The U.S. Preventive Services Task Force (USPSTF) has issued recommendations on behavioral counseling to prevent sexually transmitted infections (STIs) and recommendations about screening for individual STIs. Clinicians should obtain a sexual history to assess for behaviors that increase a patient's risk. Community and population risk factors should also be considered. The USPSTF recommends intensive behavioral counseling for all sexually active adolescents and for adults whose history indicates an increased risk of STIs. These interventions can reduce STI acquisition and risky sexual behaviors, and increase condom use and other protective behaviors. The USPSTF recommends screening for chlamydia and gonorrhea in all sexually active women 24 years and younger, and in older women at increased risk. It recommends screening for human immunodeficiency virus (HIV) infection in all patients 15 to 65 years of age regardless of risk, as well as in younger and older patients at increased risk of HIV infection. The USPSTF also recommends screening for hepatitis B virus infection and syphilis in persons at increased risk. All pregnant women should be tested for hepatitis B virus infection, HIV infection, and syphilis. Pregnant women 24 years and younger, and older women with risk factors should be tested for gonorrhea and chlamydia. The USPSTF recommends against screening for asymptomatic herpes simplex virus infection. There is inadequate evidence to determine the optimal interval for repeat screening; clinicians should rescreen patients when their sexual history reveals new or persistent risk factors.
The Centers for Disease Control and Prevention estimates that 20 million new cases of sexually transmitted infections (STIs) occur each year in the United States.1 STIs are often asymptomatic, which can lead to inadvertent transmission of these infections.2 Undiagnosed and untreated STIs can cause serious complications and adverse health outcomes, including death.
SORT: KEY RECOMMENDATIONS FOR PRACTICE

| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Provide or refer for intensive behavioral counseling in all sexually active adolescents (regardless of risk) and in adults who are at increased risk of sexually transmitted infection. | B | 8 |
| Screen for chlamydia and gonorrhea in sexually active nonpregnant female adolescents and adults 24 years and younger, and in older women who are at increased risk. | B | 3 |
| Screen for HIV infection in adolescents and adults 15 to 65 years of age. Younger adolescents and older adults who are at increased risk should also be screened. | A | 6 |
| Screen all pregnant women for syphilis, hepatitis B virus, and HIV infections. | A | 6, 9, 10 |
| Screen for chlamydia and gonorrhea in all pregnant women 24 years and younger, and in older women who are at increased risk. | B | 3 |
| Screen for syphilis, HIV infection, and hepatitis B in men and women at increased risk. | A | 5–7 |
HIV = human immunodeficiency virus.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
This article provides an overview of current recommendations from the U.S. Preventive Services Task Force (USPSTF) on behavioral counseling and screening for STIs. In addition to its general recommendations on counseling about prevention, the USPSTF has issued recommendations on screening for individual STIs, including chlamydia, gonorrhea, herpes, hepatitis B virus (HBV) infection, human immunodeficiency virus (HIV) infection, and syphilis.3–7 These recommendations, which pertain to adult men and women, adolescents, and pregnant women, are summarized in Table 13–8 and Table 2.3,4,6,9–11 The scale for rating the strength of the recommendations is presented in Table 3.12 USPSTF recommendations on screening for cervical cancer and hepatitis C are not discussed in this article and have been reviewed elsewhere.13,14 The USPSTF recommends against routine serologic screening for infection with herpes simplex virus because there is no evidence that screening reduces transmission or that antiviral therapy in asymptomatic infected persons improves health outcomes.4
Table 1. USPSTF Recommendations for Behavioral Counseling and STI Screening in Nonpregnant Patients
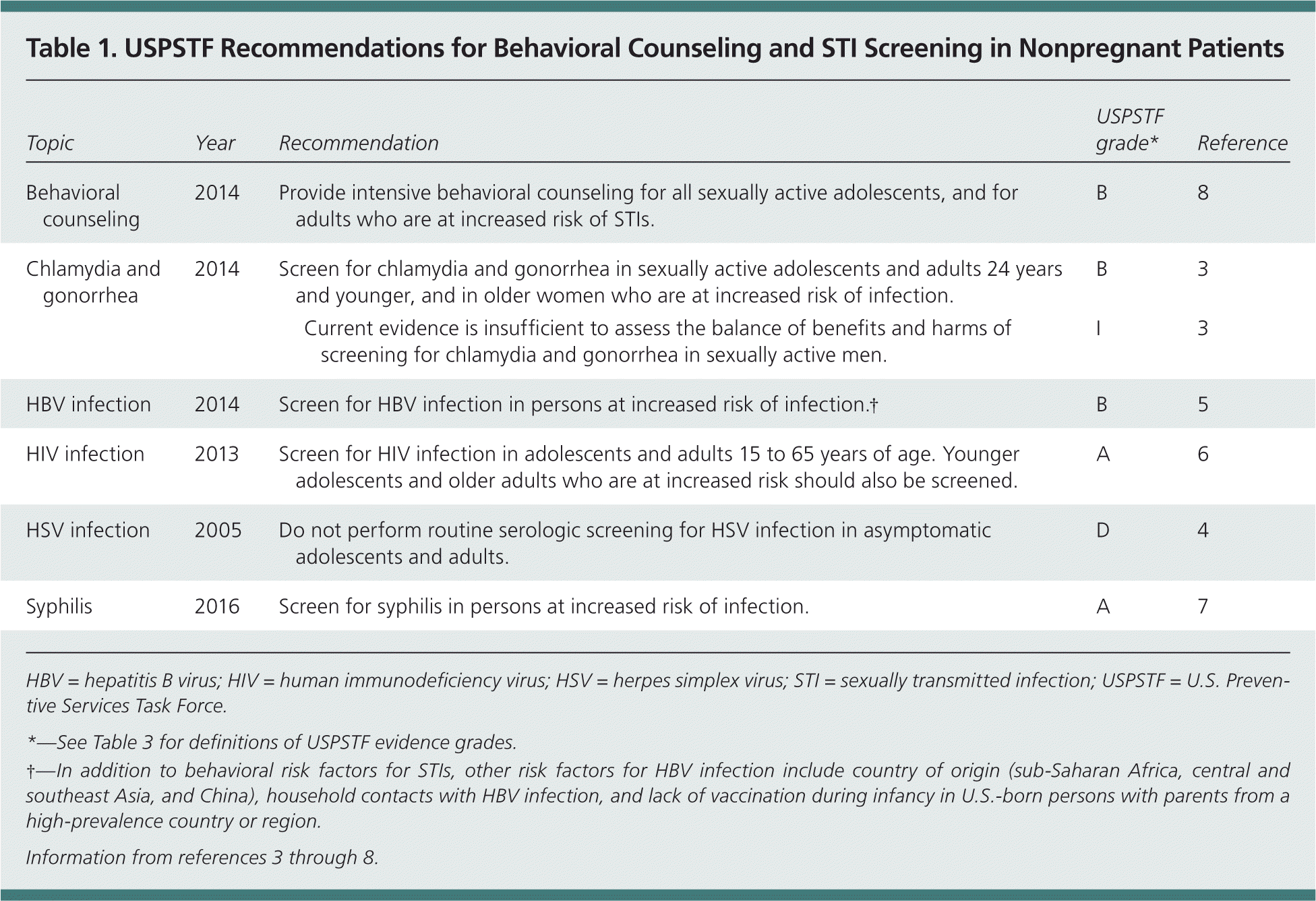
| Topic | Year | Recommendation | USPSTF grade* | Reference | |
|---|---|---|---|---|---|
| Behavioral counseling | 2014 | Provide intensive behavioral counseling for all sexually active adolescents, and for adults who are at increased risk of STIs. | B | 8 | |
| Chlamydia and gonorrhea | 2014 | Screen for chlamydia and gonorrhea in sexually active adolescents and adults 24 years and younger, and in older women who are at increased risk of infection. | B | 3 | |
| Current evidence is insufficient to assess the balance of benefits and harms of screening for chlamydia and gonorrhea in sexually active men. | I | 3 | |||
| HBV infection | 2014 | Screen for HBV infection in persons at increased risk of infection.† | B | 5 | |
| HIV infection | 2013 | Screen for HIV infection in adolescents and adults 15 to 65 years of age. Younger adolescents and older adults who are at increased risk should also be screened. | A | 6 | |
| HSV infection | 2005 | Do not perform routine serologic screening for HSV infection in asymptomatic adolescents and adults. | D | 4 | |
| Syphilis | 2016 | Screen for syphilis in persons at increased risk of infection. | A | 7 | |
HBV = hepatitis B virus; HIV = human immunodeficiency virus; HSV = herpes simplex virus; STI = sexually transmitted infection; USPSTF = U.S. Preventive Services Task Force.
*—See Table 3 for definitions of USPSTF evidence grades.
†—In addition to behavioral risk factors for STIs, other risk factors for HBV infection include country of origin (sub-Saharan Africa, central and southeast Asia, and China), household contacts with HBV infection, and lack of vaccination during infancy in U.S.-born persons with parents from a high-prevalence country or region.
Table 2. USPSTF Recommendations for Screening for Sexually Transmitted Infections in Pregnant Patients

| Topic | Year | Recommendation | USPSTF grade* | Reference |
|---|---|---|---|---|
| Chlamydia and gonorrhea† | 2014 | Screen for chlamydia and gonorrhea in all pregnant women 24 years and younger, and in older women who are at increased risk of infection. | B | 3 |
| HBV infection‡ | 2009 | Screen pregnant women for HBV infection at their first prenatal visit. | A | 9 |
| HIV infection‡ | 2013 | Screen all pregnant women for HIV infection, including those who present in labor and whose HIV status is unknown. | A | 6 |
| HSV infection | 2005 | Do not perform routine serologic screening for HSV infection in asymptomatic pregnant women at any time during pregnancy to prevent neonatal HSV infection. | D | 4 |
| Syphilis‡ | 2009 | Screen all pregnant women for syphilis. | A | 10 |
HBV = hepatitis B virus; HIV = human immunodeficiency virus; HSV = herpes simplex virus; USPSTF = U.S. Preventive Services Task Force.
*—See Table 3 for definitions of USPSTF evidence grades.
†—In 2011, the USPSTF recommended the use of prophylactic ocular topical medication for all newborns for the prevention of gonococcal ophthalmia neonatorum (A recommendation).11
‡—A pragmatic approach in all pregnant women is to screen for HBV infection, HIV infection, and syphilis in the first trimester, with repeat screening in the third trimester for women with high-risk behaviors or exposures. Women who present in labor and whose infection status for HBV, HIV, and/or syphilis is unknown should also be tested. Women screened during a previous pregnancy should be rescreened in subsequent pregnancies.
Table 3. USPSTF Evidence Grades: Definitions and Suggestions for Clinical Practice
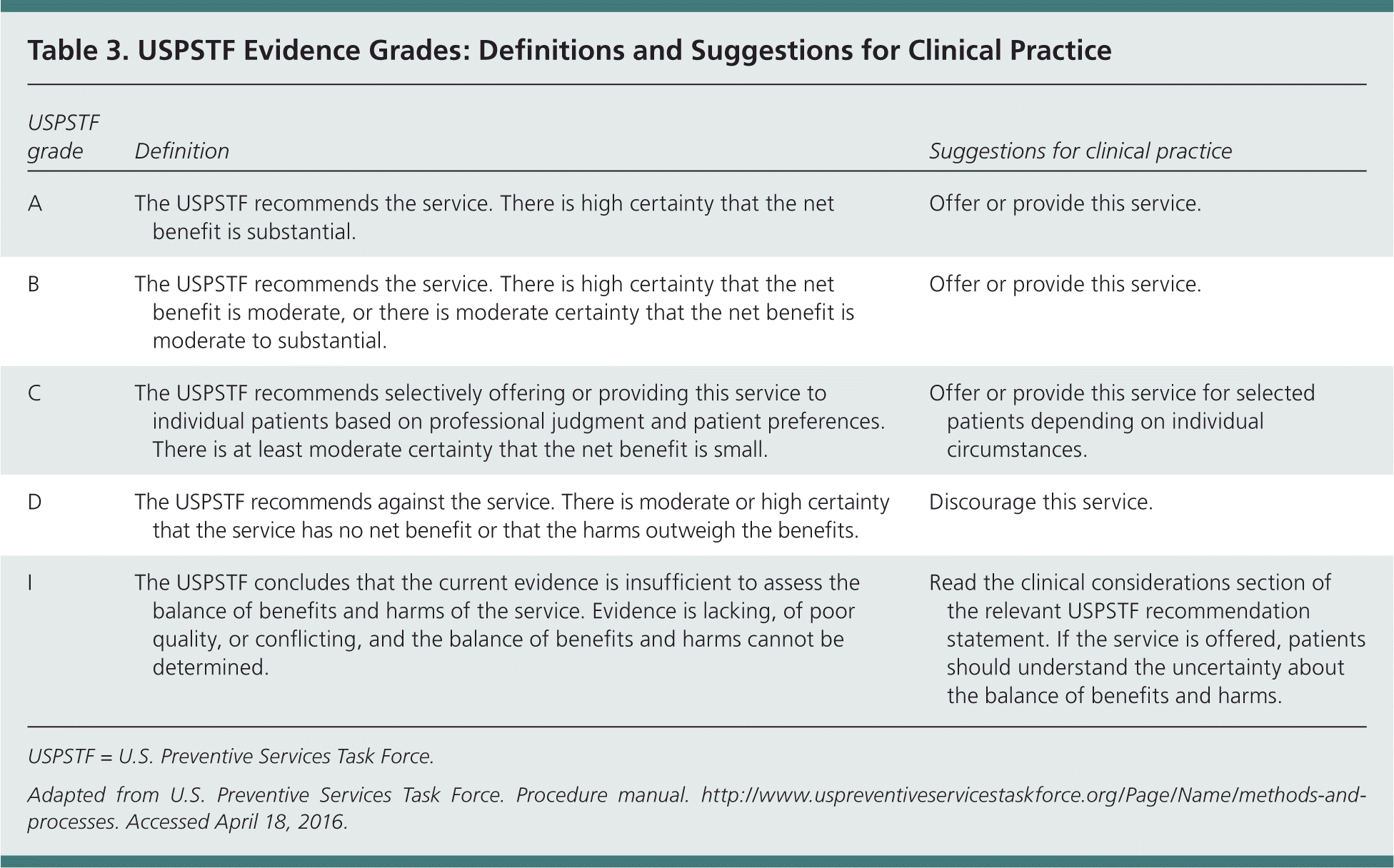
| USPSTF grade | Definition | Suggestions for clinical practice |
|---|---|---|
| A | The USPSTF recommends the service. There is high certainty that the net benefit is substantial. | Offer or provide this service. |
| B | The USPSTF recommends the service. There is high certainty that the net benefit is moderate, or there is moderate certainty that the net benefit is moderate to substantial. | Offer or provide this service. |
| C | The USPSTF recommends selectively offering or providing this service to individual patients based on professional judgment and patient preferences. There is at least moderate certainty that the net benefit is small. | Offer or provide this service for selected patients depending on individual circumstances. |
| D | The USPSTF recommends against the service. There is moderate or high certainty that the service has no net benefit or that the harms outweigh the benefits. | Discourage this service. |
| I | The USPSTF concludes that the current evidence is insufficient to assess the balance of benefits and harms of the service. Evidence is lacking, of poor quality, or conflicting, and the balance of benefits and harms cannot be determined. | Read the clinical considerations section of the relevant USPSTF recommendation statement. If the service is offered, patients should understand the uncertainty about the balance of benefits and harms. |
USPSTF = U.S. Preventive Services Task Force.
Adapted from U.S. Preventive Services Task Force. Procedure manual. http://www.uspreventiveservicestaskforce.org/Page/Name/methods-and-processes. Accessed April 18, 2016.
Assessment of STI Risk
Clinicians should obtain a sexual history from patients at visits in which preventive services are addressed, or when symptoms that may be related to an STI are present. When making decisions about STI screening, clinicians should consider each patient's individual risk behaviors and social situation. Persons who are not sexually active and those who are sexually active in mutually monogamous relationships with uninfected partners are not at increased risk of STIs.6
INDIVIDUAL BEHAVIORAL RISKS
Persons at increased risk of STIs include those who have more than one sex partner, a new sex partner, or a sex partner with concurrent partners, and those who are not in mutually monogamous relationships and have inconsistent condom use. The risk is even higher in persons who have a sex partner with an STI, those who have a previous or current STI, and those who exchange sex for drugs or money.3,6,8,15–17 Regular condom use can reduce STI risk in persons with behavioral risk factors for STIs. However, these persons should still be considered at increased risk and screened accordingly.
COMMUNITY/POPULATION RISKS
STI prevalence rates are increased in specific populations, such as men who have sex with men, persons with low incomes who live in urban settings, current or former prison inmates, military recruits, persons with mental illness or disability, current or former injection drug users, persons with a history of sexual abuse, and patients presenting at public STI clinics.18 Clinicians should consider updating the sexual history of persons in these high-prevalence populations more often.
Risks for STIs vary across and within communities. Some STIs, particularly gonorrhea, are concentrated in specific geographic locations and communities.18 These variations may be related to social and economic influences (e.g., effects of poverty and discrimination). Race and ethnicity can be surrogate markers for some of the underlying socioeconomic factors that increase community STI risk,19 with higher prevalence rates among some racial and ethnic minority groups (e.g., blacks, Hispanics, Native Americans).18 Clinicians can obtain up-to-date information about STI prevalence in different geographic areas, such as by county, at http://www.cdc.gov/std/stats/default.htm, and about STI prevalence by racial or ethnic group at http://www.cdc.gov/std/stats14/minorities.htm. Local public health agencies can also provide information on community STI prevalence rates and other pertinent epidemiologic information. Because patients may not be aware of their sex partners' STI risk factors, clinicians in communities with high STI prevalence rates may choose to consider all sexually active members of the community at increased risk.
SPECIAL RISKS FOR HEPATITIS INFECTION
In addition to behavioral risk factors, risk factors for HBV infection include country of origin (particularly sub-Saharan Africa, central and southeast Asia, and China), household contacts with HBV infection, and lack of vaccination during infancy in U.S.-born persons with parents from a high-prevalence country or region.20–23 Persons who were not properly vaccinated or who are uncertain of their vaccination status should receive the HBV vaccine or undergo serologic testing to confirm HBV status with subsequent vaccination if results are negative.
Behavioral Counseling
The USPSTF recommends intensive behavioral counseling for all sexually active adolescents (regardless of risk) and for adults who are at increased risk of STIs, based on evidence that these interventions can reduce risky sexual behaviors, increase the likelihood of condom use and other protective behaviors, and decrease the likelihood of STI acquisition.8,17
The USPSTF found that behavioral counseling interventions ranging in intensity from 30 minutes to two hours or more are generally effective, with the effect increasing in proportion to intensity.8,17 Intensive behavioral counseling may be delivered in primary care settings or other sectors of the health care system. Although the evidence is limited, the USPSTF found that some interventions of lesser intensity are also effective.24–26 Clinicians should use their judgment based on practical time constraints and patient risk in making decisions about when to offer guidance on STI prevention (e.g., when taking a sexual history) and the duration and intensity of counseling.8,17
Regardless of duration and intensity, most successful counseling approaches provide basic information about STIs and about reducing STI transmission (e.g., abstinence, mutual monogamy, condom use). They also include a discussion of the patient's risks of acquisition and transmission of STIs, and they empower patients by providing education about proper condom use, communication with partners about safe sex, and how to deal with difficult sexual situations.8,17 The USPSTF recommendations on behavioral counseling provide more detailed examples of counseling interventions of varying intensities.8
The USPSTF found no good evidence that behavioral counseling about sex is associated with increased sexual activity among adolescents.24,27–30 It also found no evidence that behavioral counseling to prevent STIs is associated with avoidance of subsequent medical care, or with making patients feel ashamed, guilty, or stigmatized.8
Approach to STI Screening
Table 4 shows a pragmatic approach to STI screening in sexually active patient populations and summarizes USPSTF recommendations for men and nonpregnant women.3,5–8
Table 4. USPSTF Recommendations for STI Screening in Sexually Active Adults, by Population
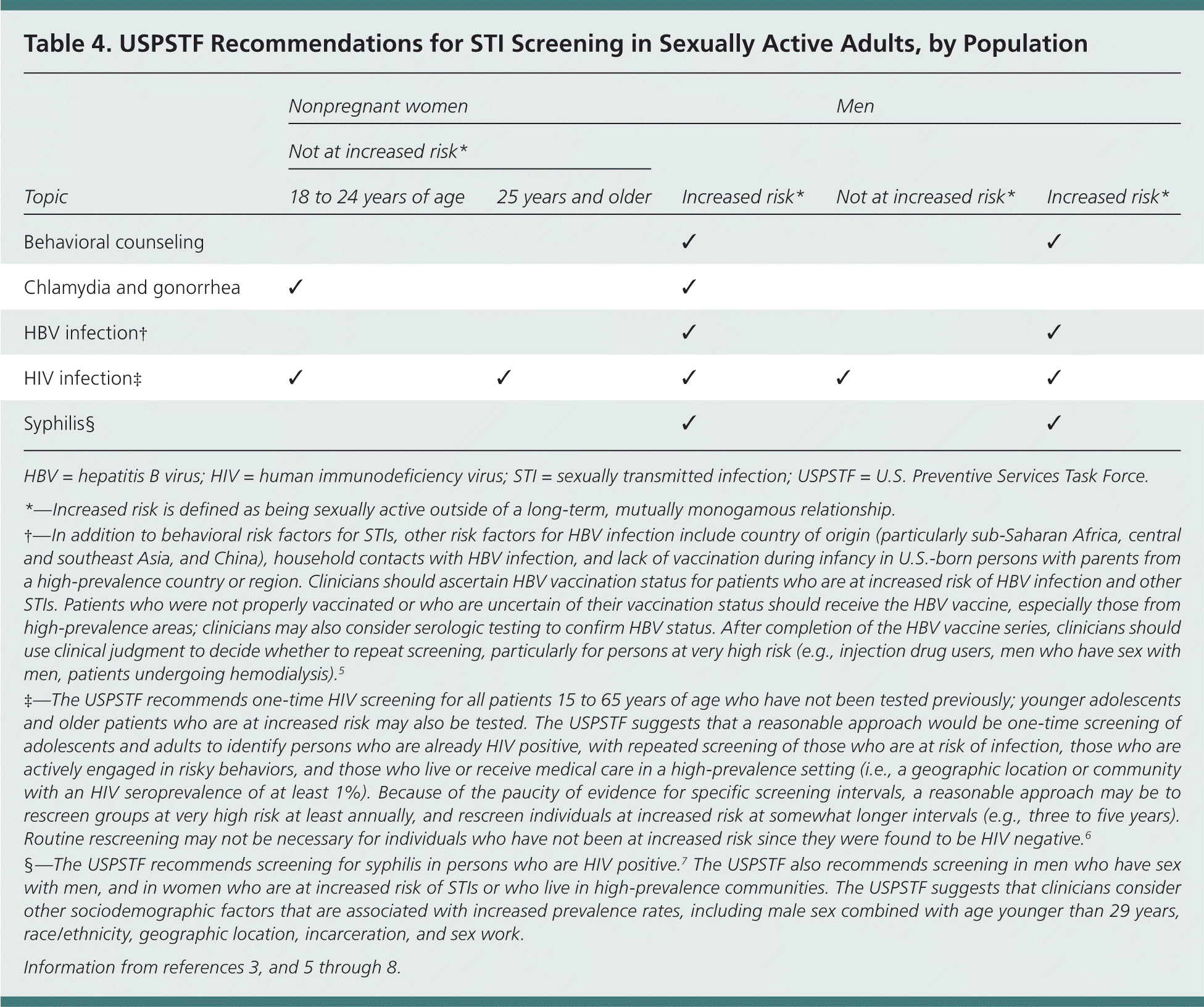
| Nonpregnant women | Men | ||||
|---|---|---|---|---|---|
| Not at increased risk* | |||||
| Topic | 18 to 24 years of age | 25 years and older | Increased risk* | Not at increased risk* | Increased risk* |
| Behavioral counseling | ✔ | ✔ | |||
| Chlamydia and gonorrhea | ✔ | ✔ | |||
| HBV infection† | ✔ | ✔ | |||
| HIV infection‡ | ✔ | ✔ | ✔ | ✔ | ✔ |
| Syphilis§ | ✔ | ✔ | |||
HBV = hepatitis B virus; HIV = human immunodeficiency virus; STI = sexually transmitted infection; USPSTF = U.S. Preventive Services Task Force.
*—Increased risk is defined as being sexually active outside of a long-term, mutually monogamous relationship.
†—In addition to behavioral risk factors for STIs, other risk factors for HBV infection include country of origin (particularly sub-Saharan Africa, central and southeast Asia, and China), household contacts with HBV infection, and lack of vaccination during infancy in U.S.-born persons with parents from a high-prevalence country or region. Clinicians should ascertain HBV vaccination status for patients who are at increased risk of HBV infection and other STIs. Patients who were not properly vaccinated or who are uncertain of their vaccination status should receive the HBV vaccine, especially those from high-prevalence areas; clinicians may also consider serologic testing to confirm HBV status. After completion of the HBV vaccine series, clinicians should use clinical judgment to decide whether to repeat screening, particularly for persons at very high risk (e.g., injection drug users, men who have sex with men, patients undergoing hemodialysis). 5
‡—The USPSTF recommends one-time HIV screening for all patients 15 to 65 years of age who have not been tested previously; younger adolescents and older patients who are at increased risk may also be tested. The USPSTF suggests that a reasonable approach would be one-time screening of adolescents and adults to identify persons who are already HIV positive, with repeated screening of those who are at risk of infection, those who are actively engaged in risky behaviors, and those who live or receive medical care in a high-prevalence setting (i.e., a geographic location or community with an HIV seroprevalence of at least 1%). Because of the paucity of evidence for specific screening intervals, a reasonable approach may be to rescreen groups at very high risk at least annually, and rescreen individuals at increased risk at somewhat longer intervals (e.g., three to five years). Routine rescreening may not be necessary for individuals who have not been at increased risk since they were found to be HIV negative.6
§—The USPSTF recommends screening for syphilis in persons who are HIV positive. 7The USPSTF also recommends screening in men who have sex with men, and in women who are at increased risk of STIs or who live in high-prevalence communities. The USPSTF suggests that clinicians consider other sociodemographic factors that are associated with increased prevalence rates, including male sex combined with age younger than 29 years, race/ethnicity, geographic location, incarceration, and sex work.
NONPREGNANT WOMEN WHO ARE NOT AT INCREASED RISK OF STI
For women who are not at increased risk of STIs, clinicians should consider the patient's age when determining which screening tests to offer. The USPSTF recommends screening for chlamydia, gonorrhea, and HIV infection (if not previously tested) in all sexually active women 18 to 24 years of age who are not at increased risk.3,6 For sexually active women 25 years and older who are not at increased risk, the USPSTF recommends screening for HIV if not previously tested.6
NONPREGNANT WOMEN WHO ARE AT INCREASED RISK OF STI
The USPSTF recommends screening for gonorrhea, chlamydia, HIV infection, syphilis, and HBV infection in sexually active women of all ages who are at increased risk.3,5–7
MEN
The USPSTF recommends screening for syphilis, HIV infection (if not previously tested), and HBV infection (if not previously vaccinated or if vaccination status is unknown) in men at increased risk.5–7 It found insufficient evidence that screening high-risk men or all men for chlamydia or gonorrhea reduces acquisition, transmission, or complications of either infection.3 Men who are not at increased risk of HIV infection should also be screened.6
PREGNANT WOMEN
In general, clinicians should determine a pregnant woman's risk status using the same risk factors as for nonpregnant women (e.g., high-risk sexual behaviors, age). Because of the implications for neonatal infection and the need to institute treatment in newborns, the USPSTF recommends screening all pregnant women for HBV infection, HIV infection, and syphilis.6,9,10 The USPSTF also recommends screening for chlamydia and gonorrhea in all pregnant women 24 years and younger, as well as in older pregnant women who are at increased risk.3
Screening Intervals
The USPSTF found inadequate evidence to make recommendations on the optimal intervals for repeat STI screening.3–7 In the absence of good studies on screening intervals, the USPSTF suggests that a reasonable approach is to screen patients whose sexual history reveals new or persistent risk factors since the last negative test result.3 For women 24 years and younger, annual screening for chlamydia and gonorrhea may be considered, regardless of interval sexual history. Additional suggestions about screening intervals are noted in the footnotes of Table 23,4,6,9–11 and Table 4.3,5–8
Recommendations from Other Organizations
Recommendations from the USPSTF, Centers for Disease Control and Prevention, American Academy of Family Physicians (AAFP), and the American Congress of Obstetricians and Gynecologists are summarized in Table 53–7,31–34 and Table 6.3–7,9,10,31,32,35 Most of the recommendations are similar, and the AAFP closely follows the USPSTF. However, the AAFP recommends routine screening for HIV infection starting at 18 years of age, whereas the USPSTF recommends routine screening starting at 15 years of age.
Table 5. Comparison of STI Screening Recommendations for Sexually Active Nonpregnant Adult and Adolescent Females
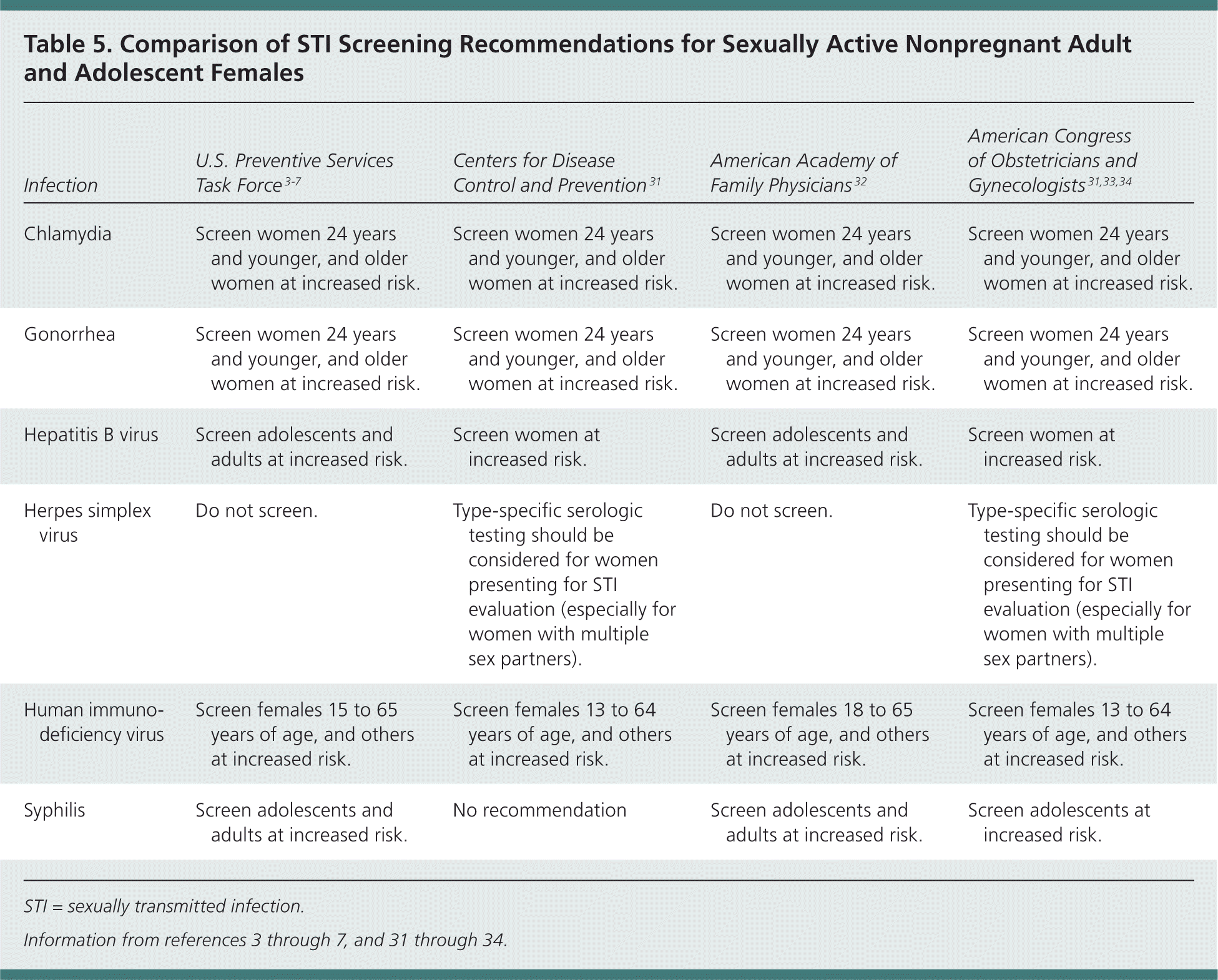
| Infection | U.S. Preventive Services Task Force3–7 | Centers for Disease Control and Prevention31 | American Academy of Family Physicians32 | American Congress of Obstetricians and Gynecologists31,33,34 |
|---|---|---|---|---|
| Chlamydia | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. |
| Gonorrhea | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. |
| Hepatitis B virus | Screen adolescents and adults at increased risk. | Screen women at increased risk. | Screen adolescents and adults at increased risk. | Screen women at increased risk. |
| Herpes simplex virus | Do not screen. | Type-specific serologic testing should be considered for women presenting for STI evaluation (especially for women with multiple sex partners). | Do not screen. | Type-specific serologic testing should be considered for women presenting for STI evaluation (especially for women with multiple sex partners). |
| Human immunodeficiency virus | Screen females 15 to 65 years of age, and others at increased risk. | Screen females 13 to 64 years of age, and others at increased risk. | Screen females 18 to 65 years of age, and others at increased risk. | Screen females 13 to 64 years of age, and others at increased risk. |
| Syphilis | Screen adolescents and adults at increased risk. | No recommendation | Screen adolescents and adults at increased risk. | Screen adolescents at increased risk. |
STI = sexually transmitted infection.
Table 6. Comparison of Screening Recommendations for Sexually Transmitted Infections in Pregnant Women

| Infection | U.S. Preventive Services Task Force3–7,9,10 | Centers for Disease Control and Prevention31 | American Academy of Family Physicians32 | American Congress of Obstetricians and Gynecologists31,35 |
|---|---|---|---|---|
| Chlamydia | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. |
| Gonorrhea | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. | Screen women 24 years and younger, and older women at increased risk. |
| Hepatitis B virus | Screen all. | Screen all. | Screen all. | Screen all. |
| HSV | Do not screen. | Evidence does not support routine HSV-2 serologic screening in asymptomatic pregnant women. However, type-specific serologic testing may be useful for identifying pregnant women at risk of HSV infection, and for guiding counseling about the risk of acquiring genital herpes during pregnancy. | Do not screen. | Evidence does not support routine HSV-2 serologic screening in asymptomatic pregnant women. However, type-specific serologic testing may be useful for identifying pregnant women at risk of HSV infection, and for guiding counseling about the risk of acquiring genital herpes during pregnancy. |
| Human immunodeficiency virus | Screen all. | Screen all. | Screen all. | Screen all. |
| Syphilis | Screen all. | Screen all. | Screen all. | Screen all. |
HSV = herpes simplex virus.
Information from references 3 through 7, 9, 10, 31, 32, and 35.
Data Sources: The authors reviewed the USP-STF's library of recommendations and supporting evidence reviews (available at http://www.uspreventiveservicestaskforce.org). In addition to the USPSTF recommendation on behavioral counseling to prevent STIs, the authors reviewed USPSTF recommendations on screening for chlamydia, gonorrhea, HBV, hepatitis C virus, genital herpes, HIV, herpes simplex virus, syphilis, and cervical cancer. Search dates: December 7, 2015, and April 21, 2016.
The views expressed in this article are those of the authors and do not necessarily reflect the official policies of the Department of Health and Human Services, the National Institutes of Health, nor the Agency of Healthcare Research and Quality, nor does mention of the department or agency imply endorsement by the U.S. government.

