Clinical Question
How should abnormal test results be managed in patients who have had cervical cancer screening?
Evidence Summary
Cervical cancer incidence and mortality have decreased significantly in the United States since the introduction of cervical cancer screening.1 In 2019, the American Society for Colposcopy and Cervical Pathology (ASCCP) published updated consensus guidelines for the management of abnormal cervical cancer screening results in asymptomatic patients. These guidelines provided recommendations that attempted to maximize cervical cancer prevention and limit harms from overtesting and overtreatment.2
The ASCCP guidelines help determine the risk of developing cervical intraepithelial neoplasia grade 3 or worse (CIN3+), including CIN3, adenocarcinoma in situ, and invasive cervical cancer, and recommend management based on the guiding principle that equal risk should receive equal management. Risk estimates were based on a prospective longitudinal cohort of more than 1.5 million patients followed for more than 10 years at Kaiser Permanente–Northern California3 and were validated in several databases that represent patients of diverse racial, ethnic, and socioeconomic backgrounds.4–9
The recommendations can be implemented in five common clinical scenarios: (1) initial management of an abnormal cervical cancer screening result; (2) return visit for surveillance of a previous abnormal cervical cancer screening result that did not require colposcopy; (3) evaluation of a colposcopic biopsy result; (4) management of test results at a surveillance visit after a colposcopic biopsy that shows less than CIN grade 2 (CIN2); and (5) follow-up after treatment for CIN2 or CIN3.2
Individualized risk of developing CIN3+ can be determined by combining the patient's age, current screening results (from primary human papillomavirus [HPV] screening, cotesting with HPV and cervical cytology, or cervical cytology alone), prior screening and biopsy results, and history of precancer treatment.2 In most cases, a persistent HPV infection is necessary for developing cervical precancer or cancer; therefore, HPV type and duration of infection strongly impact the risk for CIN3+.10–13 In an observational cohort of 990,013 women in the Kaiser Permanente–Northern California health care system, a negative HPV test or co-test in the past five years has been shown to correlate with a 50% decrease in the risk of CIN3+.14 Incorporating prior screening and biopsy results allows for the detection and treatment of CIN3+ and avoids unnecessary interventions for patients who are at lower risk.
When a patient's risk has been identified, appropriate management can be determined by referencing the clinical action thresholds that correspond to specific risk levels. Colposcopy and treatment recommendations are based on the immediate risk of CIN3+, whereas longer-term surveillance recommendations are based on the five-year risk of CIN3+. Risk estimates and management plans can be determined using the ASCCP decision aid, available via a smartphone application (ASCCP Management Guidelines; $9.99 in the Apple App Store and Google Play) and at no cost on their website (https://app.asccp.org).2 Figure 1 provides management recommendations based on immediate and five-year risk of CIN3+.
FIGURE 1. Management recommendations based on immediate and five-year risk of CIN grade 3 or worse.
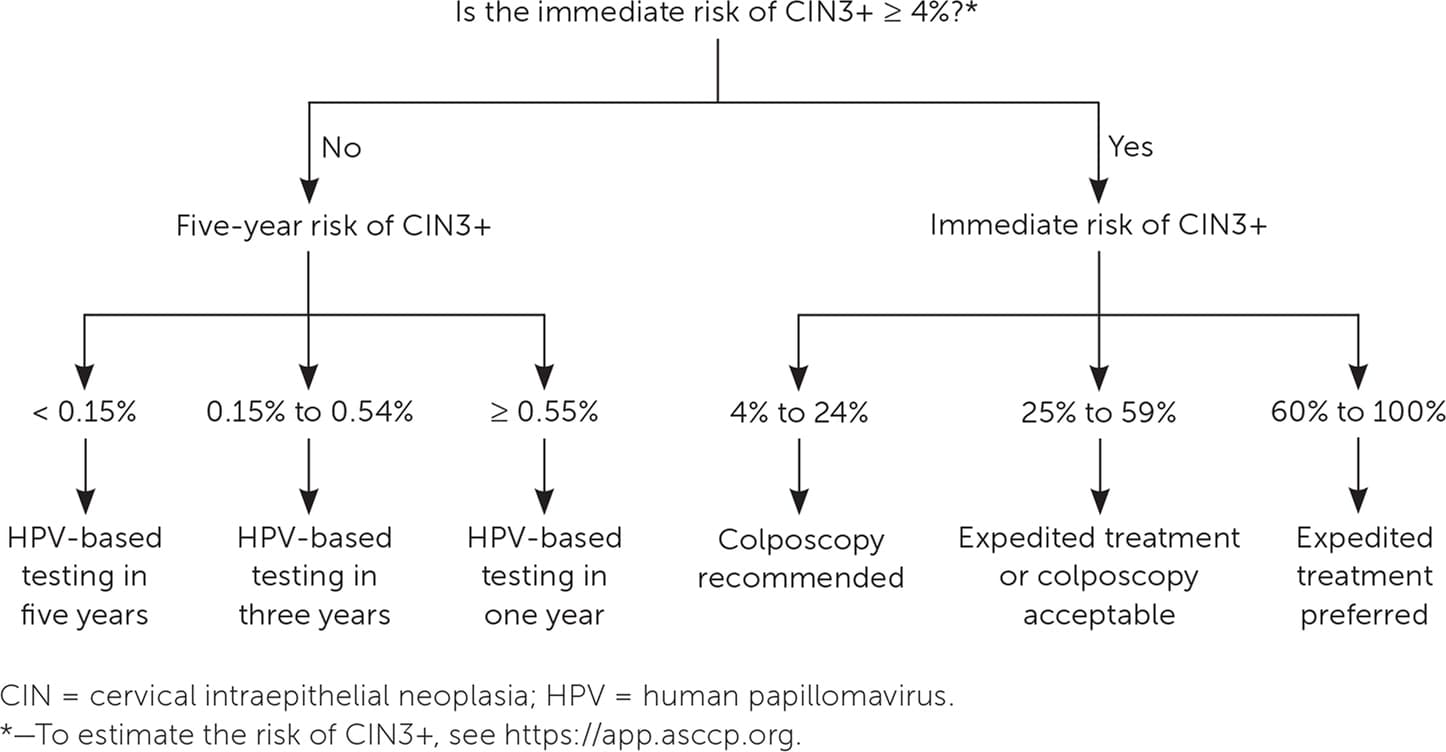
There are specific considerations for patients who are pregnant, immunosuppressed, posthysterectomy, and younger than 25 years; therefore, it is important to include these patient characteristics when determining risk. There are also special guidelines for atypical glandular cells, adenocarcinoma in situ, atypical squamous cells–cannot rule out high-grade squamous intraepithelial lesions, HPV-16 and HPV-18 with normal cytology, and two consecutive unsatisfactory cervical cancer screening tests. Management of these cases does not follow the risk algorithm outlined in Figure 1 because of limited data or a disproportionately high risk of cancer.2
Applying the Evidence
A 30-year-old woman with no prior cervical cancer screening has a co-test result that is positive for HPV and atypical squamous cells of undetermined significance.Using the ASCCP decision aid, you enter herage, current test result, and lack of prior testing. It determines that shehas an immediate risk of CIN3+ of 4.4%. Becauseher immediate risk is 4% or greater, colposcopy is recommended. Her colposcopic biopsy result isCIN grade 1, and repeat HPV screening is recommended in one year. At follow-up, her cotestresult is still positive forHPV and atypical squamous cells of undeter-mined significance. You include her prior screening and current testresults using the decision aid and determine that her immediate risk of CIN3+ is 3.1%, and her five-year risk of CIN3+ is 6.0%. Because her immediate risk is below the clinical action threshold of 4%, she does not need to repeat colposcopy. It is recommended that HPV screening be repeated in one year based on her five-year risk of CIN3+.

