Clinical Question
Does therapy with angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) prevent progression to chronic kidney disease (CKD) in normotensive patients with microalbuminuria?
Evidence-Based Answer
ACE inhibitors reduce the risk of progression to macroalbuminuria in normotensive patients with microalbuminuria and type 1 diabetes mellitus. (Strength of Recommendation [SOR]: C, based on a meta-analysis of randomized controlled trials [RCTs] with disease-oriented outcomes.) ACE inhibitors and ARBs reduce the risk of progression to macroalbuminuria in normotensive patients with microalbuminuria and type 2 diabetes. (SOR: C, based on disease-oriented evidence from small RCTs.) ACE inhibitors and ARBs do not reliably affect serum creatinine levels. Recommendations are not available for normotensive patients without diabetes who have microalbuminuria.
Evidence Summary
TYPE 1 DIABETES
A 2001 meta-analysis of 10 small RCTs (N = 698) described the effect of ACE inhibitors vs. placebo on progression to macro-albuminuria in normotensive patients with microalbuminuria and type 1 diabetes.1 Several definitions of normotension were allowed; microalbuminuria was defined as a urinary albumin excretion rate of 20 to 200 mcg per minute, whereas a rate greater than 200 mcg per minute was considered macroalbuminuria. Two-year follow-up data were available for 646 patients. Nine of the 10 trials demonstrated a significantly lower risk of progression to macroalbuminuria with ACE inhibitors compared with placebo (odds ratio [OR] = 0.38; 95% confidence interval [CI], 0.25 to 0.57). Regression to normal albuminuria occurred more often with ACE inhibitors than with placebo (OR = 3.07; 95% CI, 2.14 to 4.44). Changes in estimated creatinine clearance and glomerular filtration rate were not reported. The authors of this meta-analysis and several of the referenced studies received funding from industry.
TYPE 2 DIABETES
All studies demonstrated the benefits of therapy with ACE inhibitors and ARBs on urinary albumin excretion in patients with type 2 diabetes, but the magnitude of effect varied by comparison group. Three RCTs showed a statistically significant reduction in urinary albumin excretion in patients treated with losartan (Cozaar) or enalapril (Vasotec) over one to five years; two studies showed no difference with ramipril (Altace) or enalapril (Table 1).2–6 Only one study found a significant difference in the serum creatinine level in treated patients (significant decrease in the enalapril group).4 No study of patients with type 2 diabetes found a significant difference in blood pressure, A1C level, or body mass index between treated patients and the control group.
Table 1. Summary of Randomized Controlled Trials of Normotensive Patients with Type 2 Diabetes Mellitus and Microalbuminuria
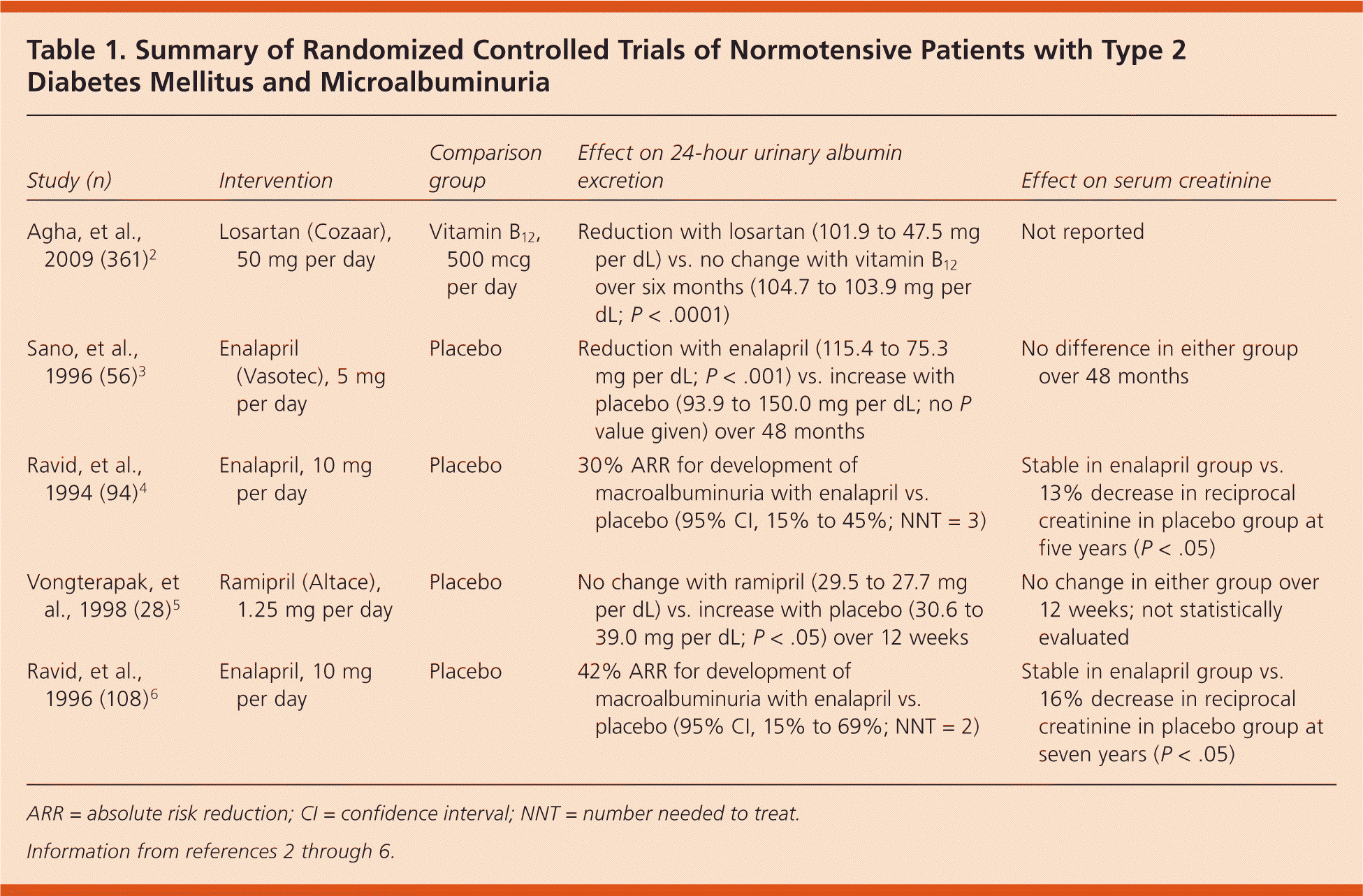
| Study (n) | Intervention | Comparison group | Effect on 24-hour urinary albumin excretion | Effect on serum creatinine |
|---|---|---|---|---|
| Agha, et al., 2009 (361)2 | Losartan (Cozaar), 50 mg per day | Vitamin B12, 500 mcg per day | Reduction with losartan (101.9 to 47.5 mg per dL) vs. no change with vitamin B12 over six months (104.7 to 103.9 mg per dL; P < .0001) | Not reported |
| Sano, et al., 1996 (56)3 | Enalapril (Vasotec), 5 mg per day | Placebo | Reduction with enalapril (115.4 to 75.3 mg per dL; P < .001) vs. increase with placebo (93.9 to 150.0 mg per dL; no P value given) over 48 months | No difference in either group over 48 months |
| Ravid, et al., 1994 (94)4 | Enalapril, 10 mg per day | Placebo | 30% ARR for development of macroalbuminuria with enalapril vs. placebo (95% CI, 15% to 45%; NNT = 3) | Stable in enalapril group vs. 13% decrease in reciprocal creatinine in placebo group at five years (P < .05) |
| Vongterapak, et al., 1998 (28)5 | Ramipril (Altace), 1.25 mg per day | Placebo | No change with ramipril (29.5 to 27.7 mg per dL) vs. increase with placebo (30.6 to 39.0 mg per dL; P < .05) over 12 weeks | No change in either group over 12 weeks; not statistically evaluated |
| Ravid, et al., 1996 (108)6 | Enalapril, 10 mg per day | Placebo | 42% ARR for development of macroalbuminuria with enalapril vs. placebo (95% CI, 15% to 69%; NNT = 2) | Stable in enalapril group vs. 16% decrease in reciprocal creatinine in placebo group at seven years (P < .05) |
ARR = absolute risk reduction; CI = confidence interval; NNT = number needed to treat.
Recommendations from Others
The Kidney Disease: Improving Global Outcomes guideline recommends ARBs or ACE inhibitors for adults with CKD with or without diabetes who have a urinary albumin excretion rate of 30 to 300 mg per 24 hours and in whom blood pressure medications are indicated. The guideline recommends a blood pressure goal of 130/80 mm Hg or less in these patients.7 The American Diabetes Association recommends ACE inhibitors or ARBs for nonpregnant patients with diabetes and microalbuminuria, but it makes no specific recommendations for blood pressure beyond its standard goals.8 The National Kidney Foundation recommends an ACE inhibitor or ARB in normotensive patients with diabetes and an albumin level greater than 30 mg per g who are at high risk of CKD or progression.9 The National Kidney Foundation guideline does not provide recommendations for normotensive patients without diabetes.

