Dystocia (abnormally slow or protracted labor) accounts for 25% to 55% of primary cesarean deliveries. The latent phase of labor begins with onset of regular, painful contractions and continues until 6 cm of cervical dilation. Current recommendations are to avoid admission to labor and delivery during the latent phase, assuming maternal/fetal status is reassuring. The active phase begins at 6 cm. An arrested active phase is defined as more than four hours without cervical change despite rupture of membranes and adequate contractions and more than six hours of no cervical change without adequate contractions. Managing a protracted active phase includes oxytocin augmentation with or without amniotomy. The second stage of labor begins at complete cervical dilation and continues to delivery. This stage is considered protracted if it lasts three hours or more in nulliparous patients without an epidural or four hours or more in nulliparous patients with an epidural. Primary interventions for a protracted second stage include use of oxytocin and manual rotation if the fetus is in the occiput posterior position. When contractions or pushing is inadequate, vacuum or forceps delivery may be needed. Effective measures for preventing dystocia and subsequent cesarean delivery include avoiding admission during latent labor, providing cervical ripening agents for induction in patients with an unfavorable cervix, encouraging the use of continuous labor support (e.g., a doula), walking or upright positioning in the first stage, and not diagnosing failed induction during the latent phase until oxytocin has been given for 12 to 18 hours after membrane rupture. Elective induction at 39 weeks' gestation in low-risk nulliparous patients may reduce the risk of cesarean delivery.
Labor dystocia refers to abnormally slow or protracted labor. It may be diagnosed in the first stage of labor (onset of contractions until complete cervical dilation) or the second stage of labor (complete cervical dilation until delivery). Dystocia is responsible for most cesarean deliveries. First- and second-stage arrest of labor may account for as many as 15% to 30% and 10% to 25%, respectively, of all primary cesarean deliveries.1 Table 1 defines protracted and arrested labor for each stage of labor.1–3
WHAT'S NEW ON THIS TOPIC
Labor Dystocia
A U.S. cohort study of 62,415 patients between 2002 and 2008 showed that the rate of cervical dilation in the active phase of labor was slower than that described by Friedman and that the transition point from latent to active labor was 6 cm rather than 4 cm.
A Cochrane review encompassing more than 15,000 patients reported that continuous labor support shortens labor by 35 minutes on average, reduces the risk of cesarean or operative vaginal delivery, and improves patients' satisfaction with their labor experience.
A 2017 meta-analysis of intravenous fluid administration showed that an infusion rate of 250 mL per hour in nulliparous patients may reduce the duration of labor and the cesarean delivery rate compared with 125 mL per hour.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
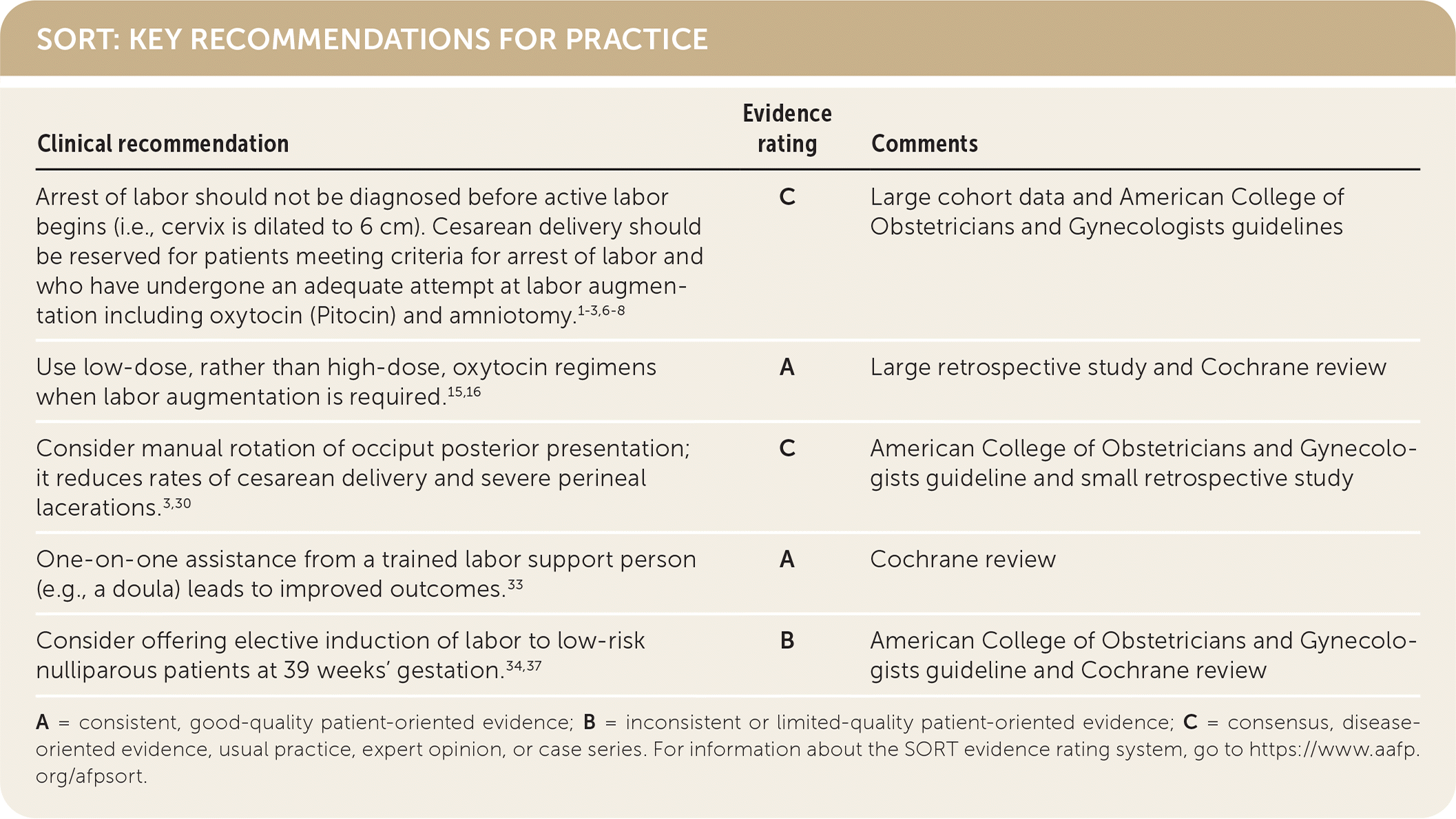
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
TABLE 1. Current Definitions of Labor Dystocia
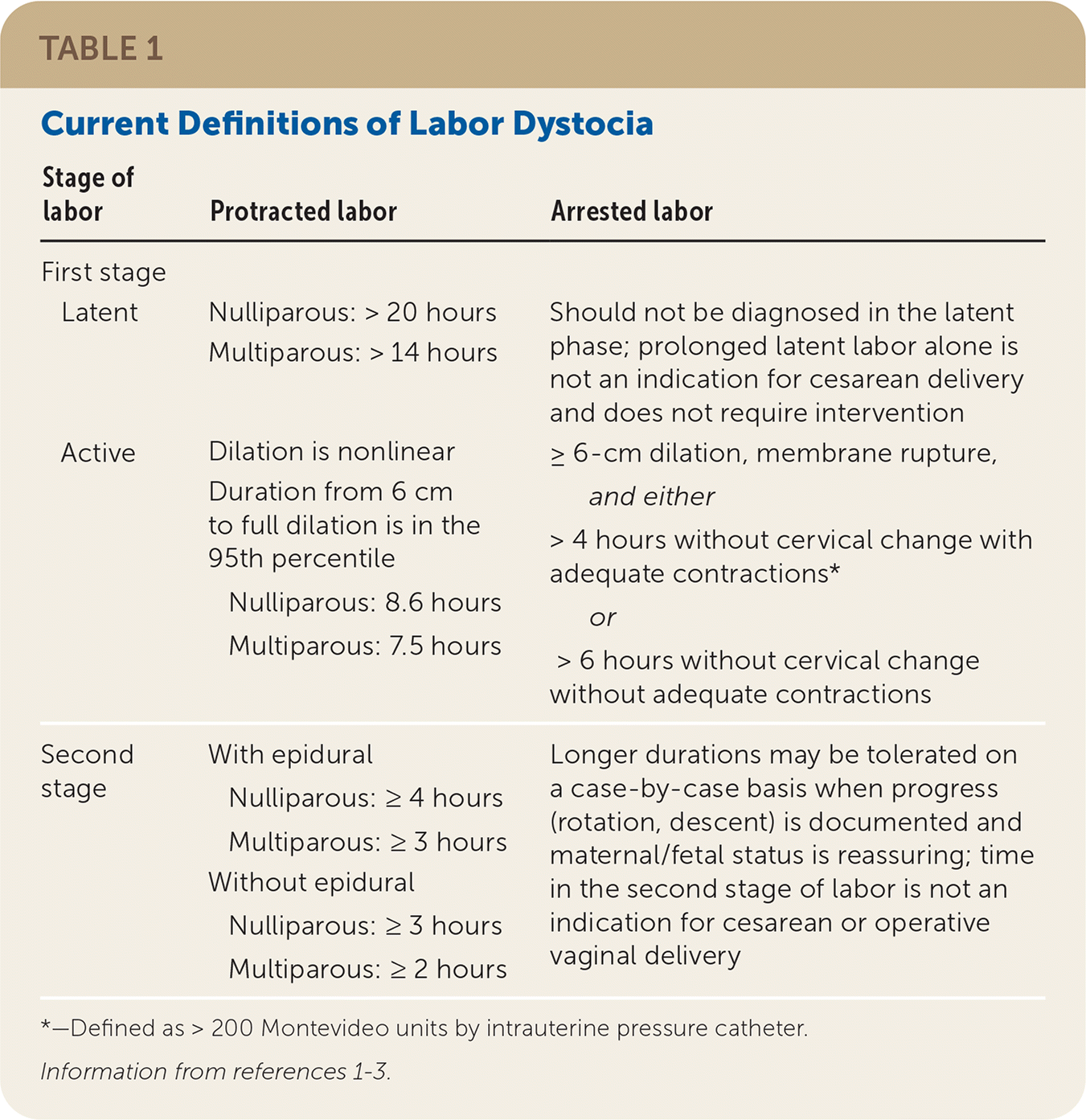
| Stage of labor | Protracted labor | Arrested labor |
|---|---|---|
| First stage | ||
| Latent | Nulliparous: > 20 hours Multiparous: > 14 hours | Should not be diagnosed in the latent phase; prolonged latent labor alone is not an indication for cesarean delivery and does not require intervention |
| Active | Dilation is nonlinear Duration from 6 cm to full dilation is in the 95th percentile Nulliparous: 8.6 hours Multiparous: 7.5 hours | ≥ 6-cm dilation, membrane rupture, and either > 4 hours without cervical change with adequate contractions* or > 6 hours without cervical change without adequate contractions |
| Second stage | With epidural Nulliparous: ≥ 4 hours Multiparous: ≥ 3 hours Without epidural Nulliparous: ≥ 3 hours Multiparous: ≥ 2 hours | Longer durations may be tolerated on a case-by-case basis when progress (rotation, descent) is documented and maternal/fetal status is reassuring; time in the second stage of labor is not an indication for cesarean or operative vaginal delivery |
*—Defined as > 200 Montevideo units by intrauterine pressure catheter.
First Stage of Labor
The first stage of labor includes latent labor (beginning with the onset of regular, painful contractions) and active labor (beginning when the rate of cervical dilation accelerates).
DYSTOCIA DEFINITION
Traditionally, labor progress was defined as normal or abnormal based on data from the Friedman labor curve and was dependent on whether the patient was in latent or active labor.3–5 A more recent study of 62,415 pregnant people in U.S. hospitals between 2002 and 2008 showed that the rate of dilation in the active phase of labor was slower than that described by Friedman and that the transition point from latent to active labor was 6 cm rather than 4 cm.2 As a result, and in an effort to reduce cesarean deliveries performed because of slow labor progress before 6 cm of dilation, the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine revised their definitions and recommendations for the management of labor dystocia and arrest.3
MANAGEMENT
In the absence of other indications for delivery, prolonged latent labor in nulliparous patients generally does not require intervention as long as cervical dilation progresses. Specifically, it is not an indication for cesarean delivery because most patients with prolonged latent labor will eventually enter into active labor or will stop contracting.1,3
Arrest of labor should not be diagnosed before active labor begins (i.e., the cervix is dilated to 6 cm).1–3 Cesarean delivery should be reserved for patients meeting criteria for arrest of labor and who have undergone an adequate attempt at labor augmentation, including administration of oxytocin (Pitocin; titrated when possible until intrauterine pressure catheter shows contraction strength of at least 200 Montevideo units in 10 minutes) and amniotomy.6–8
In normally progressing, spontaneous labor, amniotomy alone does not shorten labor or reduce the cesarean delivery rate.9 When added to oxytocin in patients with a prolonged first stage, amniotomy shortens labor but does not reduce the cesarean delivery rate.10 When combined and used as a method of dystocia prevention, amniotomy and oxytocin are associated with only modest reductions in the cesarean delivery rate.10
Low-dose oxytocin regimens start at 0.5 to 2 mU per minute and increase by 1 to 2 mU per minute every 15 to 40 minutes. High-dose regimens start at 6 mU per minute and increase by 3 to 6 mU per minute every 15 to 40 minutes.11,12 Previous data suggested that high-dose regimens decreased the time to delivery11,13,14; however, a 2019 retrospective study of nulliparous patients did not show a faster time to delivery or reduction in the cesarean delivery rate when compared with low-dose regimens.15 A Cochrane review showed a decrease in uterine tachysystole (defined as more than five contractions in a 10-minute period, averaged over 30 minutes) with low-dose regimens compared with high-dose regimens, without a difference in the vaginal delivery rate within 24 hours or in the overall cesarean delivery rate.16 Given this evidence, physicians should use low-dose, rather than high-dose, oxytocin regimens when labor augmentation is required in nulliparous patients.
Other interventions that have been studied for arrest of labor in the first stage include intravenous fluids and maternal positioning. A meta-analysis of intravenous fluid administration showed that an infusion rate of 250 mL per hour in nulliparous patients may reduce the duration of labor and the cesarean delivery rate compared with 125 mL per hour.17 Walking and upright positioning, rather than supine positioning, also shorten the first stage of labor and reduce the cesarean delivery rate.18
Second Stage of Labor
The second stage of labor begins with complete cervical dilation and continues until delivery.
DYSTOCIA DEFINITION
In an update of definitions, guidelines from ACOG and the Society for Maternal-Fetal Medicine allow an additional one hour of pushing in the second stage of labor for nulliparous and multiparous patients before diagnosing second-stage dystocia and yet another hour for patients receiving epidural anesthesia.3 Still, the chance for spontaneous vaginal delivery decreases with each hour of second-stage labor. After three or more hours, only one in four nulliparous patients delivers spontaneously.3
Maternal risks from a prolonged second stage include increased rates of operative vaginal delivery, third- and fourth-degree perineal lacerations, chorioamnionitis, and postpartum hemorrhage. Fetal risks from a prolonged second stage include increased rates of shoulder dystocia, neonatal intensive care unit admission, and lower umbilical artery pH, although no appreciable difference in neonatal neurologic outcomes occurs.19
MANAGEMENT
Time in the second stage of labor is not an indication for operative delivery if maternal and fetal status are reassuring and progress is documented. Physicians should, however, consider or address factors that might be contributing to slow progress. The most common causes are inadequate pushing or contractions, fetal malposition, and cephalopelvic disproportion.20
For patients not receiving epidural anesthesia, maintaining an upright position during the second stage of labor reduces duration of this stage by about seven minutes and reduces rates of episiotomy and operative vaginal delivery, although there is no effect on the cesarean delivery rate.21 For patients receiving epidural anesthesia, no specific position has a clear advantage, and it is thus reasonable to allow these patients to push in the position they prefer.22 If contraction frequency or strength becomes inadequate, intravenous oxytocin may be administered or the rate increased if already initiated.12
Delaying maternal pushing, also known as laboring down, is another approach to consider. This allows for spontaneous descent of the fetal head to a lower station, without pushing, after full cervical dilation is obtained. According to a Cochrane review, laboring down results in a longer (by 56 minutes) second stage of labor overall but a shorter duration of active pushing (by 19 minutes), compared with immediate pushing. It is also associated with a small increase in the rate of spontaneous vaginal delivery (risk ratio [RR] = 1.07; 95% CI, 1.02 to 1.11).23 However, a randomized controlled trial of 2,414 nulliparous patients with epidural anesthesia compared immediate pushing after complete cervical dilation with pushing after a 60-minute delay.24 The rate of spontaneous vaginal delivery was no different between the two groups, although patients who immediately pushed had a shorter total second stage (−31.8 minutes; 95% CI, −36.7 to −26.9), a longer duration of active pushing (9.2 minutes longer; 95% CI, 5.8 to 12.6), less chorioamnionitis, and fewer postpartum hemorrhages.24 ACOG supports immediate pushing at the start of the second stage in nulliparous patients with epidural anesthesia.25 Additionally, patients should be encouraged to push in the manner in which they feel most comfortable; closed glottis pushing does not reduce second-stage duration compared with spontaneous pushing.23
When contractions or pushing is inadequate, operative vaginal delivery with forceps or vacuum can also be used and is a safe alternative to cesarean delivery if the physician is well trained in the procedure.1 There is no difference in neonatal death or intracerebral hemorrhage between operative vaginal delivery and unplanned cesarean delivery in nulliparous patients.26 Epidural anesthesia increases the likelihood of operative vaginal delivery but not cesarean delivery and adds an average of 14 minutes to the second stage of labor.27,28
Malposition of the fetus, particularly occiput posterior presentation, is a common contributor to second-stage dystocia. Fetal head position can often be determined by digital examination, although intrapartum ultrasonography is more accurate.3,20,29 In the case of persistent occiput transverse or occiput posterior positioning, manually rotating to an occiput anterior position may be considered20 (Figure 1). Manual rotation in this setting reduces the rates of cesarean delivery and severe perineal laceration, but it may increase cervical lacerations.3,30 Maternal repositioning to hands and knees is often tried as an approach to occiput posterior presentation, but it has not been shown to consistently rotate the fetus to an occiput anterior position or to reduce the need for operative delivery.31
FIGURE 1.
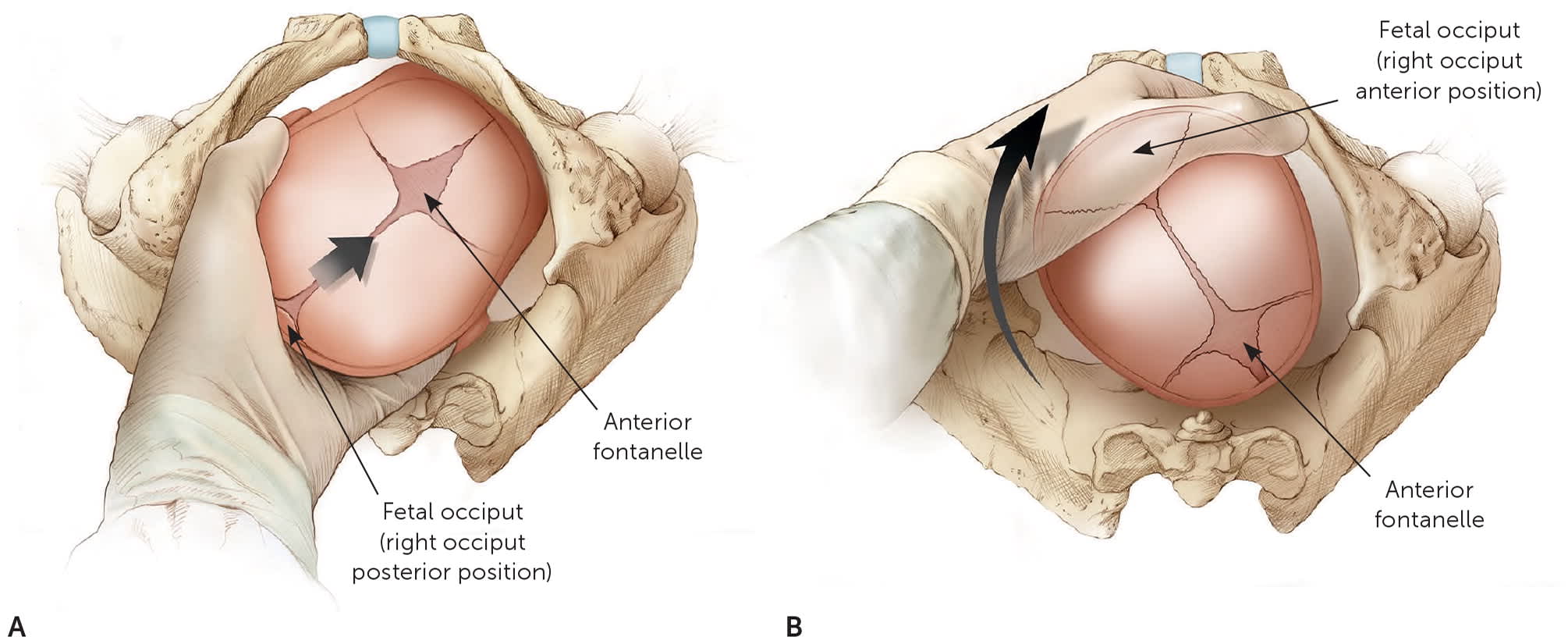
Manual rotation of a fetus from the occiput posterior position to the occiput anterior position. (A) The physician's hand is placed palm upward over the fetal occiput in the vagina. (B) The hand serves as a wedge to flex the fetal head while the fingers exert a rotating force to bring the occiput to the anterior position.
Illustration by Christy Krames
Prevention
Potential interventions to prevent labor dystocia and associated cesarean delivery in nulliparous patients include avoidance of admission to labor and delivery during latent labor; continuous labor support, such as with a doula; appropriate management of labor induction and use of cervical ripening agents; and adequate time and intervention before considering cesarean delivery for a “failed” induction (Table 2).1,3,11,25,32–41
TABLE 2. Selected Interventions to Prevent Labor Dystocia
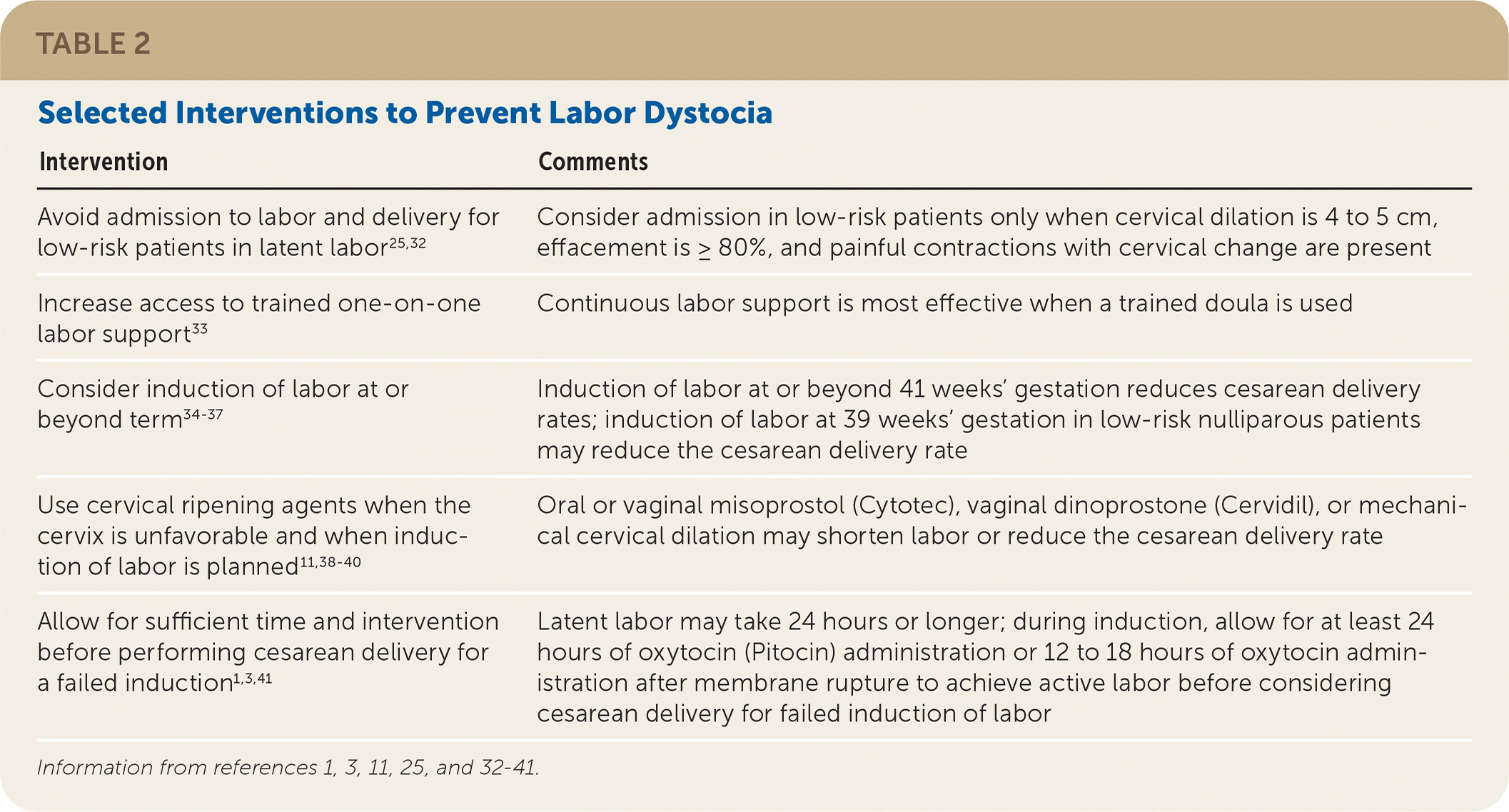
| Intervention | Comments |
|---|---|
| Avoid admission to labor and delivery for low-risk patients in latent labor25,32 | Consider admission in low-risk patients only when cervical dilation is 4 to 5 cm, effacement is ≥ 80%, and painful contractions with cervical change are present |
| Increase access to trained one-on-one labor support33 | Continuous labor support is most effective when a trained doula is used |
| Consider induction of labor at or beyond term34–37 | Induction of labor at or beyond 41 weeks' gestation reduces cesarean delivery rates; induction of labor at 39 weeks' gestation in low-risk nulliparous patients may reduce the cesarean delivery rate |
| Use cervical ripening agents when the cervix is unfavorable and when induction of labor is planned 11,38–40 | Oral or vaginal misoprostol (Cytotec), vaginal dinoprostone (Cervidil), or mechanical cervical dilation may shorten labor or reduce the cesarean delivery rate |
| Allow for sufficient time and intervention before performing cesarean delivery for a failed induction1,3,41 | Latent labor may take 24 hours or longer; during induction, allow for at least 24 hours of oxytocin (Pitocin) administration or 12 to 18 hours of oxytocin administration after membrane rupture to achieve active labor before considering cesarean delivery for failed induction of labor |
AVOIDING ADMISSION DURING LATENT LABOR
Admission to labor and delivery for patients in latent labor has been shown to increase rates of diagnosing active labor arrest along with interventions, including cesarean delivery.1,25 The Society of Obstetricians and Gynaecologists of Canada recommends not admitting low-risk patients to labor and delivery until cervical dilation is at least 4 cm.42 Similarly, the National Partnership for Maternal Safety recommends admission for low-risk patients only after significant dilation (4 to 5 cm), effacement (80% or more), documented cervical change, and painful contractions are present.32 ACOG also supports expectant management and delayed admission for patients in latent labor until cervical dilation is 4 to 6 cm, assuming maternal and fetal status are reassuring.25
LABOR SUPPORT
Multiple studies have demonstrated that one-on-one assistance from a trained labor support person (e.g., a doula) leads to improved outcomes. A Cochrane review encompassing more than 15,000 patients reported that continuous labor support shortens labor by 0.58 hours on average and reduces the risk of cesarean delivery (RR = 0.78) or operative vaginal delivery (RR = 0.90). Patients were also more satisfied with their labor experience.33
MANAGEMENT OF LABOR INDUCTION
Induction rates increased from 22% in 2006 to 27% in 2018, and appropriate management of induction at or beyond term is important because it can affect cesarean delivery and stillbirth rates.11,43 In the past, there had been concern that induction of labor could increase the rate of labor arrest and subsequent cesarean delivery, particularly with cervical ripening in nulliparous patients.44 More recent evidence, however, has challenged this concern. A 2018 Cochrane review reported a lower rate of cesarean deliveries in patients undergoing induction at or beyond term (RR = 0.92) when compared with expectant management.34 Similarly, a 2018 multicenter randomized controlled trial showed that, compared with expectant management, labor induction between 39 and 39 4/7 weeks' gestation in low-risk nulliparous patients reduces, not increases, the cesarean delivery rate (18.6% vs. 22.2%; RR = 0.84; number needed to treat = 28), along with other improved outcomes.36 As a result of these newer data, ACOG and the Society for Maternal-Fetal Medicine advise that it is reasonable to offer elective induction to low-risk nulliparous patients at 39 weeks' gestation.37,45
When the cervix is not favorable for induction, traditionally defined as a Bishop score of 6 or less (see calculator at https://www.mdcalc.com/bishop-score-vaginal-delivery-induction-labor), cervical ripening is used before starting induction. Mechanical dilation with a transcervical Foley catheter (filled with 30 to 80 mL of saline) or a cervical ripening balloon is a commonly used method and is associated with a reduction in cesarean delivery rates and duration of labor compared with the use of oxytocin alone.11,38
Oral, vaginal, or sublingual misoprostol (Cytotec) can also be used for cervical ripening.11 A Cochrane review reported a reduction in cesarean delivery rates when oral misoprostol was used for cervical ripening, rather than induction with intravenous oxytocin alone.39 Similarly, vaginal dinoprostone (Cervidil) and vaginal misoprostol both reduce cesarean delivery rates and shorten labor compared with placebo ripening agents, but they were not compared directly with intravenous oxytocin alone in the most recent Cochrane review.40 Two randomized controlled trials have shown that the combination of vaginal misoprostol with transcervical Foley catheter shortens labor compared with misoprostol alone.46,47
It should be noted, however, that dinoprostone and misoprostol are prostaglandins. Studies cited in a 2019 Cochrane review indicate that a higher rate of uterine tachysystole occurs with prostaglandins (3% with fetal heart rate changes, 5.9% without fetal heart rate changes) compared with only about 1% with mechanical ripening methods.11,38
FAILED INDUCTION
Finally, when a patient is unable to reach the active stage of labor despite appropriate interventions, a cesarean delivery may be performed for failed induction of labor. There is, however, no universal definition of what length of labor constitutes failure. A review of 10,000 nulliparous patients found that 96% had entered active labor within 15 hours after membrane rupture and oxytocin administration.41 ACOG recommends allowing longer (24 hours or more) durations of the latent phase and avoiding cesarean delivery for failed induction in the latent phase until oxytocin has been given for 12 to 18 hours after membrane rupture.3
This article updates a previous article on this topic by Shields, et al.20
Data Sources: The authors performed PubMed searches for systematic reviews, meta-analyses, and randomized controlled trials since the year 2000. Relevant guidelines from the Society for Maternal-Fetal Medicine, American College of Obstetricians and Gynecologists, Society of Obstetricians and Gynaecologists of Canada, and Royal College of Obstetricians and Gynaecologists were reviewed. The Cochrane Database of Systematic Reviews was searched. An Essential Evidence Plus summary of patient-oriented evidence that matters was reviewed. Individual references were reviewed from the bibliographies of other specialty guidelines with relevant articles reviewed in full text. Search dates: December 2019 and August 2020.
The authors thank the medical librarians at John Peter Smith Hospital for their help with comprehensive literature searches.

