Serum protein electrophoresis is used to identify patients with multiple myeloma and other serum protein disorders. Electrophoresis separates proteins based on their physical properties, and the subsets of these proteins are used in interpreting the results. Plasma protein levels display reasonably predictable changes in response to acute inflammation, malignancy, trauma, necrosis, infarction, burns, and chemical injury. A homogeneous spike-like peak in a focal region of the gamma-globulin zone indicates a monoclonal gammopathy. Monoclonal gammopathies are associated with a clonal process that is malignant or potentially malignant, including multiple myeloma, Waldenström’s macroglobulinemia, solitary plasmacytoma, smoldering multiple myeloma, monoclonal gammopathy of undetermined significance, plasma cell leukemia, heavy chain disease, and amyloidosis. The quantity of M protein, the results of bone marrow biopsy, and other characteristics can help differentiate multiple myeloma from the other causes of monoclonal gammopathy. In contrast, polyclonal gammopathies may be caused by any reactive or inflammatory process.
Serum protein electrophoresis is a laboratory examination that commonly is used to identify patients with multiple myeloma and other disorders of serum protein. Many subspecialists include serum protein electrophoresis screening in the initial evaluation for numerous clinical conditions. Sometimes, however, the results of this examination can be confusing or difficult to interpret.
This article provides a comprehensive review of serum protein electrophoresis, including a discussion of how the examination is performed, what it measures, and when it is indicated. The article also provides a simple guide to result interpretation and suggestions on follow-up of abnormal results.
Definitions
Electrophoresis is a method of separating proteins based on their physical properties. Serum is placed on a specific medium, and a charge is applied. The net charge (positive or negative) and the size and shape of the protein commonly are used in differentiating various serum proteins.1
Several subsets of serum protein electrophoresis are available. The names of these subsets are based on the method that is used to separate and differentiate the various serum components. In zone electrophoresis, for example, different protein subtypes are placed in separate physical locations on a gel made from agar, cellulose, or other plant material.2,3 The proteins are stained, and their densities are calculated electronically to provide graphical data on the absolute and relative amounts of the various proteins. Further separation of protein subtypes is achieved by staining with an immunologically active agent, which results in immunofluorescence and immunofixation.
Components of Serum Protein Electrophoresis
The pattern of serum protein electrophoresis results depends on the fractions of two major types of protein: albumin and globulins. Albumin, the major protein component of serum, is produced by the liver under normal physiologic conditions. Globulins comprise a much smaller fraction of the total serum protein content. The subsets of these proteins and their relative quantity are the primary focus of the interpretation of serum protein electrophoresis.1,3
Albumin, the largest peak, lies closest to the positive electrode. The next five components (globulins) are labeled alpha1, alpha2, beta1, beta2, and gamma. The peaks for these components lie toward the negative electrode, with the gamma peak being closest to that electrode. Figure 1 shows a typical normal pattern for the distribution of proteins as determined by serum protein electrophoresis.
Figure 1
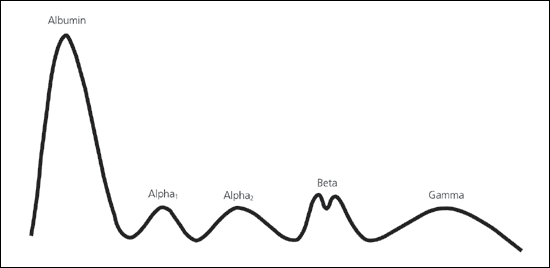
Typical normal pattern for serum protein electrophoresis.
ALBUMIN
The albumin band represents the largest protein component of human serum. The albumin level is decreased under circumstances in which there is less production of the protein by the liver or in which there is increased loss or degradation of this protein. Malnutrition, significant liver disease, renal loss (e.g., in nephrotic syndrome), hormone therapy, and pregnancy may account for a low albumin level. Burns also may result in a low albumin level. Levels of albumin are increased in patients with a relative reduction in serum water (e.g., dehydration).
ALPHA FRACTION
Moving toward the negative portion of the gel (i.e., the negative electrode), the next peaks involve the alpha1 and alpha2 components. The alpha1-protein fraction is comprised of alpha1-antitrypsin, thyroid-binding globulin, and transcortin. Malignancy and acute inflammation (resulting from acute-phase reactants) can increase the alpha1-protein band. A decreased alpha1-protein band may occur because of alpha1-antitrypsin deficiency or decreased production of the globulin as a result of liver disease. Ceruloplasmin, alpha2-macroglobulin, and haptoglobin contribute to the alpha2-protein band. The alpha2 component is increased as an acute-phase reactant.
BETA FRACTION
The beta fraction has two peaks labeled beta1 and beta2. Beta1 is composed mostly of transferrin, and beta2 contains beta-lipoprotein. IgA, IgM, and sometimes IgG, along with complement proteins, also can be identified in the beta fraction.
GAMMA FRACTION
Much of the clinical interest is focused on the gamma region of the serum protein spectrum because immunoglobulins migrate to this region. It should be noted that immunoglobulins often can be found throughout the electrophoretic spectrum. C-reactive protein (CRP) is located in the area between the beta and gamma components.1
Indications
Serum protein electrophoresis commonly is performed when multiple myeloma is suspected. The examination also should be considered in other “red flag” situations (Table 1).2–4
TABLE 1 Indications for Serum Protein Electrophoresis
| Suspected multiple myeloma, Waldenström’s macroglobulinemia, primary amyloidosis, or related disorder |
| Unexplained peripheral neuropathy (not attributed to longstanding diabetes mellitus, toxin exposure, chemotherapy, etc.) |
| New-onset anemia associated with renal failure or insufficiency and bone pain |
| Back pain in which multiple myeloma is suspected |
| Hypercalcemia attributed to possible malignancy (e.g., associated weight loss, fatigue, bone pain, abnormal bleeding) |
| Rouleaux formations noted on peripheral blood smear |
| Renal insufficiency with associated serum protein elevation |
| Unexplained pathologic fracture or lytic lesion identified on radiograph |
| Bence Jones proteinuria |
If the examination is normal but multiple myeloma, Waldenström’s macroglobulinemia, primary amyloidosis, or a related disorder still is suspected, immunofixation also should be performed because this technique may be more sensitive in identifying a small monoclonal (M) protein.5
Interpretation of Results
Plasma protein levels display reasonably predictable changes in response to acute inflammation, malignancy, trauma, necrosis, infarction, burns, and chemical injury. This so-called “acute-reaction protein pattern” involves increases in fibrinogen, alpha1-antitrypsin, haptoglobin, ceruloplasmin, CRP, the C3 portion of complement, and alpha1 acid glycoprotein. Often, there are associated decreases in the albumin and transferrin levels.6 Table 26 lists characteristic patterns of acute-reaction proteins found on serum protein electrophoresis, along with associated conditions or disorders.
TABLE 2 Characteristic Patterns of Acute-Reaction Proteins Found on Serum Protein Electrophoresis and Associated Conditions or Disorders
| Increased albumin Dehydration Decreased albumin Chronic cachectic or wasting diseases Chronic infections Hemorrhage, burns, or protein-losing enteropathies Impaired liver function resulting from decreased synthesis of albumin Malnutrition Nephrotic syndrome Pregnancy Increased alpha1 globulins Pregnancy Decreased alpha1 globulins Alpha1-antitrypsin deficiency Increased alpha2 globulins Adrenal insufficiency Adrenocorticosteroid therapy Advanced diabetes mellitus Nephrotic syndrome Decreased alpha2 globulins Malnutrition Megaloblastic anemia Protein-losing enteropathies Severe liver disease Wilson’s disease | Increased beta1 or beta2 globulins Biliary cirrhosis Carcinoma (sometimes) Cushing’s disease Diabetes mellitus (some cases) Hypothyroidism Iron deficiency anemia Malignant hypertension Nephrosis Polyarteritis nodosa Obstructive jaundice Third-trimester pregnancy Decreased beta1 or beta2 globulins Protein malnutrition Increased gamma globulins Amyloidosis Chronic infections (granulomatous diseases) Chronic lymphocytic leukemia Cirrhosis Hodgkin’s disease Malignant lymphoma Multiple myeloma Rheumatoid and collagen diseases (connective tissue disorders) Waldenström’s macroglobulinemia Decreased gamma globulins Agammaglobulinemia Hypogammaglobulinemia |
Information from reference 6.
In the interpretation of serum protein electrophoresis, most attention focuses on the gamma region, which is composed predominantly of antibodies of the IgG type. The gamma-globulin zone is decreased in hypogammaglobulinemia and agammaglobulinemia. Diseases that produce an increase in the gamma-globulin level include Hodgkin’s disease, malignant lymphoma, chronic lymphocytic leukemia, granulomatous diseases, connective tissue diseases, liver diseases, multiple myeloma, Waldenström’s macroglobulinemia, and amyloidosis.3,7
Although many conditions can cause an increase in the gamma region, several disease states cause a homogeneous spike-like peak in a focal region of the gamma-globulin zone (Figure 2). These so-called “monoclonal gammopathies” constitute a group of disorders that are characterized by proliferation of a single clone of plasma cells that produce a homogeneous M protein.6
Figure 2
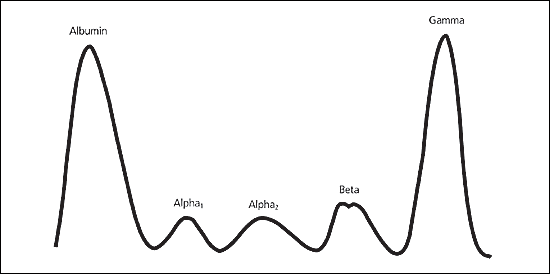
Abnormal serum protein electrophoresis pattern in a patient with multiple myeloma. Note the large spike in the gamma region.
Monoclonal Versus Polyclonal Gammopathies
It is extremely important to differentiate monoclonal from polyclonal gammopathies. Monoclonal gammopathies are associated with a clonal process that is malignant or potentially malignant. In contrast, polyclonal gammopathies may be caused by any reactive or inflammatory process, and they usually are associated with nonmalignant conditions. The most common conditions in the differential diagnosis of polyclonal gammopathy are listed in Table 3.8,9
TABLE 3 Differential Diagnosis of Polyclonal Gammopathy
| Infections | Malignancies |
|---|---|
| Viral infections, especially hepatitis, human immunodeficiency virus infection, mononucleosis, and varicella Focal or systemic bacterial infections, including endocarditis, osteomyelitis, and bacteremia Tuberculosis Connective tissue diseases Systemic lupus erythematosus Mixed connective tissue Temporal arteritis Rheumatoid arthritis Sarcoid Liver diseases Cirrhosis Ethanol abuse Autoimmune hepatitis Viral-induced hepatitis Primary biliary cirrhosis Primary sclerosing cholangitis | Solid tumors Ovarian tumors Lung cancer Hepatocellular cancer Renal tumors Gastric tumors Hematologic cancers (see below) Hematologic and lymphoproliferative disorders Lymphoma Leukemia Thalassemia Sickle cell anemia Other inflammatory conditions Gastrointestinal conditions, including ulcerative colitis and Crohn’s disease Pulmonary disorders, including bronchiectasis, cystic fibrosis, chronic bronchitis, and pneumonitis Endocrine diseases, including Graves’ disease and hashimoto’s thyroiditis |
An M protein is characterized by the presence of a sharp, well-defined band with a single heavy chain and a similar band with a kappa or lambda light chain. A polyclonal gammopathy is characterized by a broad diffuse band with one or more heavy chains and kappa and lambda light chains.7
Once a monoclonal gammopathy is identified by serum protein electrophoresis, multiple myeloma must be differentiated from other causes of this type of gammopathy. Among these other causes are Waldenström’s macroglobulinemia, solitary plasmacytoma, smoldering multiple myeloma, monoclonal gammopathy of undetermined significance, plasma cell leukemia, heavy chain disease, and amyloidosis.4,7
The quantity of M protein can help differentiate multiple myeloma from monoclonal gammopathy of undetermined significance. Definitive diagnosis of multiple myeloma requires 10 to 15 percent plasma cell involvement as determined by bone marrow biopsy. Characteristic differentiating features of the monoclonal gammopathies are listed in Table 4.7
TABLE 4 Characteristic Features of Monoclonal Gammopathies
| Disease | Distinctive features |
|---|---|
| Multiple myeloma | M protein appears as a narrow spike in the gamma, beta, or alpha2 regions. |
| M-protein level is usually greater than 3 g per dL. | |
| Skeletal lesions (e.g., lytic lesions, diffuse osteopenia, vertebral compression fractures) are present in 80 percent of patients. | |
| Diagnosis requires 10 to 15 percent plasma cell involvement on bone marrow biopsy. | |
| Anemia, pancytopenia, hypercalcemia, and renal disease may be present. | |
| Monoclonal gammopathy of undetermined significance | M-protein level is less than 3 g per dL. |
| There is less than 10 percent plasma cell involvement on bone marrow biopsy. | |
| Affected patients have no M protein in their urine, no lytic bone lesions, no anemia, no hypercalcemia, and no renal disease. | |
| Smoldering multiple myeloma | M-protein level is greater than 3 g per dL. |
| There is greater than 10 percent plasma cell involvement on bone marrow biopsy. | |
| Affected patients have no lytic bone lesions, no anemia, no hypercalcemia, and no renal disease. | |
| Plasma cell leukemia | Peripheral blood contains more than 20 percent plasma cells. |
| M-protein levels are low | |
| Affected patients have few bone lesions and few hematologic disturbances. | |
| This monoclonal gammopathy occurs in younger patients. | |
| Solitary plasmacytoma | Affected patients have only one tumor, with no other bone lesions and no urine or serum abnormalities. |
| Waldenström’s macroglobulinemia | IgM M protein is present. |
| Affected patients have hyperviscosity and hypercellular bone marrow with extensive infiltration by lymphoplasma cells. | |
| Heavy chain disease | The M protein has an incomplete heavy chain and no light chain. |
Adapted with permission from George ED, Sadovsky R. Multiple myeloma: recognition and management. Am Fam Physician 1999;59:1889.
In some patients with a plasma cell dyscrasia, serum protein electrophoresis may be normal because the complete monoclonal immunoglobulin is absent or is present at a very low level.7 In one series,6 serum protein electrophoresis showed a spike or localized band in only 82 percent of patients with multiple myeloma. The remainder had hypogammaglobulinemia or a normal-appearing pattern. Consequently, urine protein electrophoresis is recommended in all patients suspected of having a plasma cell dyscrasia.10
An additional point to consider is the size of the M-protein spike. Although this spike is usually greater than 3 g per dL in patients with multiple myeloma, up to one fifth of patients with this tumor may have an M-protein spike of less than 1 g per dL.10 Hypogammaglobulinemia on serum protein electrophoresis occurs in about 10 percent of patients with multiple myeloma who do not have a serum M-protein spike.11 Most of these patients have a large amount of Bence Jones protein (monoclonal free kappa or lambda chain) in their urine.11 Thus, the size of the M-protein spike is not helpful in excluding multiple myeloma.
If multiple myeloma still is considered clinically in a patient who does not have an M-protein spike on serum protein electrophoresis, urine protein electrophoresis should be performed.
Evaluation of an Abnormal Serum Protein Electrophoresis
Monoclonal gammopathy is present in up to 8 percent of healthy geriatric patients.12 All patients with monoclonal gammopathy require further evaluation to determine the cause of the abnormality. Patients with monoclonal gammopathy of undetermined significance require close follow-up because about 1 percent per year develop multiple myeloma or another malignant monoclonal gammopathy.13 [Evidence level B, prospective cohort study] An algorithm for the follow-up of patients with a monoclonal gammopathy is provided in Figure 3.6
Figure 3
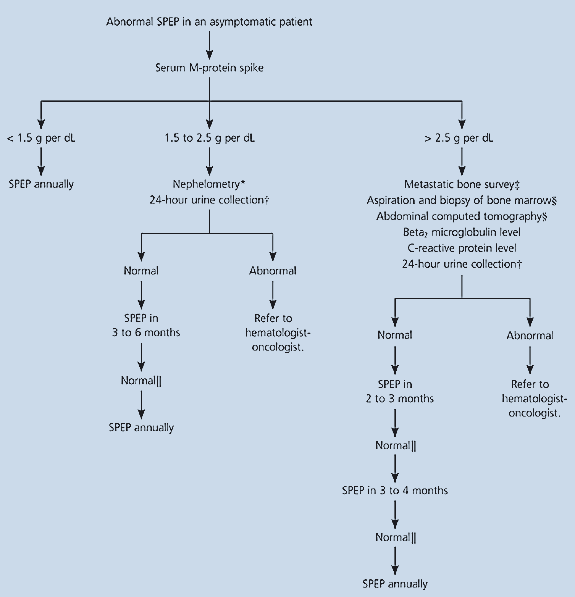
Suggested algorithm for follow-up of a monoclonal gammopathy. (SPEP = serum protein electrophoresis)Information from reference6.
If the serum M-protein spike is 1.5 to 2.5 g per dL, it is important to perform nephelometry to quantify the immunoglobulins present and to obtain a 24-hour urine collection for electrophoresis and immunofixation. If these examinations are normal, serum protein electrophoresis should be repeated in three to six months; if that examination is normal, serum protein electrophoresis should be repeated annually. If the repeat examination is abnormal or future patterns are abnormal, the next step is to refer the patient to a hematologist-oncologist.
An M-protein spike of greater than 2.5 g per dL should be assessed with a metastatic bone survey that includes a single view of the humeri and femurs. In addition, a beta2 microglobulin test, a CRP test, and a 24-hour urine collection for electrophoresis and immunofixation should be performed. If Waldenström’s macroglobulinemia or other lymphoproliferative process is suspected, an abdominal computed tomographic scan and bone marrow aspiration and biopsy should be performed. Abnormalities in any of these tests should result in a referral to a hematologist-oncologist. If all tests are normal, the pattern of follow-up in Figure 36 can be undertaken. If serum protein electrophoresis is abnormal at any time during the follow-up, a referral should be made

