Approximately 1% of primary care office visits are for chest pain, and 2% to 4% of these patients will have unstable angina or myocardial infarction. Initial evaluation is based on determining whether the patient needs to be referred to a higher level of care to rule out acute coronary syndrome (ACS). A combination of age, sex, and type of chest pain can predict the likelihood of coronary artery disease as the cause of chest pain. The Marburg Heart Score and the INTERCHEST clinical decision rule can also help estimate ACS risk. Twelve-lead electrocardiography is recommended to look for ST segment changes, new-onset left bundle branch block, presence of Q waves, and new T-wave inversions. Patients with suspicion of ACS or changes on electrocardiography should be transported immediately to the emergency department. Those at low or intermediate risk of ACS can undergo exercise stress testing, coronary computed tomography angiography, or cardiac magnetic resonance imaging. In those with low suspicion for ACS, consider other diagnoses such as chest wall pain or costochondritis, gastroesophageal reflux disease, and panic disorder or anxiety states. Other less common, but important, diagnostic considerations include acute pericarditis, pneumonia, heart failure, pulmonary embolism, and acute thoracic aortic dissection.
Approximately 1% of all ambulatory visits in primary care settings are for chest pain.1 Cardiac disease is the leading cause of death in the United States, yet only 2% to 4% of patients presenting to a primary care office with chest pain will have unstable angina or an acute myocardial infarction.2–4 The most common causes of chest pain in the primary care population are chest wall pain (20% to 50%), reflux esophagitis (10% to 20%), and costochondritis (13%).2 Other potential factors include pulmonary etiologies (pneumonia, pulmonary embolism [PE]), psychological etiologies (panic disorder), and nonischemic cardiovascular disorders (congestive heart failure, thoracic aortic dissection).2,3,5,6 No definitive diagnosis may be found in as many as 15% of patients.2 Differentiating ischemic from nonischemic causes is often challenging because patients with ischemic chest pain may appear well. As such, the initial diagnostic approach should always consider a cardiac etiology for the chest pain unless other causes are apparent.7
SORT: KEY RECOMMENDATIONS FOR PRACTICE
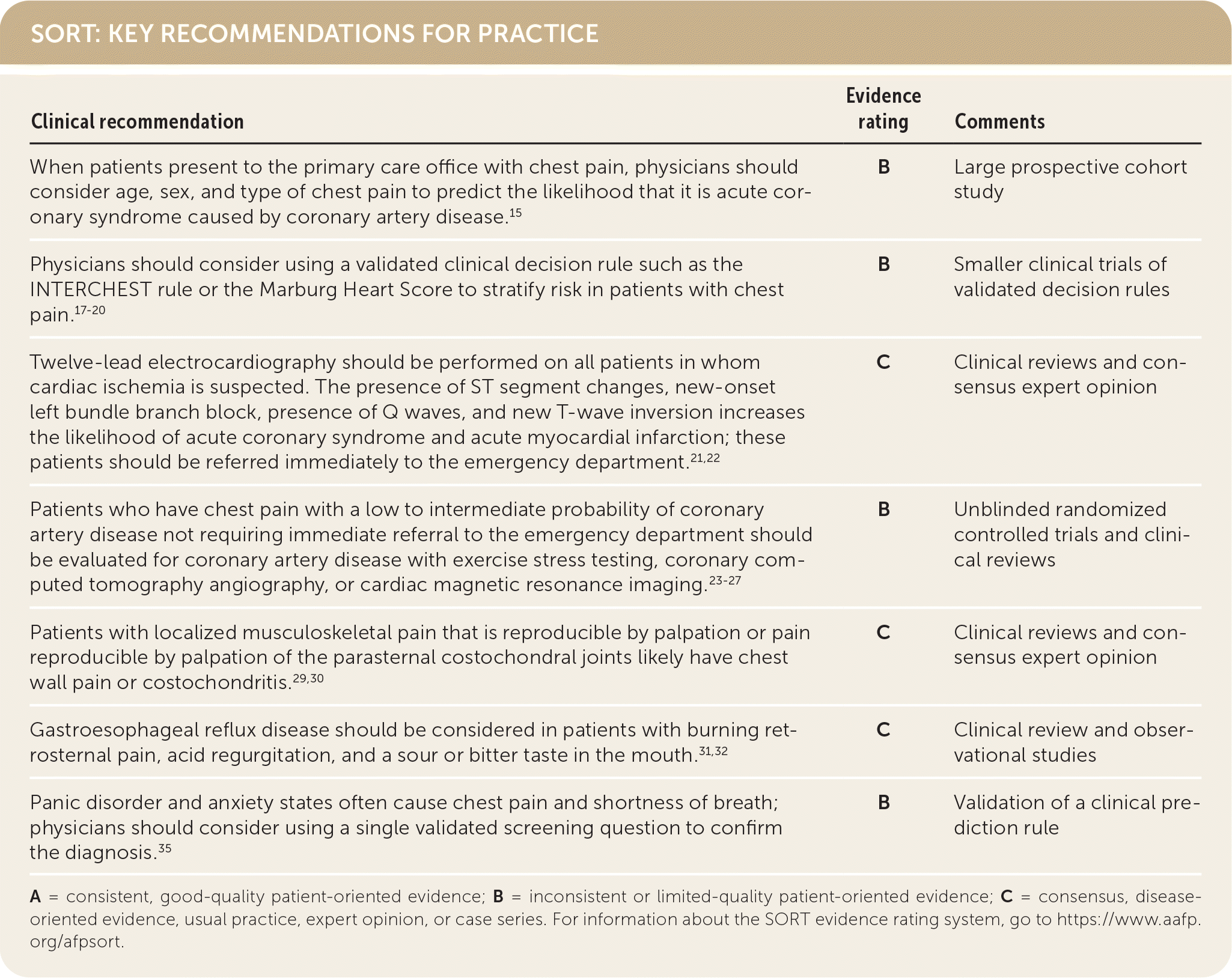
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
BEST PRACTICES IN CARDIOLOGY
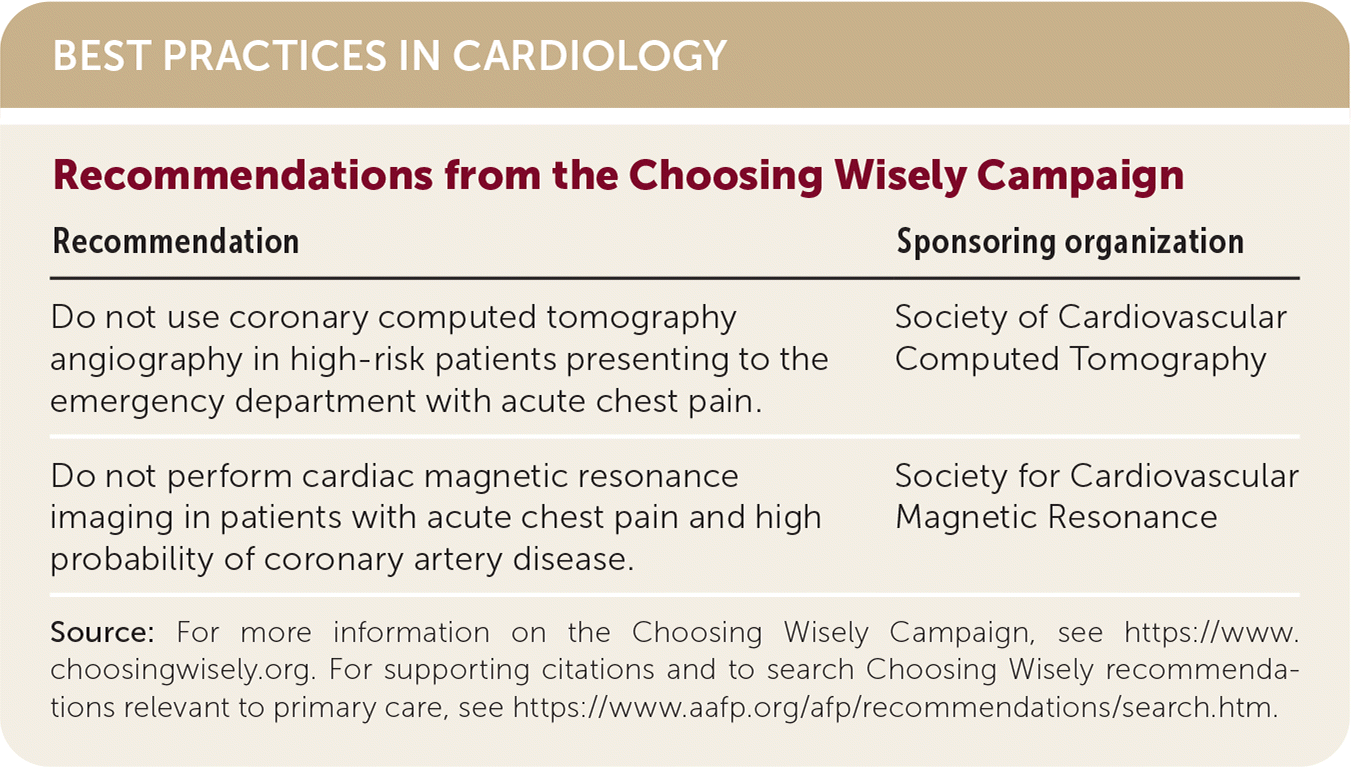
| Recommendation | Sponsoring organization |
|---|---|
| Do not use coronary computed tomography angiography in high-risk patients presenting to the emergency department with acute chest pain. | Society of Cardiovascular Computed Tomography |
| Do not perform cardiac magnetic resonance imaging in patients with acute chest pain and high probability of coronary artery disease. | Society for Cardiovascular Magnetic Resonance |
Source: For more information on the Choosing Wisely Campaign, see https://www.choosingwisely.org. For supporting citations and to search Choosing Wisely recommendations relevant to primary care, see https://www.aafp.org/afp/recommendations/search.htm.
Initial Evaluation
The first decision point for most physicians is to determine whether the patient needs immediate referral to the emergency department for further testing to determine whether the chest pain is an acute coronary syndrome (ACS) caused by coronary ischemia.7 ACS is a clinical diagnosis that includes unstable angina, ST segment elevation myocardial infarction, and non–ST segment elevation myocardial infarction. Definitions of chest pain have evolved over time. Typical chest pain or angina is a deep, poorly localized chest or arm discomfort (pain or pressure) associated with physical exertion or emotional stress and relieved with rest or sublingual nitroglycerin within five minutes.8 Unstable angina is new-onset angina, angina at rest, or angina that becomes more frequent, severe, or prolonged.9 Acute myocardial infarction is myocardial injury resulting in elevated cardiac biomarkers in the setting of acute ischemia caused by ST segment elevation myocardial infarction or non–ST segment elevation myocardial infarction.10 The impression of chest pain is often determined by a combination of clinical symptoms at the time of presentation, physical examination, initial electrocardiography (ECG), and risk factors for ACS.11 Patients often do not use the term pain to describe their symptoms but frequently use other terms such as pressure, aching, discomfort, tightness, squeezing, or indigestion.12
A meta-analysis of studies that evaluated the role of previous chest pain in diagnosing ACS concluded that chest pain that is pleuritic, positional, or reproducible with palpation and not related to exertion is low risk for ACS. Pain that is described as pressure (similar to that of prior myocardial infarction), worse than prior anginal pain, associated with exertion, accompanied by nausea or diaphoresis, and/or radiates to one or both arms/shoulders is higher risk for ACS.13
Although individual characteristics generally do not support or rule out a diagnosis, a combination of these may increase diagnostic accuracy.14 The combination of age, sex, and type of chest pain can predict the likelihood of coronary artery disease (CAD) as the cause of chest pain.15 Table 1 outlines updated predicted pretest probabilities of CAD in patients with chest pain based on these three factors.15 U.S. guidelines recommend that patients with a probability of less than 5% be classified as low risk and not undergo further testing.16 Those with a probability greater than 70% should undergo invasive angiography, and those with a probability of 5% to 70% should undergo noninvasive testing.16 European guidelines use cutoffs of 15% and 85%, respectively.15
TABLE 1. Predicted Pretest Probabilities of Coronary Artery Disease in Patients with Chest Pain Based on Age, Sex, and Type of Chest Pain
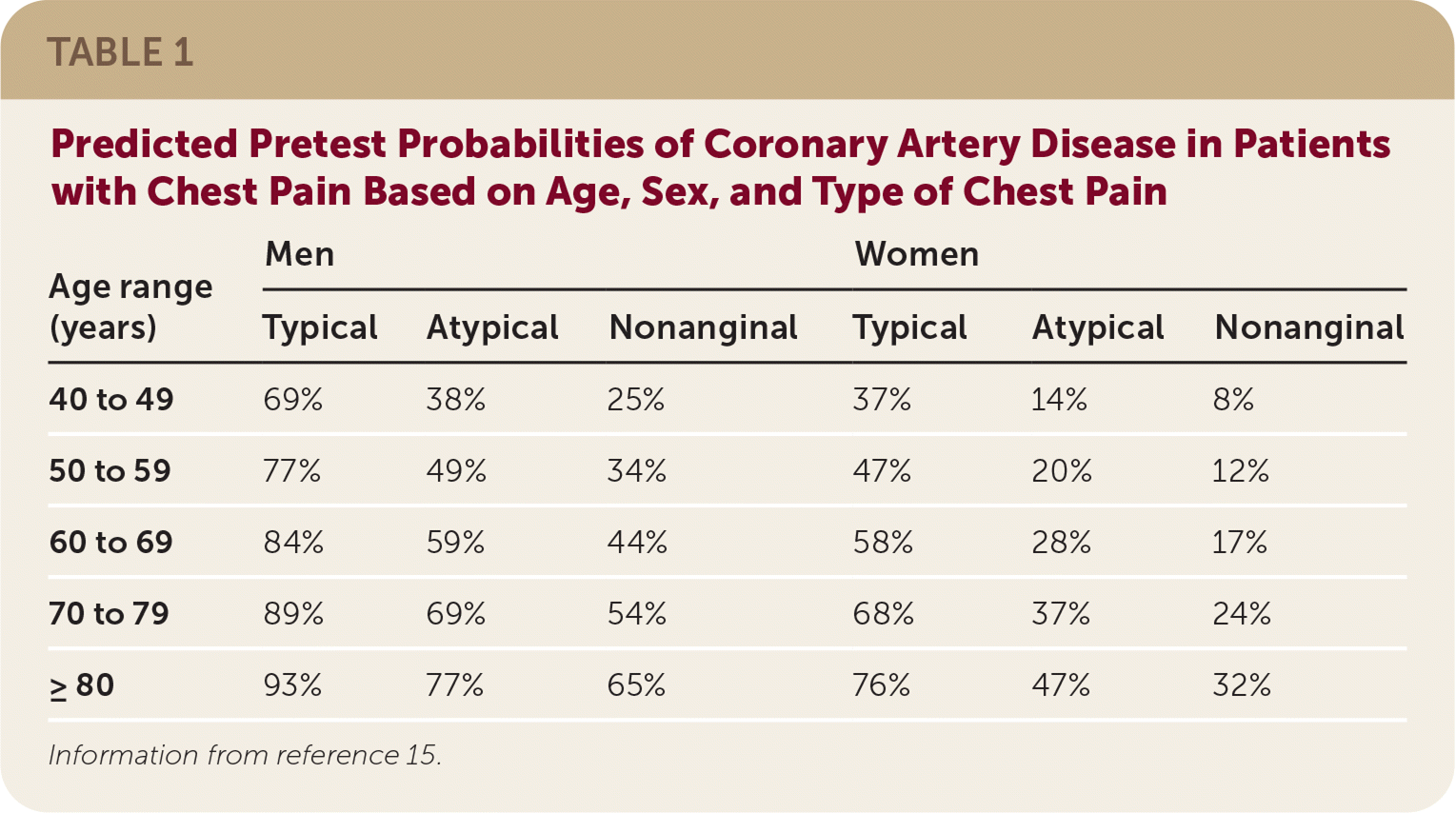
| Age range (years) | Men | Women | ||||
|---|---|---|---|---|---|---|
| Typical | Atypical | Nonanginal | Typical | Atypical | Nonanginal | |
| 40 to 49 | 69% | 38% | 25% | 37% | 14% | 8% |
| 50 to 59 | 77% | 49% | 34% | 47% | 20% | 12% |
| 60 to 69 | 84% | 59% | 44% | 58% | 28% | 17% |
| 70 to 79 | 89% | 69% | 54% | 68% | 37% | 24% |
| ≥ 80 | 93% | 77% | 65% | 76% | 47% | 32% |
Information from reference 15.
Validated clinical decision rules can help determine whether chest pain is caused by CAD. One systematic review found that the validated Marburg Heart Score is better than clinical judgment alone for predicting whether chest pain is cardiac in origin.17,18 Table 2 outlines the scoring for this clinical rule and presents the probability of CAD as the cause of chest pain for pretest probabilities of 2%, 10%, and 20%.18
TABLE 2. Marburg Heart Score to Predict CAD as a Cause of Chest Pain
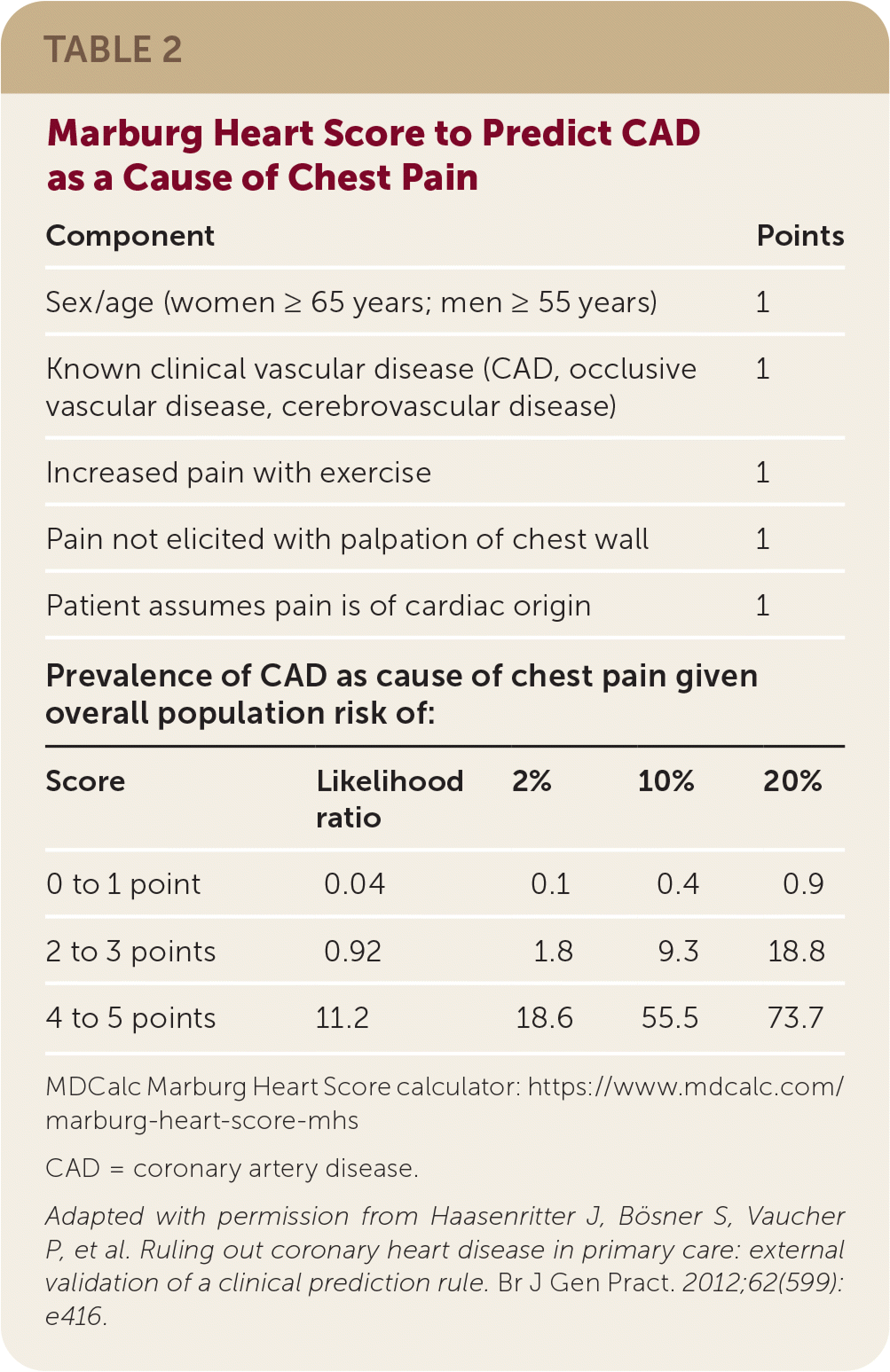
| Component | Points | |||
|---|---|---|---|---|
| Sex/age (women ≥ 65 years; men ≥ 55 years) | 1 | |||
| Known clinical vascular disease (CAD, occlusive vascular disease, cerebrovascular disease) | 1 | |||
| Increased pain with exercise | 1 | |||
| Pain not elicited with palpation of chest wall | 1 | |||
| Patient assumes pain is of cardiac origin | 1 | |||
| Prevalence of CAD as cause of chest pain given overall population risk of: | ||||
| Score | Likelihood ratio | 2% | 10% | 20% |
| 0 to 1 point | 0.04 | 0.1 | 0.4 | 0.9 |
| 2 to 3 points | 0.92 | 1.8 | 9.3 | 18.8 |
| 4 to 5 points | 11.2 | 18.6 | 55.5 | 73.7 |
MDCalc Marburg Heart Score calculator: https://www.mdcalc.com/marburg-heart-score-mhs
CAD = coronary artery disease.
Adapted with permission from Haasenritter J, Bösner S, Vaucher P, et al. Ruling out coronary heart disease in primary care: external validation of a clinical prediction rule. Br J Gen Pract. 2012;62(599):e416.
The INTERCHEST clinical decision rule is a second validated decision rule that can predict the presence or absence of CAD in patients who present with chest pain in the primary care setting (Table 3).19,20 Patients with a score of less than 2 have only a 2% chance of having CAD, whereas 43% of patients with a score of 2 or more have CAD, making the test useful for ruling out CAD as a cause of the patient's chest pain.20
TABLE 3. INTERCHEST Rule for Predicting CAD as the Etiology of Chest Pain
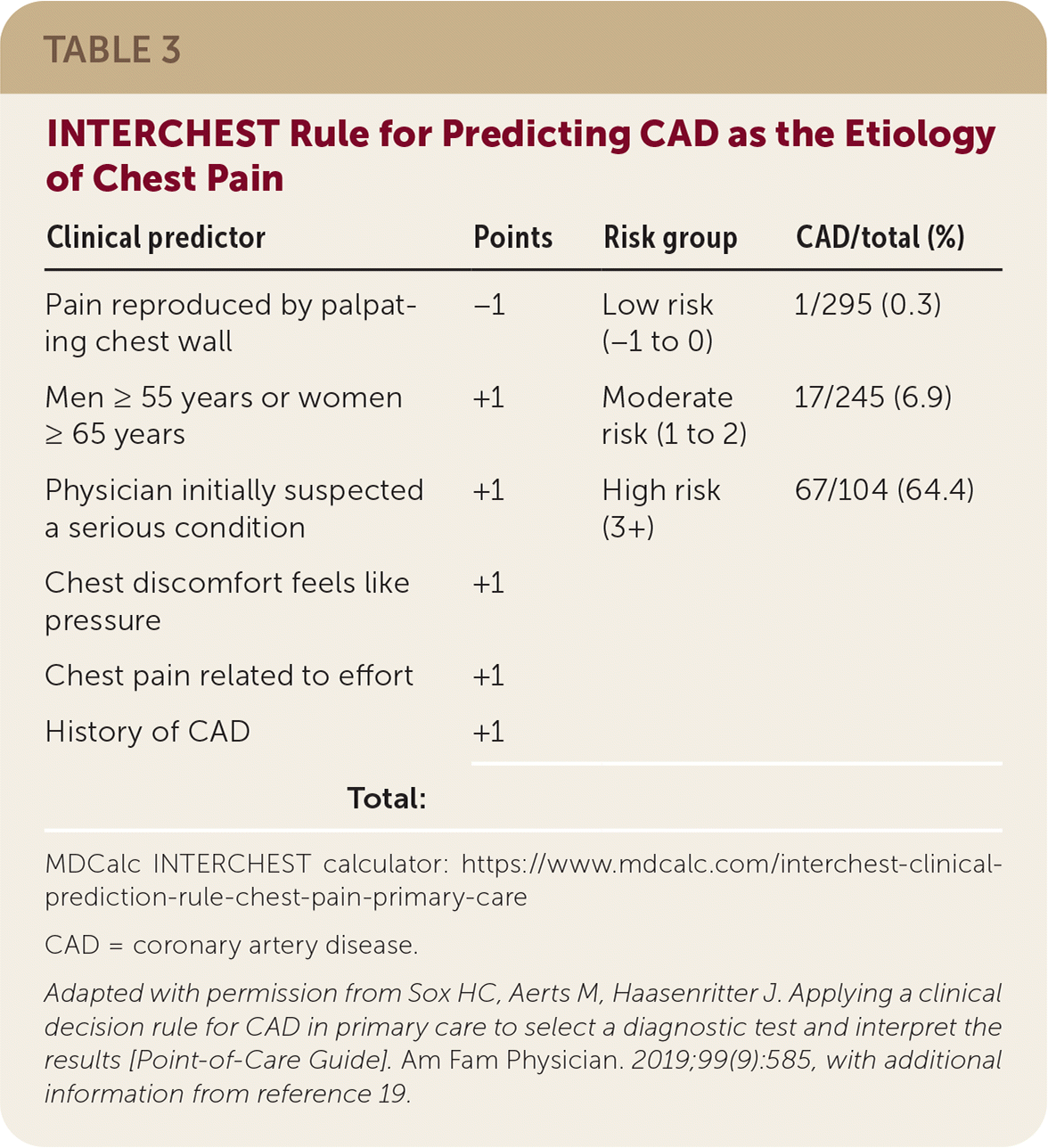
| Clinical predictor | Points | Risk group | CAD/total (%) |
|---|---|---|---|
| Pain reproduced by palpating chest wall | −1 | Low risk (−1 to 0) | 1/295 (0.3) |
| Men ≥ 55 years or women ≥ 65 years | +1 | Moderate risk (1 to 2) | 17/245 (6.9) |
| Physician initially suspected a serious condition | +1 | High risk (3+) | 67/104 (64.4) |
| Chest discomfort feels like pressure | +1 | ||
| Chest pain related to effort | +1 | ||
| History of CAD | +1 | ||
| Total: |
MDCalc INTERCHEST calculator: https://www.mdcalc.com/interchest-clinical-prediction-rule-chest-pain-primary-care
CAD = coronary artery disease.
Adapted with permission from Sox HC, Aerts M, Haasenritter J. Applying a clinical decision rule for CAD in primary care to select a diagnostic test and interpret the results [Point-of-Care Guide]. Am Fam Physician. 2019;99(9):585, with additional information from reference 19.
Because history alone usually cannot determine whether a patient is actively experiencing cardiac ischemia, a 12-lead ECG should be performed on all patients in whom cardiac ischemia is suspected.21 ECG findings that increase the likelihood of ACS include ST segment elevation, new-onset left bundle branch block, presence of Q waves, or new T-wave inversions.22 Similar ECG findings may be observed in non-ACS conditions, including acute pericarditis and left ventricular hypertrophy. Patients with suspicion of ACS based on clinical presentation (history, physical examination, risk factors) with changes seen on ECG should be transported immediately to the emergency department.16
For patients with chest pain not requiring immediate referral who have a low to intermediate pretest probability of CAD, exercise stress testing should be considered.23 Adding myocardial perfusion or echocardiography to the stress test increases test accuracy with a negative predictive value for acute myocardial infarction and cardiac death of 98%.24 Evaluating with coronary computed tomography angiography (CCTA) decreases the number of nonfatal acute myocardial infarctions 25 and is moderately more accurate than stress ECG in ruling out CAD in patients with chest pain (positive likelihood ratio [LR+] = 5.62; negative likelihood ratio [LR–] = 0.05).26 As a result, CCTA is becoming a first-line test for patients presenting with chest pain in the emergency department and should be a consideration for family physicians evaluating and managing patients with stable chest pain in the office. Cardiac magnetic resonance imaging may be useful in the evaluation of typical angina. For the evaluation of acute chest pain, cardiac magnetic resonance imaging is comparable to angiography in mortality at one year; however, it results in less need for invasive angiography and fewer subsequent revascularization procedures.27 Cost is a barrier to the use of CCTA and cardiac magnetic resonance imaging. According to Healthcare Bluebook, the cost of an exercise stress test is $171, whereas the price of CCTA is $667 and cardiac magnetic resonance imaging angiography is $1,075.28 Consideration should also be given to the harms of radiation and contrast exposure from CCTA.
Other Diagnostic Considerations
If the initial evaluation indicates that ACS is less likely or the diagnostic evaluation for ACS in higher-risk patients is negative, other non-ACS conditions that may cause symptoms similar to coronary ischemia should be considered (Table 4). Understanding the presentation of these common conditions with the clinical impression will help lead to a correct diagnosis.
TABLE 4. Nonischemic Causes of Chest Pain
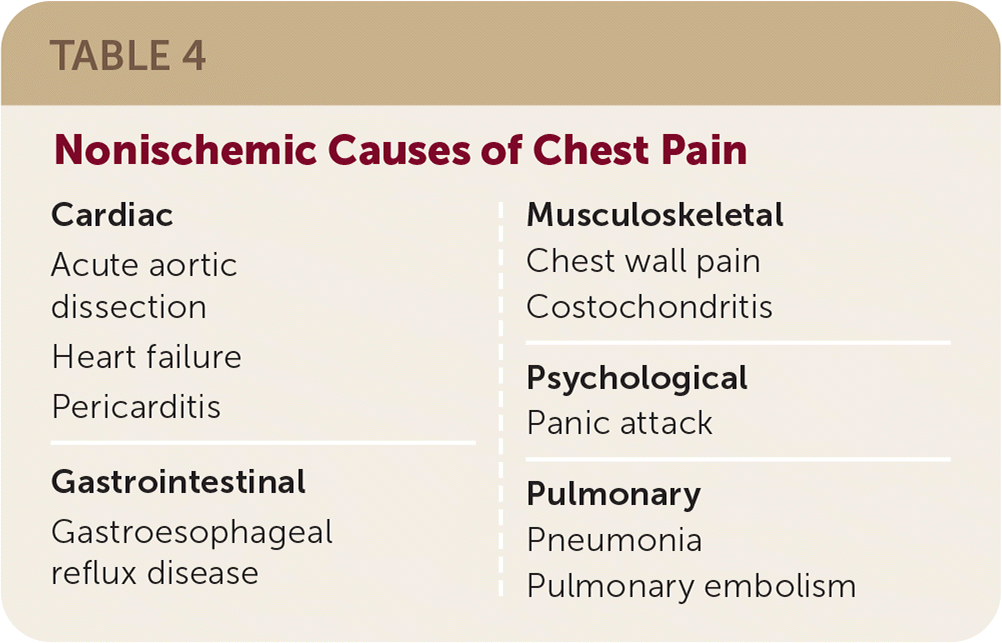
| Cardiac Acute aortic dissection Heart failure Pericarditis Gastrointestinal Gastroesophageal reflux disease Musculoskeletal Chest wall pain Costochondritis Psychological Panic attack Pulmonary Pneumonia Pulmonary embolism |
CHEST WALL PAIN
Chest wall pain is the most common cause of chest pain in the outpatient setting, accounting for 33% to 50% of chest pain.29 One prospective cohort study identified four clinical factors that predict a final diagnosis of chest wall pain in patients presenting to the primary care office with chest pain: localized muscle tension, stinging pain, pain reproducible by palpation, and the absence of a cough. In a study population with a prevalence of chest wall pain of 47%, patients with at least two of these findings had a 77% likelihood of chest wall pain as the cause of their discomfort (LR+ = 3.02), and those with none or one of the findings had only an 18% likelihood (LR− = 0.47).29
COSTOCHONDRITIS
Often considered a subset of chest wall pain, costochondritis is a self-limited condition characterized by pain that is reproducible with palpation in the parasternal costochondral joints. Costochondritis is a clinical diagnosis and does not require specific diagnostic testing in the absence of concomitant cardiopulmonary symptoms or risk factors.30
GASTROESOPHAGEAL REFLUX DISEASE
Classic symptoms of gastroesophageal reflux disease (GERD) include a burning retrosternal pain, acid regurgitation, and a sour or bitter taste in the mouth.31,32 There are no useful physical examination maneuvers or standard tests to establish the diagnosis or to support or rule it out. A one-week trial of a high-dose proton pump inhibitor is modestly sensitive and specific for GERD, with a 50% reduction in reflux symptoms being moderately accurate for a final diagnosis of GERD (LR+ = 5.5; LR− = 0.24).33 ACS symptoms can often be mistaken for those of GERD; if clinical suspicion is high for ACS, an ECG should be obtained.
Panic Disorder and Anxiety State
Panic disorder and anxiety states are common. One in four people with a panic attack will have chest pain and shortness of breath.34 Yet, concomitant panic disorder and chest pain are often not recognized, leading to more testing, follow-up, and higher costs of care.34 A moderately accurate assessment for detecting panic disorder is had by asking the following validated screening question: “In the past four weeks, have you had an anxiety attack (suddenly feeling fear or panic)?” This question is good at supporting a diagnosis of panic disorder when patients answer yes (LR+ = 4.2) and is good at ruling it out when the answer is no (LR− = 0.09).35
Less Common, but Important, Diagnostic Considerations
PERICARDITIS
Pericarditis manifests as a clinical triad of pleuritic chest pain, a pericardial friction rub, and diffuse ECG ST–T-wave changes often preceded by a viral illness.36 Acute pericarditis should be considered in patients presenting with new-onset chest pain that increases with inspiration or when reclining and is lessened by leaning forward.36 ECG usually demonstrates diffuse ST segment elevation and PR interval depression.
PNEUMONIA
Common symptoms of pneumonia include fever, chills, productive cough, and pleuritic chest pain.37 Egophony (LR+ = 8.6), dullness to percussion of the posterior thorax (LR+ = 4.3), and respiratory rate greater than 20 breaths per minute (LR+ = 3.5) are suggestive of pneumonia.38 Normal temperature, heart rate, and respiratory rate with a normal pulmonary examination rules out pneumonia (LR− = 0.10).39 Chest radiography can assist in the diagnosis of pneumonia; however, a Cochrane review suggests that routine chest radiography does not affect outcomes in patients who present with signs of lower respiratory tract infection.40
HEART FAILURE
Most patients with heart failure present with dyspnea on exertion, although some will present with chest pain.41 Clinical impression is predictive of heart failure (LR+ = 9.9; LR− = 0.65), as is pulmonary edema on chest radiography (LR+ = 11.0).41 Patients with acute dyspnea and one or more of the MICE criteria (Male sex, history of myocardial Infarction, basal lung Crepitations, and ankle Edema) likely have heart failure and should be evaluated with echocardiography.42,43
PULMONARY EMBOLISM
Diagnosing PE in the office is challenging because its presentation is highly variable. Although dyspnea, tachycardia, and/or chest pain are present in 97% of those diagnosed with PE, no single clinical feature effectively supports or rules out its diagnosis.44 Risk of PE can be estimated by using a validated clinical decision rule, such as the Wells criteria (Table 5).45 Patients at moderate or higher risk should undergo additional testing with a d-dimer assay, ventilation-perfusion scan, or helical computed tomography of the pulmonary arteries.45 The Pulmonary Embolism Rule-out Criteria were developed to specifically rule out PE in the primary care setting.46 Patients meeting all eight criteria (50 years or younger, heart rate less than 100 beats per minute, oxygen saturation greater than 94%, no unilateral leg swelling, no hemoptysis, no surgery or trauma within four weeks, no previous deep venous thrombosis or PE, no oral hormone use) have a less than 1% likelihood of PE and thus do not need d-dimer testing or imaging.46,47
TABLE 5. Wells Clinical Prediction Rule for PE
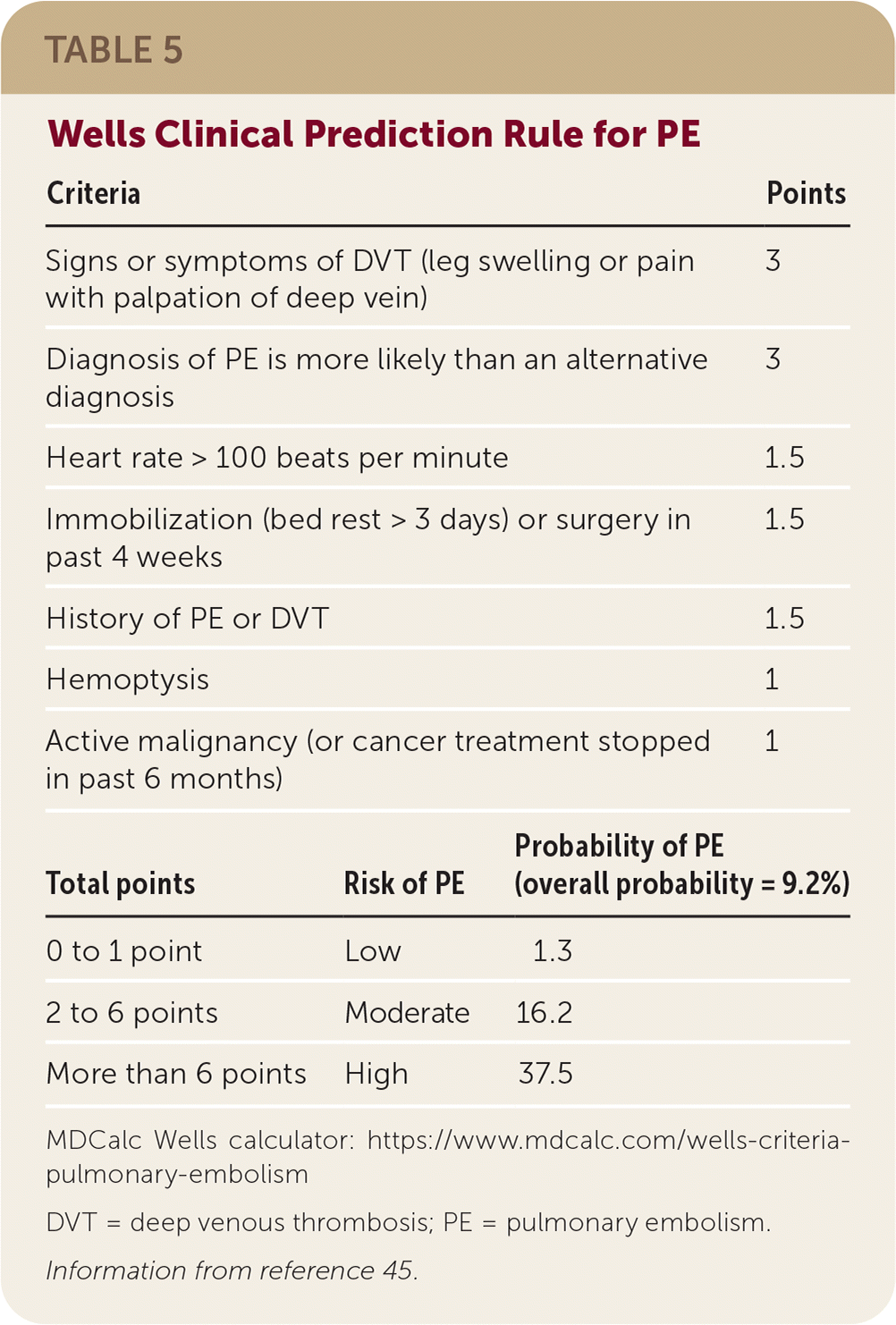
| Criteria | Points | ||
|---|---|---|---|
| Signs or symptoms of DVT (leg swelling or pain with palpation of deep vein) | 3 | ||
| Diagnosis of PE is more likely than an alternative diagnosis | 3 | ||
| Heart rate > 100 beats per minute | 1.5 | ||
| Immobilization (bed rest > 3 days) or surgery in past 4 weeks | 1.5 | ||
| History of PE or DVT | 1.5 | ||
| Hemoptysis | 1 | ||
| Active malignancy (or cancer treatment stopped in past 6 months) | 1 | ||
| Total points | Risk of PE | Probability of PE(overall probability = 9.2%) | |
| 0 to 1 point | Low | 1.3 | |
| 2 to 6 points | Moderate | 16.2 | |
| More than 6 points | High | 37.5 | |
MDCalc Wells calculator: https://www.mdcalc.com/wells-criteria-pulmonary-embolism
DVT = deep venous thrombosis; PE = pulmonary embolism.
Information from reference 45.
ACUTE THORACIC AORTIC DISSECTION
Patients with acute thoracic aortic dissection may present with chest or back pain.48 History and physical examination are only modestly useful for supporting or ruling out the diagnosis; acute chest or back pain and a pulse differential in the upper extremities modestly increases the likelihood of an acute thoracic aortic dissection (LR+ = 5.3).49 Clinical suspicion for thoracic dissection warrants immediate referral to the emergency department.
This article updates previous articles on this topic by McConaghy and Oza50 and Cayley.51
Data Sources: A PubMed search was completed using the key terms chest pain, chest pain evaluation, diagnosis, clinical decision rule, differential diagnosis, acute coronary syndrome, and angina. The search included meta-analyses, reviews, randomized controlled trials, point-of-care guides, and clinical trials. We also searched the Cochrane Database of Systematic Reviews, the National Guideline Clearinghouse, Essential Evidence Plus, Database of Abstracts of Reviews of Effects, and Agency for Healthcare Research and Quality Evidence Reports. Search date: literature search was completed on several occasions; last date was October 11, 2020.

