopsied cohort, true negatives were determined by a histologic diagnosis of nonmelanoma (Table 1).4
Benefits
The PLA allows for noninvasive testing of the entire surface of a melanocytic lesion. It reduces the number of biopsies needed to diagnose one melanoma from 20 (reported range of 8 to 30) to between 2.7 and 5.1.4–6 The PLA is useful for testing lesions occurring in cosmetically sensitive areas. It can also be beneficial in patients who are not good candidates for biopsy or who develop hypertrophic scars.1
Harms
The PLA should not be used for testing lesions that are clearly malignant.1 It is not validated in children or in adults with Fitzpatrick skin types IV through VI (light-brown skin that rarely burns to brown and black skin that never burns and tans easily). The PLA is not recommended in patients with psoriasis, eczema, ulcerations, bleeding, or scarring. Testing on the palms, soles, nails, mucous membranes, or areas covered with hair that cannot be trimmed should be avoided. Lesions must be a minimum of 5 mm in diameter, and multiple kits should be used for lesions larger than 16 mm. About 85% of adhesive patch samples yield sufficient RNA for analysis.2,4 The manufacturer requires clinicians to complete training before administering the PLA. It can take up to five days to receive PLA results, and patients may need to return to the clinic if a biopsy is needed.
Cost
Medicare pays $760 for the DermTech Melanoma Test (RNA biomarkers only).1 Most patients pay less than $75 out-of-pocket for the PLA.1 Medicare does not cover the optional DNA TERT add-on assay. Medicaid and some commercial insurance plans do not cover the test, but the manufacturer offers financial assistance to patients.1
An economic ana lysis based on consensus treatment guidelines and fee schedules from the Centers for Medicare and Medicaid Services estimated a cost reduction of $447 per assessed pigmented lesion using PLA compared with the current histopathologic standard of care. This was primarily due to the increased sensitivity and specificity of PLA relative to the standard of care, leading to fewer biopsies, surgical excisions, and missed melanomas.6
Bottom Line
The PLA is an objective, noninvasive tool that decreases the need to biopsy melanocytic lesions in some skin regions. However, its accuracy requires improvement. The PLA is not validated in children or in adults with Fitzpatrick skin types IV through VI. Use of the PLA has not been studied in the primary care setting, and additional validation is needed before widespread implementation.