A patient's sense of empowerment and control is most predictive of maternal satisfaction with childbirth. Analgesia during labor greatly affects this experience. Individual patient priorities for labor pain management should be explored as part of routine prenatal care. Continuous labor support, water immersion, and upright positioning in the first stage of labor are associated with decreased use of pharmacologic analgesia. Despite the increased risk of adverse effects, self-administered inhaled nitrous oxide appears to be safe and effective for pain relief; however, its negative environmental impact as a greenhouse gas is a drawback. Evidence is lacking that any one opioid is superior in maximizing pain relief while minimizing adverse effects. Neuraxial anesthesia provides the most effective pharmacologic analgesia and is used in nearly three-fourths of labors in the United States. Neuraxial regional anesthesia is not associated with increased rates of cesarean delivery or assisted vaginal delivery, and it has only a small effect on the length of the second stage of labor. Epidural, spinal, combined spinal-epidural, and dural puncture epidural anesthesia are commonly used neuraxial techniques. Paracervical and pudendal blocks are safe and effective pain management options in specific circumstances. Both transversus abdominis plane block and subcutaneous wound infiltration with local anesthetic can decrease the use of postoperative analgesia. Patients with opioid use disorder require individualized pain management plans throughout perinatal care, and judicious opioid prescribing practices are encouraged for all patients.
Most patients will use some form of pharmacologic analgesia for pain management during labor. Although timeliness and adequacy of pain relief are important, they are not as predictive of maternal satisfaction with childbirth as the patient's sense of empowerment and control. Therefore, creating a supportive and nurturing environment should be a priority throughout the perinatal period.1
SORT: KEY RECOMMENDATIONS FOR PRACTICE
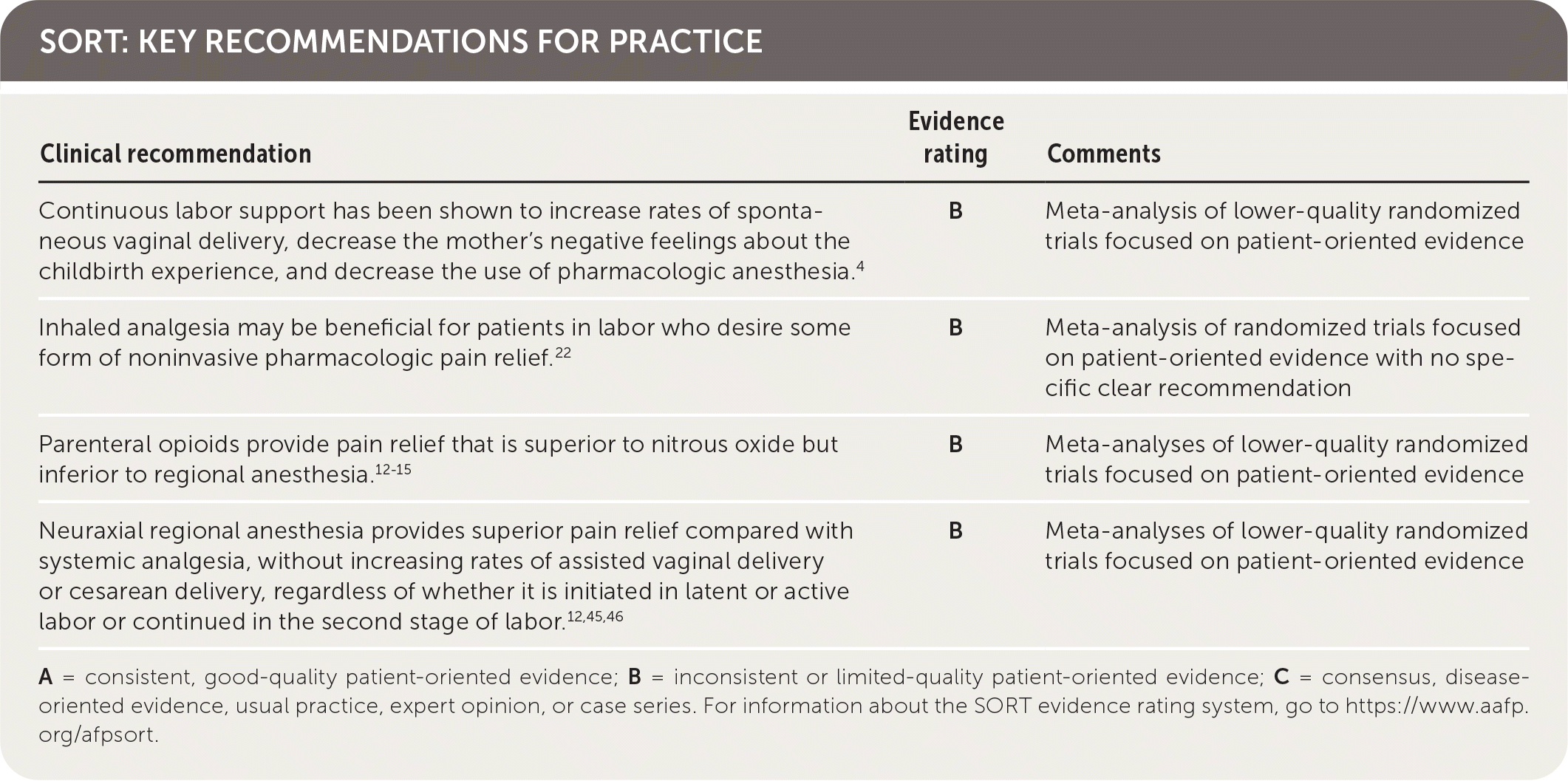
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Health care professionals providing prenatal care should discuss options for labor pain management during routine prenatal visits and explore the patient's priorities for the labor experience. The effectiveness and associated risks of the various pain management approaches should be reviewed. Additionally, physicians should be familiar with and educate their patients on the available analgesia options; these options may vary among hospitals and birth centers. Anesthesia consultation should be considered in complicated cases (Table 1).2
TABLE 1. Considerations for Anesthesia Consultation During Labor
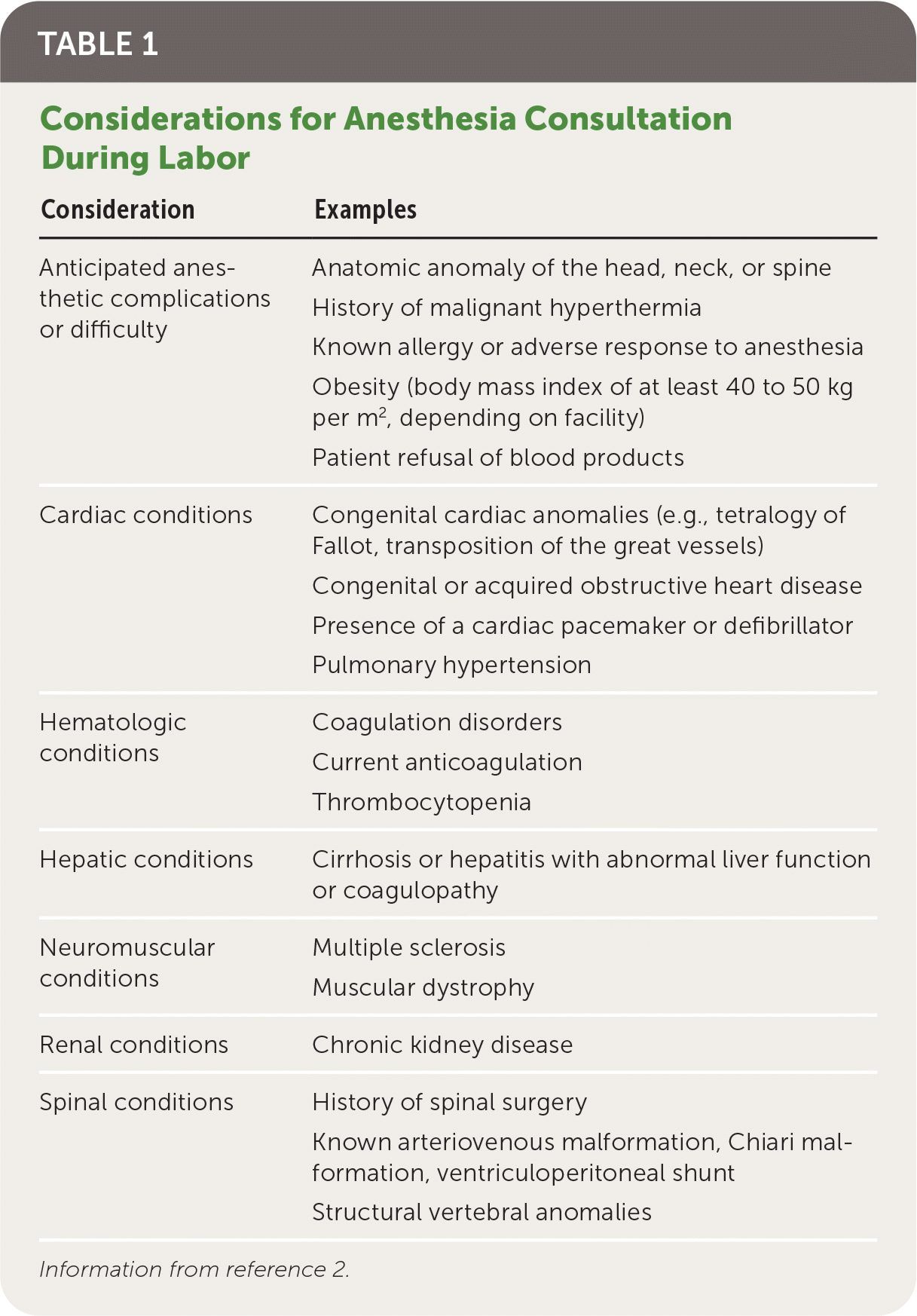
| Consideration | Examples |
|---|---|
| Anticipated anesthetic complications or difficulty | Anatomic anomaly of the head, neck, or spine History of malignant hyperthermia Known allergy or adverse response to anesthesia Obesity (body mass index of at least 40 to 50 kg per m2, depending on facility) Patient refusal of blood products |
| Cardiac conditions | Congenital cardiac anomalies (e.g., tetralogy of Fallot, transposition of the great vessels) Congenital or acquired obstructive heart disease Presence of a cardiac pacemaker or defibrillator Pulmonary hypertension |
| Hematologic conditions | Coagulation disorders Current anticoagulation Thrombocytopenia |
| Hepatic conditions | Cirrhosis or hepatitis with abnormal liver function or coagulopathy |
| Neuromuscular conditions | Multiple sclerosis Muscular dystrophy |
| Renal conditions | Chronic kidney disease |
| Spinal conditions | History of spinal surgery Known arteriovenous malformation, Chiari malformation, ventriculoperitoneal shunt Structural vertebral anomalies |
Information from reference 2.
Nonpharmacologic Approaches
Nonpharmacologic pain relief interventions are often used during labor, in place of or in addition to pharmacologic methods. Common nonpharmacologic interventions are summarized in Table 2.3
TABLE 2. Nonpharmacologic Options for Pain Management During Labor
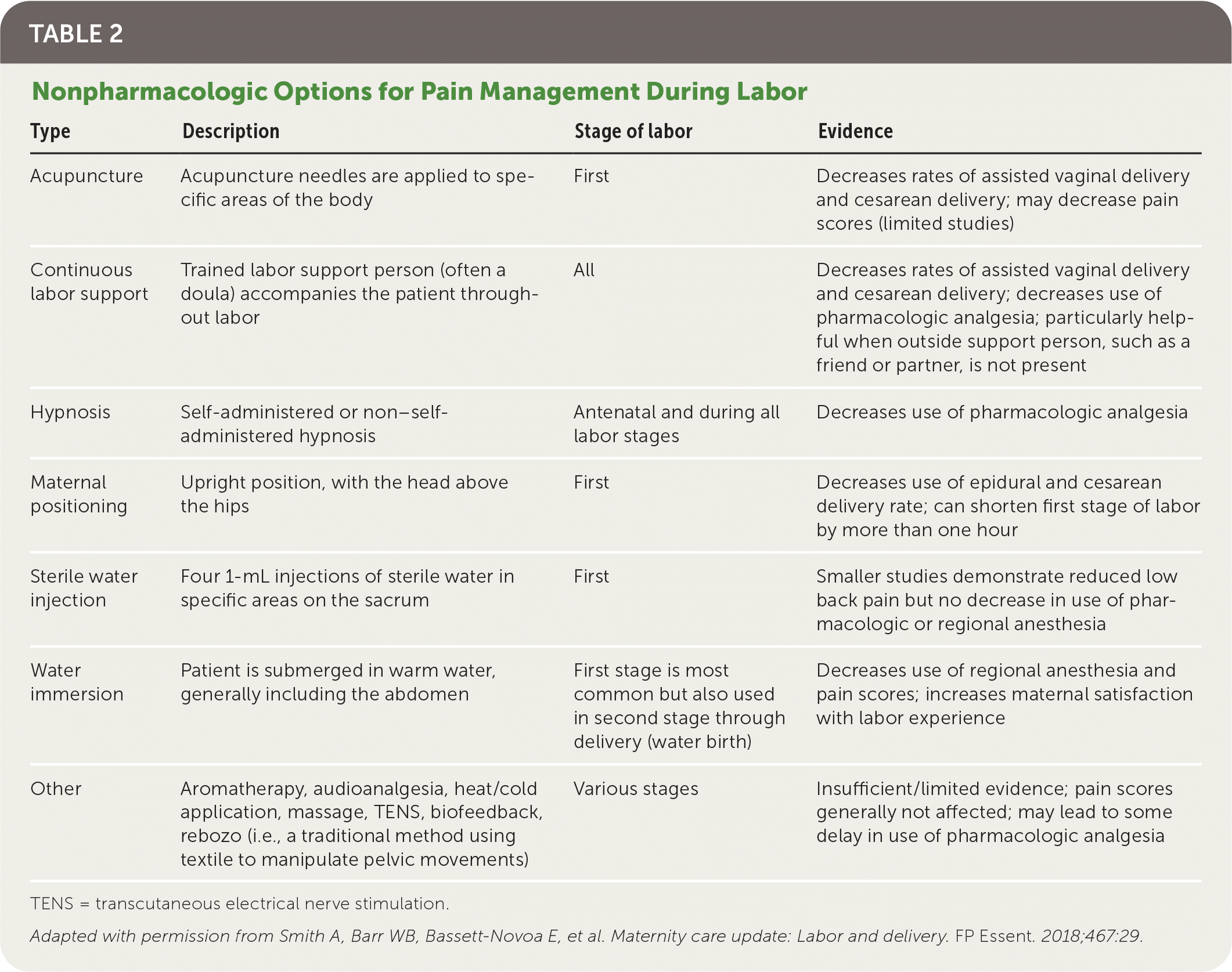
| Type | Description | Stage of labor | Evidence |
|---|---|---|---|
| Acupuncture | Acupuncture needles are applied to specific areas of the body | First | Decreases rates of assisted vaginal delivery and cesarean delivery; may decrease pain scores (limited studies) |
| Continuous labor support | Trained labor support person (often a doula) accompanies the patient throughout labor | All | Decreases rates of assisted vaginal delivery and cesarean delivery; decreases use of pharmacologic analgesia; particularly helpful when outside support person, such as a friend or partner, is not present |
| Hypnosis | Self-administered or non–self-administered hypnosis | Antenatal and during all labor stages | Decreases use of pharmacologic analgesia |
| Maternal positioning | Upright position, with the head above the hips | First | Decreases use of epidural and cesarean delivery rate; can shorten first stage of labor by more than one hour |
| Sterile water injection | Four 1-mL injections of sterile water in specific areas on the sacrum | First | Smaller studies demonstrate reduced low back pain but no decrease in use of pharmacologic or regional anesthesia |
| Water immersion | Patient is submerged in warm water, generally including the abdomen | First stage is most common but also used in second stage through delivery (water birth) | Decreases use of regional anesthesia and pain scores; increases maternal satisfaction with labor experience |
| Other | Aromatherapy, audioanalgesia, heat/cold application, massage, TENS, biofeedback, rebozo (i.e., a traditional method using textile to manipulate pelvic movements) | Various stages | Insufficient/limited evidence; pain scores generally not affected; may lead to some delay in use of pharmacologic analgesia |
TENS = transcutaneous electrical nerve stimulation.
Adapted with permission from Smith A, Barr WB, Bassett-Novoa E, et al. Maternity care update: Labor and delivery. FP Essent. 2018;467:29.
Continuous labor support, often from a doula, has been shown to increase rates of spontaneous vaginal delivery (relative risk [RR] = 1.08; 95% CI, 1.04 to 1.12), decrease the mother's negative feelings about the childbirth experience (RR = 0.69; 95% CI, 0.59 to 0.79), and decrease the use of pharmacologic analgesia (RR = 0.90; 95% CI, 0.84 to 0.96), especially when an outside support person, such as a friend or partner, is not present.4
Immersion in water during the first stage of labor can reduce the use of regional anesthesia (RR = 0.91; 95% CI, 0.83 to 0.99) with no difference in rates of spontaneous vaginal delivery, assisted vaginal delivery, postpartum hemorrhage, or third- or fourth-degree lacerations, and no difference in the duration of any stage of labor.5 Smaller studies suggest that injections of sterile water into the skin over the sacrum reduce labor-associated low back pain but do not meaningfully decrease the use of pharmacologic or regional anesthesia (RR = 0.86; 95% CI, 0.44 to 1.69).6
A Cochrane review demonstrated that patients in the first stage of labor who were in the upright position with the head above the hips, as opposed to recumbent positions, were less likely to use an epidural (RR = 0.81; 95% CI, 0.66 to 0.99) or have a cesarean delivery (RR = 0.71; 95% CI, 0.54 to 0.94).7 Another Cochrane review showed that upright positioning in the second stage of labor does not affect rates of cesarean delivery in patients who already have an epidural in place.8
Exercise ball maneuvers, lumbosacral massage, and warm showers during the first stage of labor may lower pain severity and delay or reduce the use of analgesic medications.9 Patients who use hypnosis during labor are less likely to use analgesic medications (RR = 0.73; 95% CI, 0.57 to 0.94), with no clear differences in spontaneous vaginal delivery rates, maternal satisfaction, or neonatal outcomes.10 Other interventions, such as aromatherapy and audioanalgesia, have not been shown to be beneficial.11
Systemic Pharmacologic Analgesia
TABLE 3. Systemic Analgesia Options for Pain Management During Labor
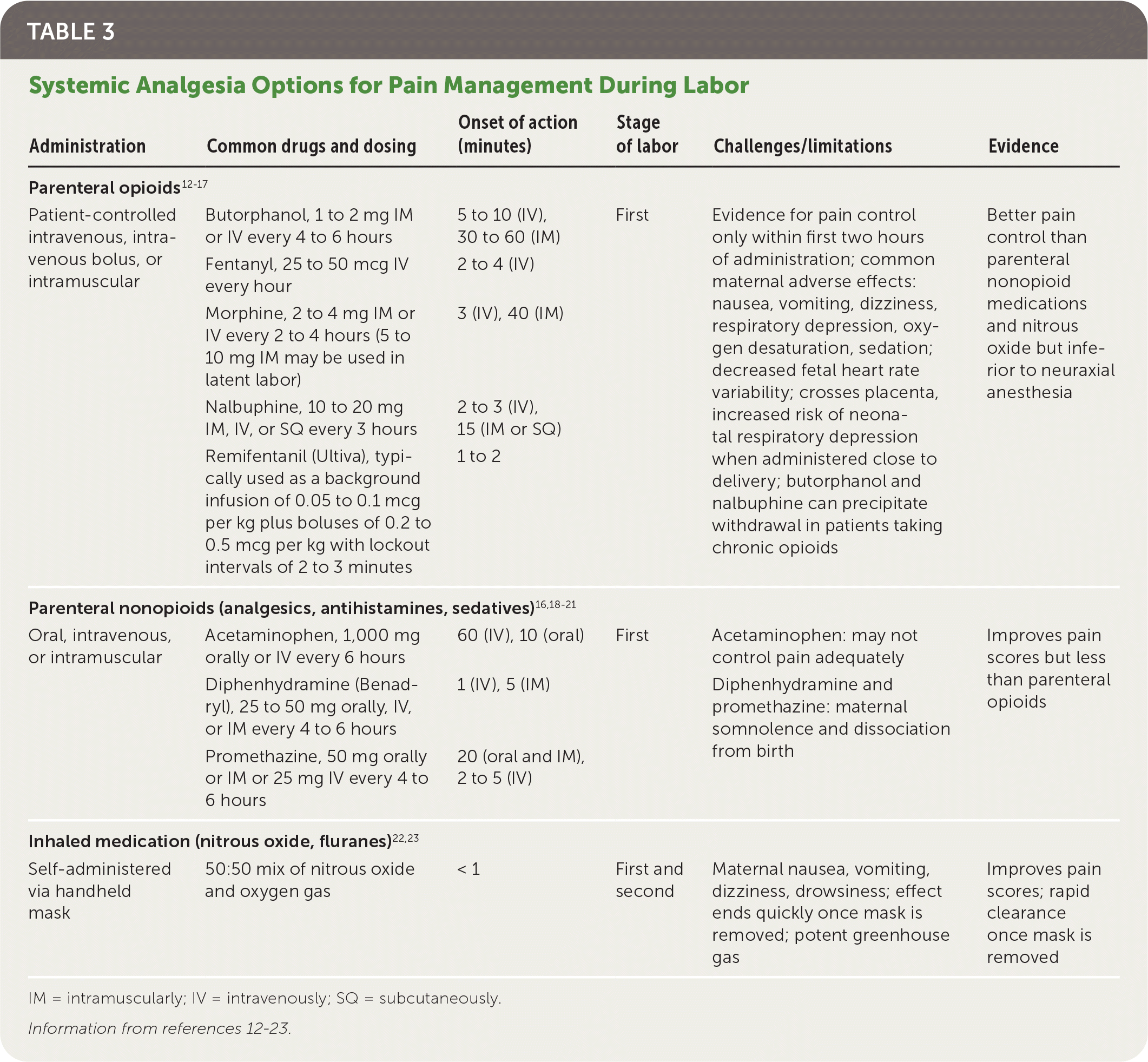
| Administration | Common drugs and dosing | Onset of action (minutes) | Stage of labor | Challenges/limitations | Evidence |
|---|---|---|---|---|---|
| Parenteral opioids12–17 | |||||
| Patient-controlled intravenous, intravenous bolus, or intramuscular | Butorphanol, 1 to 2 mg IM or IV every 4 to 6 hours | 5 to 10 (IV), 30 to 60 (IM) | First | Evidence for pain control only within first two hours of administration; common maternal adverse effects: nausea, vomiting, dizziness, respiratory depression, oxygen desaturation, sedation; decreased fetal heart rate variability; crosses placenta, increased risk of neonatal respiratory depression when administered close to delivery; butorphanol and nalbuphine can precipitate withdrawal in patients taking chronic opioids | Better pain control than parenteral nonopioid medications and nitrous oxide but inferior to neuraxial anesthesia |
| Fentanyl, 25 to 50 mcg IV every hour | 2 to 4 (IV) | ||||
| Morphine, 2 to 4 mg IM or IV every 2 to 4 hours (5 to 10 mg IM may be used in latent labor) | 3 (IV), 40 (IM) | ||||
| Nalbuphine, 10 to 20 mg IM, IV, or SQ every 3 hours | 2 to 3 (IV), 15 (IM or SQ) | ||||
| Remifentanil (Ultiva), typically used as a background infusion of 0.05 to 0.1 mcg per kg plus boluses of 0.2 to 0.5 mcg per kg with lockout intervals of 2 to 3 minutes | 1 to 2 | ||||
| Parenteral nonopioids (analgesics, antihistamines, sedatives)16,18–21 | |||||
| Oral, intravenous, or intramuscular | Acetaminophen, 1,000 mg orally or IV every 6 hours | 60 (IV), 10 (oral) | First | Acetaminophen: may not control pain adequately | Improves pain scores but less than parenteral opioids |
| Diphenhydramine (Benadryl), 25 to 50 mg orally, IV, or IM every 4 to 6 hours | 1 (IV), 5 (IM) | Diphenhydramine and promethazine: maternal somnolence and dissociation from birth | |||
| Promethazine, 50 mg orally or IM or 25 mg IV every 4 to 6 hours | 20 (oral and IM), 2 to 5 (IV) | ||||
| Inhaled medication (nitrous oxide, fluranes)22,23 | |||||
| Self-administered via handheld mask | 50:50 mix of nitrous oxide and oxygen gas | < 1 | First and second | Maternal nausea, vomiting, dizziness, drowsiness; effect ends quickly once mask is removed; potent greenhouse gas | Improves pain scores; rapid clearance once mask is removed |
IM = intramuscularly; IV = intravenously; SQ = subcutaneously.
NITROUS OXIDE
Inhaled analgesia may be beneficial for patients in labor who desire some form of noninvasive pharmacologic pain relief.22 Self-administered inhaled nitrous oxide may provide patients with a greater sense of control in labor and, with an onset of action within one minute, may be safe and effective for pain relief. A Cochrane review demonstrated that placebo or no treatment provides inferior pain relief compared with nitrous oxide in the first stage of labor (RR = 0.06; 95% CI, 0.01 to 0.34).22 However, nitrous oxide increases the risk of maternal adverse effects, such as nausea (RR = 43.10; 95% CI, 2.63 to 706.74), vomiting (RR = 9.05; 95% CI, 7.09 to 1,833.69), and drowsiness (RR = 77.59; 95% CI, 4.80 to 1,254.96).22 Because anesthetic gases contribute to greenhouse gas emissions, the environmental impact of routine use of nitrous oxide should also be considered.23
PARENTERAL PHARMACOLOGIC ANESTHESIA
Parenteral opioids provide pain relief that is superior to nitrous oxide but inferior to regional anesthesia, although maternal satisfaction is comparable between regional anesthesia and parenteral opioids.12–15 Most patients are moderately satisfied with pain control shortly after opioid administration; however, two-thirds of patients report the return of moderate to severe pain within two hours of administration.13 The use of parenteral opioids in labor is associated with maternal adverse effects, such as nausea, vomiting, dizziness, respiratory depression, oxygen desaturation, and sedation.13,24 Evidence is lacking that any one opioid is superior in maximizing pain relief while minimizing adverse effects.13
There is growing interest in ultra-rapid opioids (e.g., remifentanil [Ultiva]), which have rapid and predictable elimination, because they have demonstrated less neonatal sedation than other opioids.25,26 Although rapid-acting opioids have some benefits, remifentanil has been associated with an increased incidence of maternal pruritus and sedation compared with other opioids.27
Nonopioid systemic agents, such as antihistamines, acetaminophen, and sedatives (e.g., benzodiazepines, barbiturates) can be used to treat labor pains. There is limited evidence demonstrating superiority of benzodiazepines over barbiturates and antihistamines for pain control in labor.21
Regional Anesthesia
NEURAXIAL TECHNIQUES
Epidural, spinal, combined spinal-epidural, and dural puncture epidural anesthesia are neuraxial techniques commonly used in labor (Figure 1 and Table 415,28–33 ). In the 1980s, neuraxial regional anesthesia was used in 9% to 22% of labors; this had increased to 73% of labors by 2016.34,35 Neuraxial anesthesia is an effective pain control option, but there are concerns about adverse effects, such as decreased mobilization in labor, increased rates of assisted vaginal delivery and cesarean delivery, and increased adverse maternal and fetal outcomes. However, many of these concerns are readily managed by dosing refinements and modern approaches to administration of medications.
FIGURE 1.
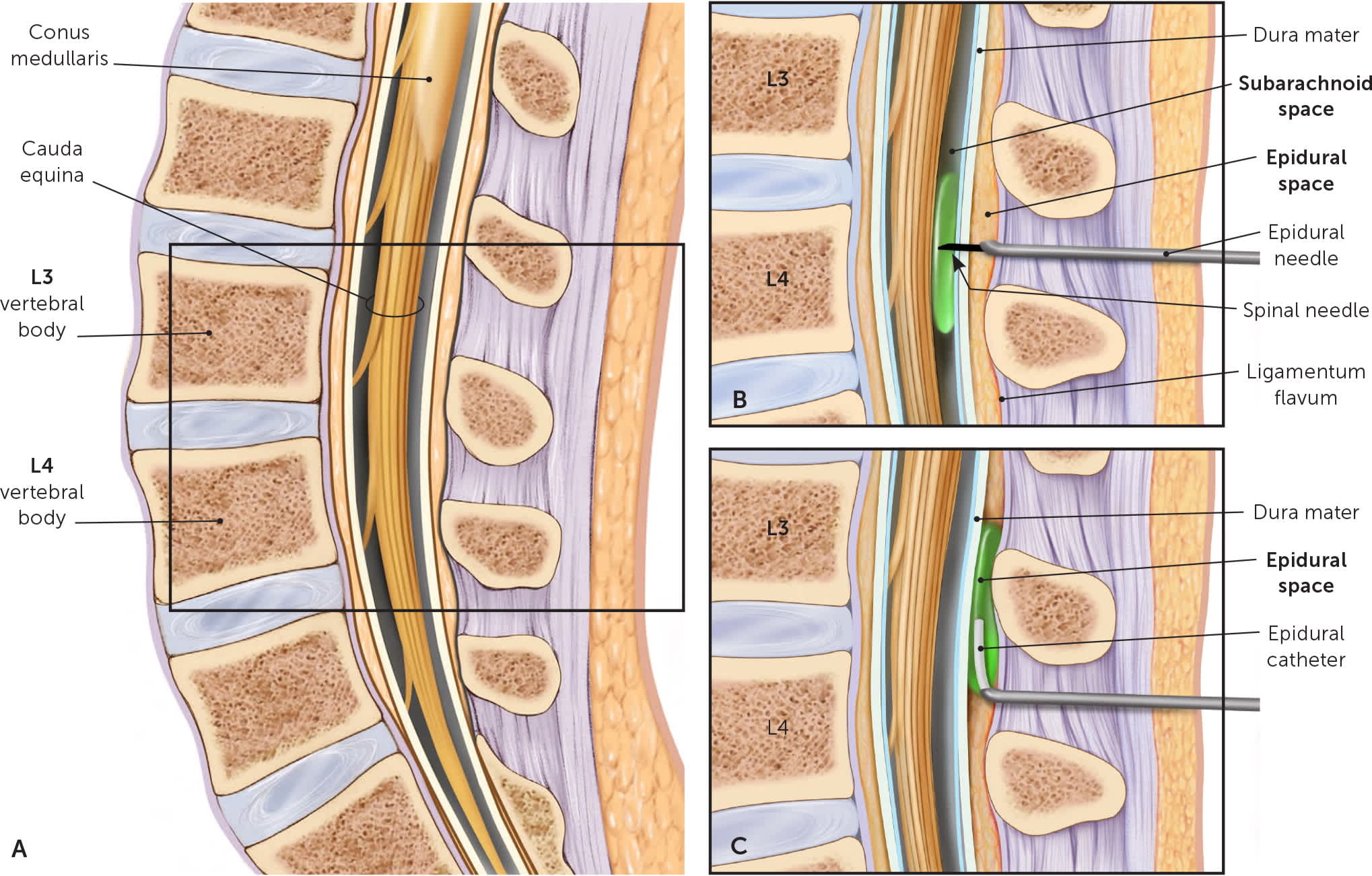
Methods of neuraxial anesthesia. (A) Sagittal cross section through lumbar spine. (B) In a combined spinal-epidural block, an epidural needle is first inserted into the epidural space. A spinal needle is inserted through the epidural needle to inject medication into the subarachnoid space and is then removed, leaving an epidural catheter in the epidural space. (C) With the epidural catheter left in place, intermittent or continuous medication may be administered.
Illustration by Christy Krames
TABLE 4. Neuraxial Regional Anesthesia Options for Pain Management During Labor
| Type | Stage of labor | Challenges/limitations* | Evidence |
|---|---|---|---|
| Epidural15,28–30 | First; second if delivery not imminent Give on request at any point once committed to delivery | Time to pain relief is about 15 minutes Decreased mobility Hypotension (with possible fetal heart rate deceleration and maternal nausea) Risk of maternal respiratory depression, fever, and urinary retention Risk of postdural puncture headache (< 1%) | Better pain relief than opioids More sensory than motor blockade No effect on rate of cesarean delivery or assisted vaginal delivery May lead to slightly longer second stage |
| Combined spinal-epidural29–31 | First; second if delivery not imminent | Risks associated with epidural (see above) Increased risk of pruritus compared with traditional epidural30 Catheter placement cannot be confirmed until the spinal component has worn off | Rapid onset of pain relief (5 minutes) Better pain relief than opioids More sensory than motor blockade No effect on rate of cesarean delivery or assisted vaginal delivery Slightly longer second stage |
| Dural puncture epidural29,30,32 | First; second if delivery not imminent | Risks associated with epidural (see above) Increased risk of pruritus compared with traditional epidural30 | Mixed results from small studies Faster onset of pain relief than traditional epidural Lower rates of asymmetric block than with traditional epidural or combined spinal-epidural Lower rates of need for rescue anesthesia than with combined spinal-epidural Less maternal hypotension than with combined spinal-epidural Significant sensory and motor blockade |
Note: Local anesthetic is often combined with an opioid, which typically leads to faster onset of pain relief.33 Commonly used local anesthetics and opioids are summarized in Table 5.
*—Contraindications to neuraxial regional anesthesia are listed in Table 6, and complications and considerations are discussed further in eTable A.
An epidural block is achieved by inserting a catheter into the epidural space and administering a continuous or intermittent infusion of medication. Pain relief generally occurs within 15 minutes and can be adjusted or augmented with boluses as needed.29 In contrast, a spinal block involves administering a single injection into the subarachnoid space. Spinal anesthesia generally provides pain relief within minutes, lasts 45 to 90 minutes, and is more likely to affect motor function than epidural anesthesia.36
A combined spinal-epidural block, also known as a walking epidural, allows the patient greater control over positioning and achieves rapid pain relief followed by continued analgesia. The epidural needle is inserted into the epidural space, and then a spinal needle is inserted through the epidural needle to puncture the dura and administer medication into the subarachnoid space. The spinal needle is removed, and an epidural catheter is left in place to administer continuous or intermittent medication.
Compared with traditional epidurals, combined spinal-epidural anesthesia leads to faster onset of pain relief, decreased use of rescue anesthesia (RR = 0.31; 95% CI, 0.14 to 0.70), and fewer assisted vaginal deliveries (RR = 0.81; 95% CI, 0.67 to 0.97) but is associated with increased pruritus (RR = 1.80; 95% CI, 1.22 to 2.65) and increased likelihood of nonreassuring fetal heart tracings (RR = 1.31; 95% CI, 1.02 to 1.67).30,31 There is an increased likelihood of maternal adverse effects (such as pruritus and nausea) and of nonreassuring fetal heart tracings when a combination of an opioid and local anesthetic is injected into the subarachnoid space, compared with the use of local anesthetic alone.37 There are no known differences between combined spinal-epidural anesthesia and epidural anesthesia in maternal mobilization during labor, cesarean delivery rate, maternal hypotension, umbilical artery pH, or Apgar scores. Moreover, studies of modern techniques, such as use of atraumatic needle type and smaller needle gauge, show no difference in the incidence of postdural puncture headaches between the groups.38
Dural puncture epidural anesthesia similarly involves puncture of the dura with a spinal needle through the epidural needle. The spinal needle is removed before the administration of medication through the epidural catheter into the epidural space. The dural puncture allows some of the anesthetic to move into the subarachnoid space, with the goal of hastening analgesia onset without the risks of combined spinal-epidural anesthesia, such as nonreassuring fetal heart tracings.32
A single-injection spinal block may be considered in the later phases of labor because it can be administered more quickly than an epidural block and is associated with low risk of neonatal respiratory distress. Multiparous patients with cervical dilation of at least 6 cm are ideal candidates for this technique given the short duration of action and high likelihood of imminent delivery following use.39
Pharmacologic Options. Neuraxial techniques typically use a local anesthetic combined with an opioid. The addition of an opioid allows for faster analgesia onset and lower dosing of the local anesthetic, which in turn permits increased maternal mobility during labor and less maternal hemodynamic instability.33 Commonly used local anesthetics and opioids are summarized in Table 5.28,29,33
TABLE 5. Common Medications Used in Neuraxial Regional Anesthesia During Labor
| Medications | Common concentrations | Onset of pain relief |
|---|---|---|
| Bupivacaine (Marcaine) | 0.0625% to 0.125% | Slow |
| Chloroprocaine (Nesacaine) | 2% to 3% | Rapid |
| Lidocaine | 0.75% to 1.0% | Rapid |
| Ropivacaine (Naropin) | 0.125% to 1.0% | Slow |
| Fentanyl Sufentanil | 50 mcg per mL 50 mcg per mL | Opioid is typically added to local anesthetic to decrease time to onset of pain relief, increase duration of analgesia, and improve patient satisfaction with analgesia |
Continuous Regional Anesthesia. Epidural anesthesia is administered by continuous infusion or programmed intermittent bolus. Regular intermittent boluses result in lower total doses to achieve adequate analgesia compared with continuous infusion, with improved maternal satisfaction and no effect on mode of delivery or risk of adverse outcomes.40,41
Complications and Contraindications. Of all pregnancy-related deaths in 1990, 2.5% were due to anesthesia-related complications.42 With improved anesthesia practices, that number had dropped to 0.2% by 2013.43 Complications, such as a high neuraxial block (i.e., a block that exceeds the requirements of analgesia and is associated with cardiorespiratory compromise), respiratory arrest, and unrecognized spinal catheter placement, are rare and affect approximately one per 4,000, one per 10,000, and one per 15,000 patients, respectively.44
The use of neuraxial anesthesia in labor is not associated with increased rates of assisted vaginal delivery or cesarean delivery, regardless of whether it is initiated in latent or active labor or continued in the second stage of labor.12,45–47 One Cochrane review demonstrated increased rates of assisted vaginal delivery (RR = 1.44; 95% CI, 1.29 to 1.60) with epidural use; however, this effect was negated when trials published before 2005 were removed from analysis, which is thought to be attributable to development of modern anesthesia techniques.12 The second stage of labor is roughly 15 minutes longer in nulliparous and multiparous patients when neuraxial anesthesia is used.
Table 6 lists contraindications to neuraxial anesthesia,48–51 and eTable A discusses the considerations and possible complications.
TABLE 6. Contraindications to Neuraxial Regional Anesthesia in Labor

| Contraindications | Comments |
|---|---|
| Absolute | |
| Increased intracranial pressure | Intracranial mass, hydrocephalus |
| Infection overlying area of necessary skin puncture | Risk of meningitis |
| Lack of patient consent for anesthesia | — |
| Relative | |
| Coagulopathy | Inherited bleeding disorders increase the risk of vertebral canal hematoma (one per 168,000 patients) Patients with von Willebrand disease have rare bleeding risk because von Willebrand factor concentrations can increase up to 300% during pregnancy Hemophilia carriers should undergo normalization of factor concentrations before anesthesia administration and for three to five days after delivery to avoid bleeding* |
| Hemodynamic instability | Severe dehydration, sepsis, cardiomyopathy, pulmonary embolism, anaphylaxis |
| Inability to maintain appropriate positioning for the procedure | Advanced dilation or anxiety may inhibit the patient from safely achieving the necessary positioning |
| Some obstructive cardiomyopathies | Includes severe mitral or aortic stenosis and left ventricular outflow obstruction as seen with hypertrophic obstructive cardiomyopathy |
| Thrombocytopenia | No specific platelet count at which complications are increased Generally considered acceptable with platelet count of 70 × 103 per μL (70 × 109 per L) or higher Many feel comfortable with lower levels in gestational thrombocytopenia and idiopathic thrombocytopenia |
*—Most experts agree that patients with a normalized von Willebrand ristocetin cofactor assay and normalized factor VIII and von Willebrand antigen concentrations should be candidates for neuraxial anesthesia.
eTABLE A Considerations and Complications for Neuraxial Regional Anesthesia in Labor
| Complication | Background | Risk | Evidence-based management/additional considerations |
|---|---|---|---|
| Assisted vaginal delivery | Older studies that do not reflect modern anesthesia practices have demonstrated increased rates of assisted vaginal delivery associated with epidural use | No association based on more recent studies | Lack of association with assisted vaginal delivery, regardless of whether neuraxial anesthesia is initiated in the latent, active, or second stage of labor A1 No change in the rate of assisted vaginal delivery with cessation of neuraxial anesthesia in the second stage of laborA2 |
| Cesarean delivery | Older studies that do not reflect modern anesthesia practices have demonstrated increased rates of cesarean delivery associated with epidural use | No association based on newer studiesA1,A3 | Newer studies show a lack of association with cesarean delivery, regardless of whether neuraxial anesthesia is initiated in the latent, active, or second stage of laborA1 No change in cesarean delivery rate with cessation of neuraxial anesthesia in the second stage of laborA1,A2 |
| Maternal fever response | Neuraxial anesthesia is associated with an increase in maternal temperatureA4–A6 May be related to inflammation of the placenta and membranes, altered thermoregulation, or lack of additional pain medications that would confer antipyretic effectsA4–A6 | 30%A4 | Antipyretic treatments Maternal intrapartum fever is associated with neonatal brain injury (e.g., encephalopathy, cerebral palsy, neurocognitive delay)A4,A7 and may lead to unnecessary neonatal sepsis workup or prophylactic neonatal antibioticsA5,A7,A8 |
| Maternal hypotension | Sympathetic nerve blockade decreases maternal systemic vascular resistance and increases venous capacity | Approximately 30% (high-dose epidural only)A9 | Lower limb compression during epiduralA10 Inconsistent evidence for preloading crystalloid intravenous fluid before epiduralA9 Vasopressors (most commonly ephedrine [Akovaz] or phenylephrine) are the mainstay of treatment if abnormal fetal heart rate patterns occurA11 |
| Postdural puncture headache | Accidental puncture of the dura with the epidural needle may result in headache, neck stiffness, and hearing changes, as well as increased pushing time | < 1% (about one in 144 patients)A12 | Aggressive hydration, nonsteroidal anti-inflammatory drugs, recumbent positioning, abdominal binders A13 About 50% of patients require an epidural blood patchA12,A14 Prophylactic epidural blood patch, intravenous neostigmine (Bloxiverz) and atropine, and intrathecal morphine have not demonstrated a clear benefitA15–A17 |
| Prolonged second stage of labor | Improved pain control in the second stage of labor is thought to be associated with decreased quality of pushing | Varies | Second stage of labor is generally about 15 minutes longer in nulliparous and multiparous patients when neuraxial anesthesia is usedA18 Modern anesthesia practices use lower concentrations of local anesthetic, which may prolong duration of labor less significantly than traditional dosingA1,A19 |
Information from:
A1. Sng BL, Leong WL, Zeng Y, et al. Early versus late initiation of epidural analgesia for labour. Cochrane Database Syst Rev. 2014;(10):CD007238.
A2. Torvaldsen S, Roberts CL, Bell JC, et al. Discontinuation of epidural analgesia late in labour for reducing the adverse delivery outcomes associated with epidural analgesia. Cochrane Database Syst Rev. 2004;(4):CD004457.
A3. Shen X, Li Y, Xu S, et al. Epidural analgesia during the second stage of labor: a randomized controlled trial. Obstet Gynecol. 2017;130(5):1097–1103.
A4. Segal S. Labor epidural analgesia and maternal fever. Anesth Analg. 2010;111(6):1467–1475.
A5. Riley LE, Celi AC, Onderdonk AB, et al. Association of epidural-related fever and noninfectious inflammation in term labor. Obstet Gynecol. 2011;117(3):588–595.
A6. Sultan P, David AL, Fernando R, et al. Inflammation and epidural-related maternal fever: proposed mechanisms. Anesth Analg. 2016;122(5):1546–1553.
A7. Arendt KW, Segal BS. The association between epidural labor analgesia and maternal fever. Clin Perinatol. 2013;40(3):385–398.
A8. Jansen S, Lopriore E, Naaktgeboren C, et al. Epidural-related fever and maternal and neonatal morbidity: a systematic review and meta-analysis [published online January 28, 2020]. Neonatology. Accessed June 6, 2020. https://www.karger.com/Article/FullText/504805
A9. Hofmeyr G, Cyna A, Middleton P. Prophylactic intravenous preloading for regional analgesia in labour. Cochrane Database Syst Rev. 2004;(4):CD000175.
A10. Peyronnet V, Roses A, Girault A, et al. Lower limbs venous compression reduces the incidence of maternal hypotension following epidural analgesia during term labor. Eur J Obstet Gynecol Reprod Biol. 2017;219:94–99.
A11. Lee A, Ngan Kee WD, Gin T. A quantitative, systematic review of randomized controlled trials of ephedrine versus phenylephrine for the management of hypotension during spinal anesthesia for cesarean delivery. Anesth Analg. 2002;94(4):920–926.
A12. D'Angelo R, Smiley RM, Riley ET, et al. Serious complications related to obstetric anesthesia: the serious complication repository project of the Society for Obstetric Anesthesia and Perinatology. Anesthesiology. 2014;120(6):1505–1512.
A13. Katz D, Beilin Y. Review of the alternatives to epidural blood patch for treatment of postdural puncture headache in the parturient. Anesth Analg. 2017;124(4):1219–1228.
A14. Costa AC, Satalich JR, Al-Bizri E, et al. A ten-year retrospective study of post-dural puncture headache in 32,655 obstetric patients. Can J Anaesth. 2019;66(12):1464–1471.
A15. Gaiser RR. Postdural puncture headache: an evidence-based approach. Anesthesiol Clin. 2017;35(1):157–167.
A16. Abdelaal Ahmed Mahmoud A, Mansour AZ, Yassin HM, et al. Addition of neostigmine and atropine to conventional management of postdural puncture headache: a randomized controlled trial. Anesth Analg. 2018;127(6):1434–1439.
A17. Al-metwalli RR. Epidural morphine injections for prevention of post dural puncture headache. Anaesthesia. 2008;63(8):847–850.
A18. Cheng YW, Shaffer BL, Nicholson JM, et al. Second stage of labor and epidural use: a larger effect than previously suggested [published correction appears in Obstet Gynecol. 2014;123(5):1109]. Obstet Gynecol. 2014;123(3):527–535.
A19. Anim-Somuah M, Smyth RM, Cyna AM, et al. Epidural versus non-epidural or no analgesia for pain management in labour. Cochrane Database Syst Rev. 2018;(5):CD000331.
PERIPHERAL TECHNIQUES
Peripheral regional techniques include paracervical and pudendal blocks. In a paracervical block, anesthesia to the cervix and uterus can be achieved within five minutes of injecting a local anesthetic into the fornix of the vagina, lateral to the cervix.52 A paracervical block is less effective than an epidural in the first stage of labor but may be more effective than systemic opioids.53 The incidence of fetal bradycardia, the most common adverse effect of a paracervical block, has not been associated with increased cesarean delivery and has decreased with the introduction of a superficial injection technique using low-dose anesthetic.54
A pudendal block involves injecting local anesthetic transvaginally, near the ischial spines. It provides anesthesia to the perineum, vulva, and lower vagina and is used in the second stage of labor and for perineal laceration repairs. Because a loss of the bearing down reflex is common, pudendal blocks are typically used immediately before expected birth. Although rare, potential risks of pudendal block include hematoma, infection, and nerve damage.55 A 2013 audit demonstrated that most obstetricians could not describe how to perform a pudendal block, suggesting that the popularity of this form of anesthesia is diminishing.56
Postpartum Analgesia
Untreated pain in the postpartum period can lead to increased postpartum depression, opioid use, and chronic pain. Major maternal-child health organizations endorse a stepwise and multimodal approach to postpartum analgesia, as well as considering the mode of delivery when choosing analgesia modalities.57
After vaginal delivery, the most common sources of pain include perineal lacerations, nipple trauma, and uterine cramping. Perineal pain is best managed with direct application of cold packs, whereas nipple pain requires careful assessment of infant latch and positioning if breastfeeding, as well as evaluation for infection.58
After cesarean delivery, opioids administered through spinal or epidural routes are the mainstay of immediate postoperative pain control. Evidence does not clearly demonstrate superiority of systemic opioids, which should be reserved for pain that is uncontrolled by nonopioid medications.59
Subcutaneous wound infiltration with local anesthetic, as well as a transversus abdominis plane block in which local anesthetic is injected in the plane between the internal oblique and transversus abdominis muscles, may decrease postoperative pain and opioid requirements. This is particularly beneficial when intrathecal narcotics are not used in the epidural or spinal block.60,61
Patients with opioid use disorder may have a higher need for postpartum analgesia because of opioid tolerance. For a patient using methadone or buprenorphine medication-assisted therapy, standard dosing is typically continued postpartum, with the addition of multimodal nonopioid medications.
At discharge, approximately one-third of patients who had a vaginal delivery and two-thirds of patients who had a cesarean delivery receive an opioid prescription, and approximately 2% of patients start using opioids persistently in the postpartum period.62 Judicious opioid prescribing, smaller prescription quantities, and options for partial refills can help address problematic opioid use postpartum.
This article updates previous articles on this topic by Schrock and Harraway-Smith63; Leeman, et al.64; Leeman, et al.65; and Vincent and Chestnut.66
Data Sources: A PubMed search was completed in Clinical Queries using the key terms: obstetric delivery, labor presentation, epidural analgesia, pudendal block, paracervical block, nitrous oxide, inhaled anesthetic, labor support, and analgesia in labor. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Also searched were the Cochrane database and Essential Evidence Plus. Search dates: January 21 to April 20, 2020; June 6, 2020; and November 8, 2020.

