Cerebrospinal fluid (CSF) analysis is a diagnostic tool for many conditions affecting the central nervous system. Urgent indications for lumbar puncture include suspected central nervous system infection or subarachnoid hemorrhage. CSF analysis is not necessarily diagnostic but can be useful in the evaluation of other neurologic conditions, such as spontaneous intracranial hypotension, idiopathic intracranial hypertension, multiple sclerosis, Guillain-Barré syndrome, and malignancy. Bacterial meningitis has a high mortality rate and characteristic effects on CSF white blood cell counts, CSF protein levels, and the CSF:serum glucose ratio. CSF culture can identify causative organisms and antibiotic sensitivities. Viral meningitis can present similarly to bacterial meningitis but usually has a low mortality rate. Adjunctive tests such as CSF lactate measurement, latex agglutination, and polymerase chain reaction testing can help differentiate between bacterial and viral causes of meningitis. Immunocompromised patients may have meningitis caused by tuberculosis, neurosyphilis, or fungal or parasitic infections. Subarachnoid hemorrhage has a high mortality rate, and rapid diagnosis is key to improve outcomes. Computed tomography of the head is nearly 100% sensitive for subarachnoid hemorrhage in the first six hours after symptom onset, but CSF analysis may be required if there is a delay in presentation or if imaging findings are equivocal. Xanthochromia and an elevated red blood cell count are characteristic CSF findings in patients with subarachnoid hemorrhage. Leptomeningeal carcinomatosis can mimic central nervous system infection. It has a poor prognosis, and large-volume CSF cytology is diagnostic.
Cerebrospinal fluid (CSF) analysis is a diagnostic tool for many conditions affecting the central nervous system (CNS). The correct selection and interpretation of CSF tests can assist with diagnosing and treating a variety of conditions. Table 1 lists the normal values of CSF components.1–4
SORT: KEY RECOMMENDATIONS FOR PRACTICE
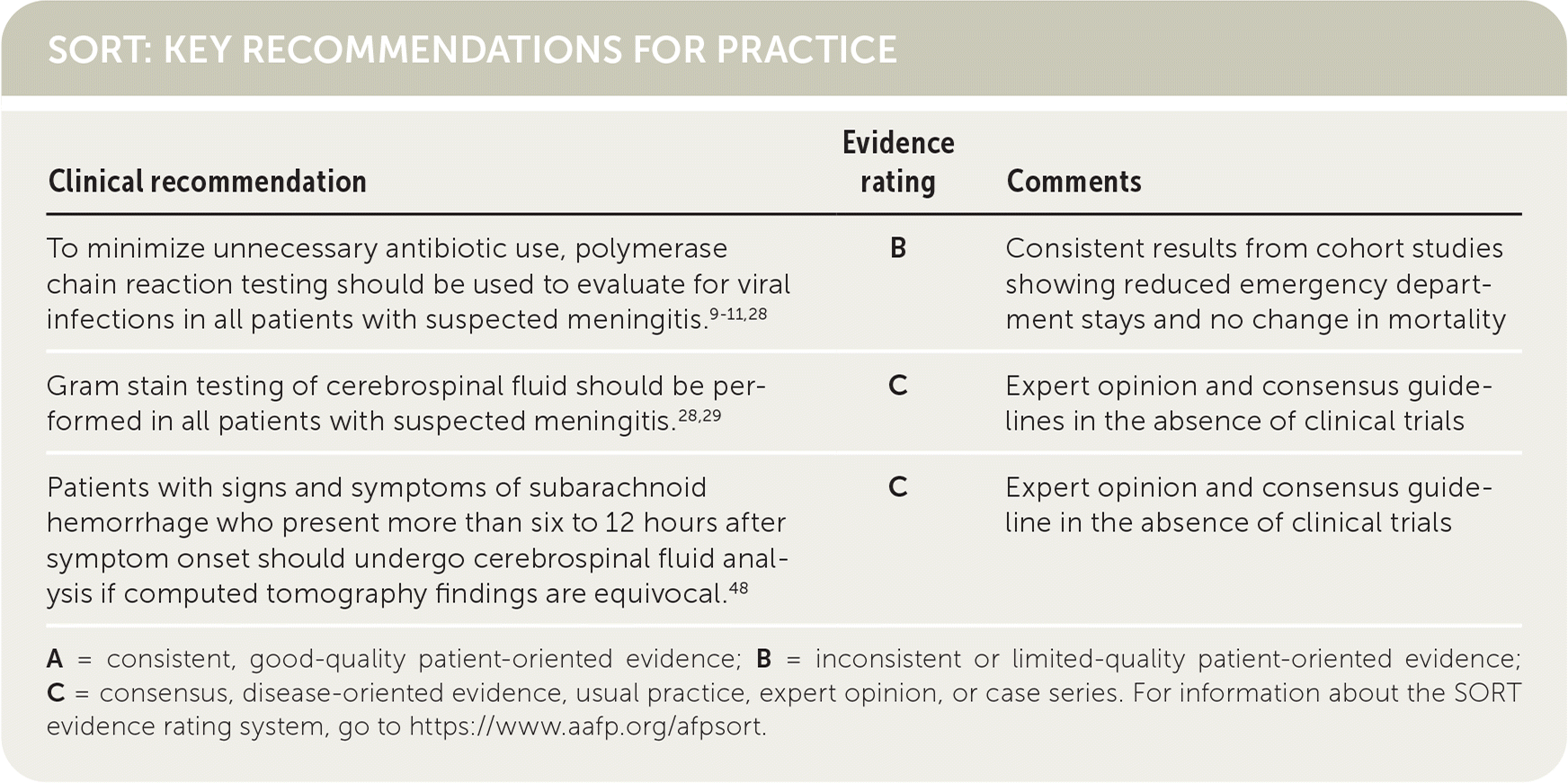
| Clinical recommendation | Evidence rating | Comments |
|---|---|---|
| To minimize unnecessary antibiotic use, polymerase chain reaction testing should be used to evaluate for viral infections in all patients with suspected meningitis.9–11,28 | B | Consistent results from cohort studies showing reduced emergency department stays and no change in mortality |
| Gram stain testing of cerebrospinal fluid should be performed in all patients with suspected meningitis.28,29 | C | Expert opinion and consensus guidelines in the absence of clinical trials |
| Patients with signs and symptoms of subarachnoid hemorrhage who present more than six to 12 hours after symptom onset should undergo cerebrospinal fluid analysis if computed tomography findings are equivocal.48 | C | Expert opinion and consensus guideline in the absence of clinical trials |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
TABLE 1. Normal Values of CSF Components
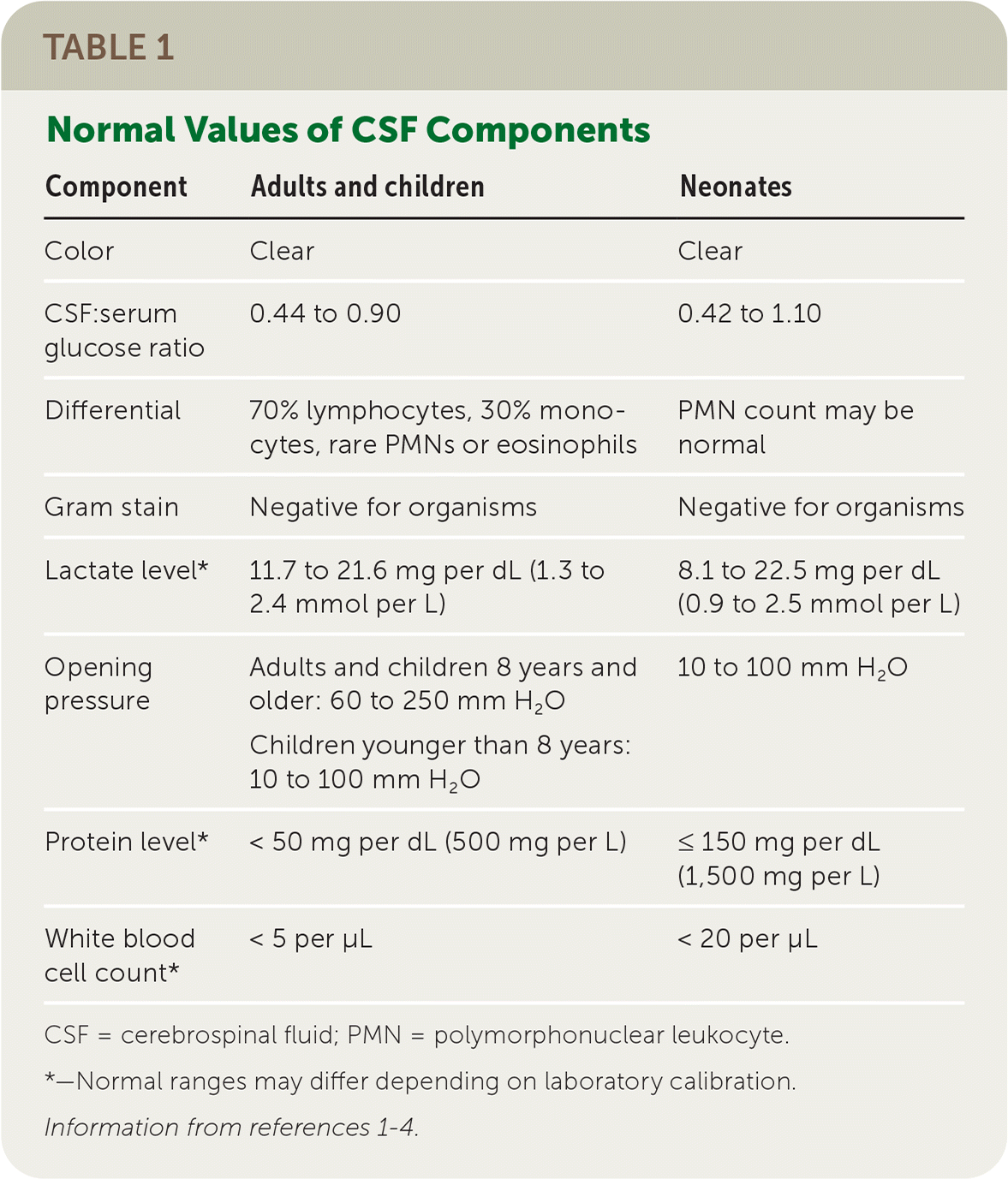
| Component | Adults and children | Neonates |
|---|---|---|
| Color | Clear | Clear |
| CSF:serum glucose ratio | 0.44 to 0.90 | 0.42 to 1.10 |
| Differential | 70% lymphocytes, 30% monocytes, rare PMNs or eosinophils | PMN count may be normal |
| Gram stain | Negative for organisms | Negative for organisms |
| Lactate level* | 11.7 to 21.6 mg per dL (1.3 to 2.4 mmol per L) | 8.1 to 22.5 mg per dL (0.9 to 2.5 mmol per L) |
| Opening pressure | Adults and children 8 years and older: 60 to 250 mm H2O Children younger than 8 years: 10 to 100 mm H2O | 10 to 100 mm H2O |
| Protein level* | < 50 mg per dL (500 mg per L) | ≤ 150 mg per dL (1,500 mg per L) |
| White blood cell count* | < 5 per μL | < 20 per μL |
CSF = cerebrospinal fluid; PMN = polymorphonuclear leukocyte.
*—Normal ranges may differ depending on laboratory calibration.
CNS Infections
Suspected CNS infection is a medical emergency and is the most common indication for lumbar puncture. Bacterial meningitis has a 14% to 25% mortality rate; therefore, rapid CSF evaluation and early empiric antibiotic treatment are critical.5,6 CSF analysis is required to identify the causative organism and select appropriate antimicrobial coverage. An article on meningitis has been published previously in American Family Physician.7
When available, polymerase chain reaction (PCR) testing should be used to rapidly diagnose viral meningitis, which allows for early discontinuation of antibiotics.8–11 It can also detect common viral and bacterial etiologies with more than 95% sensitivity and specificity,8,12 whereas viral cultures require days to weeks and have lower accuracy.13 However, PCR testing does not assess drug sensitivities, is not universally available, and does not detect every infectious organism. Table 2 lists other suggested tests for various types of CNS infections.1,8,14–25
TABLE 2. CSF Characteristics by Infection Type
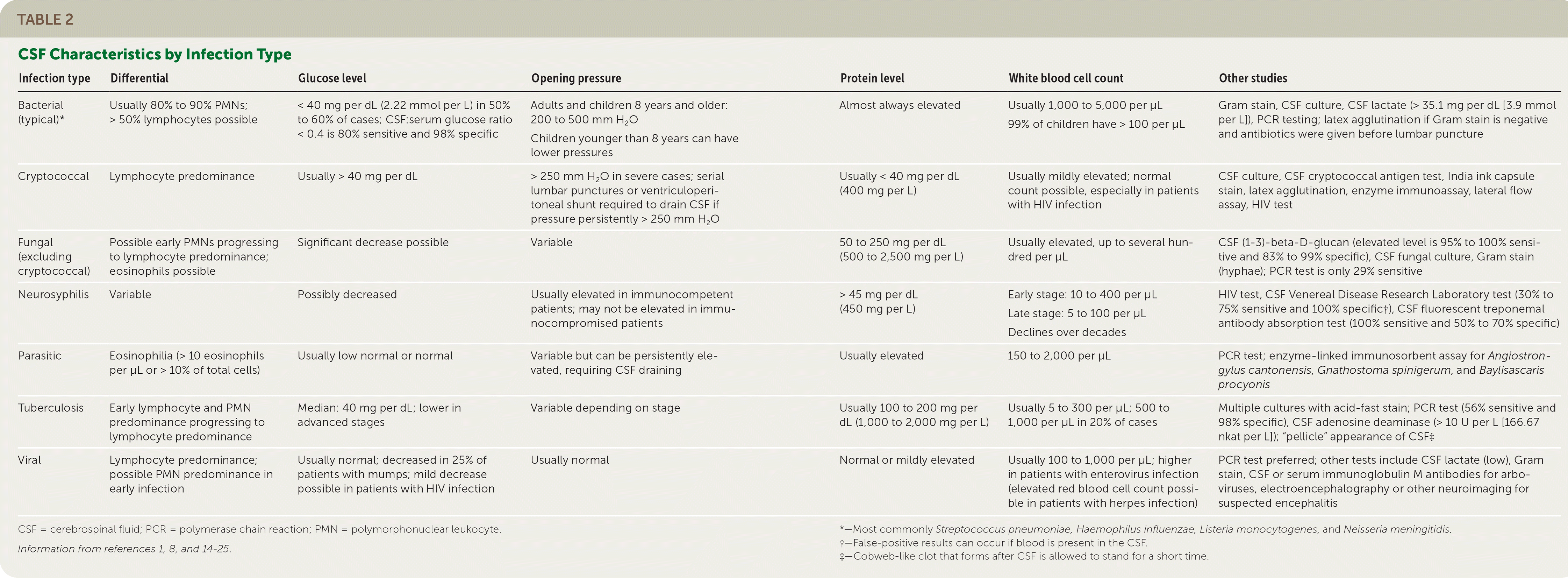
| Infection type | Differential | Glucose level | Opening pressure | Protein level | White blood cell count | Other studies |
|---|---|---|---|---|---|---|
| Bacterial (typical)* | Usually 80% to 90% PMNs; > 50% lymphocytes possible | < 40 mg per dL (2.22 mmol per L) in 50% to 60% of cases; CSF:serum glucose ratio < 0.4 is 80% sensitive and 98% specific | Adults and children 8 years and older: 200 to 500 mm H2O Children younger than 8 years can have lower pressures | Almost always elevated | Usually 1,000 to 5,000 per μL 99% of children have > 100 per μL | Gram stain, CSF culture, CSF lactate (> 35.1 mg per dL [3.9 mmol per L]), PCR testing; latex agglutination if Gram stain is negative and antibiotics were given before lumbar puncture |
| Cryptococcal | Lymphocyte predominance | Usually > 40 mg per dL | > 250 mm H2O in severe cases; serial lumbar punctures or ventriculoperitoneal shunt required to drain CSF if pressure persistently > 250 mm H2O | Usually < 40 mg per dL (400 mg per L) | Usually mildly elevated; normal count possible, especially in patients with HIV infection | CSF culture, CSF cryptococcal antigen test, India ink capsule stain, latex agglutination, enzyme immunoassay, lateral flow assay, HIV test |
| Fungal (excluding cryptococcal) | Possible early PMNs progressing to lymphocyte predominance; eosinophils possible | Significant decrease possible | Variable | 50 to 250 mg per dL (500 to 2,500 mg per L) | Usually elevated, up to several hundred per μL | CSF (1–3)-beta-D-glucan (elevated level is 95% to 100% sensitive and 83% to 99% specific), CSF fungal culture, Gram stain (hyphae); PCR test is only 29% sensitive |
| Neurosyphilis | Variable | Possibly decreased | Usually elevated in immunocompetent patients; may not be elevated in immunocompromised patients | > 45 mg per dL (450 mg per L) | Early stage: 10 to 400 per μL Late stage: 5 to 100 per μL Declines over decades | HIV test, CSF Venereal Disease Research Laboratory test (30% to 75% sensitive and 100% specific†), CSF fluorescent treponemal antibody absorption test (100% sensitive and 50% to 70% specific) |
| Parasitic | Eosinophilia (> 10 eosinophils per μL or > 10% of total cells) | Usually low normal or normal | Variable but can be persistently elevated, requiring CSF draining | Usually elevated | 150 to 2,000 per μL | PCR test; enzyme-linked immunosorbent assay for Angiostrongylus cantonensis, Gnathostoma spinigerum, and Baylisascaris procyonis |
| Tuberculosis | Early lymphocyte and PMN predominance progressing to lymphocyte predominance | Median: 40 mg per dL; lower in advanced stages | Variable depending on stage | Usually 100 to 200 mg per dL (1,000 to 2,000 mg per L) | Usually 5 to 300 per μL; 500 to 1,000 per μL in 20% of cases | Multiple cultures with acid-fast stain; PCR test (56% sensitive and 98% specific), CSF adenosine deaminase (> 10 U per L [166.67 nkat per L]); “pellicle” appearance of CSF‡ |
| Viral | Lymphocyte predominance; possible PMN predominance in early infection | Usually normal; decreased in 25% of patients with mumps; mild decrease possible in patients with HIV infection | Usually normal | Normal or mildly elevated | Usually 100 to 1,000 per μL; higher in patients with enterovirus infection (elevated red blood cell count possible in patients with herpes infection) | PCR test preferred; other tests include CSF lactate (low), Gram stain, CSF or serum immunoglobulin M antibodies for arboviruses, electroencephalography or other neuroimaging for suspected encephalitis |
CSF = cerebrospinal fluid; PCR = polymerase chain reaction; PMN = polymorphonuclear leukocyte.
Information from references 1, 8, and 14–25.
*—Most commonly Streptococcus pneumoniae, Haemophilus influenzae, Listeria monocytogenes, and Neisseria meningitidis.
†—False-positive results can occur if blood is present in the CSF.
‡—Cobweb-like clot that forms after CSF is allowed to stand for a short time.
BACTERIAL MENINGITIS
Opening pressure is the first CSF component assessed when performing a lumbar puncture. It is obtained before draining CSF fluid and should be measured with the patient in the lateral decubitus position. Opening pressure is greater than 300 mm H2O in 39% of patients with bacterial meningitis.26 The color of the CSF supernatant can be cloudy, green, or purulent.8 The white blood cell (WBC) count is often greater than 1,000 per μL, and polymorphonuclear leukocytes are typically predominant.27 However, 6% of patients with culture-diagnosed bacterial meningitis do not have an elevated WBC count, and lymphocytes are predominant in up to 10% of patients.28,29 WBC elevations can be caused by traumatic lumbar punctures rather than infections. The classic method to correct for traumatic WBC elevations has been to subtract one WBC for every 500 to 1,500 red blood cells (RBCs)26; a more accurate method is to use the formula WBCs (predicted) = CSF RBCs × (blood WBCs/blood RBCs).30
Nearly all patients with bacterial meningitis have an elevated CSF protein level.8 The ratio of CSF glucose to serum glucose can be normal to significantly decreased depending on the type of pathogen, time since infection onset, and presence of dextrose in any intravenous fluids the patient has received.31 Bacterial meningitis can be diagnosed with more than 99% certainty in patients with any of the following CSF measurements: glucose less than 34 mg per dL (1.89 mmol per L), protein greater than 220 mg per dL (2,200 mg per L), WBCs greater than 2,000 per μL, or neutrophils greater than 1,180 per μL.32 Gram stain testing is recommended for all patients with suspected CNS infection.28,29 It has a specificity of 97% for bacterial meningitis and a sensitivity of 33% to 90%,8,27,33 depending on the type of pathogen, cytocentrifugation, infection severity, laboratory personnel experience, the number of slides prepared, and the patient's exposure to antibiotics.27,31,34,35 Therefore, a negative CSF Gram stain cannot exclude bacterial infection, particularly in patients who have already started antibiotic therapy.
Bacterial meningitis may still be present if results of the CSF analysis are normal or only mildly abnormal. Further testing can help clarify the diagnosis if results of the CSF analysis are equivocal. The Infectious Diseases Society of America does not recommend routine measurement of CSF lactate in patients with suspected bacterial meningitis, but two recent meta-analyses demonstrate its potential value.28,29,36 A CSF lactate level greater than 35.1 mg per dL (3.9 mmol per L) is 93% to 95% sensitive and 94% to 99% specific for diagnosing bacterial meningitis if the CSF is obtained before antibiotic treatment begins.37,38 The CSF lactate level is particularly helpful in neurosurgical patients because inflammation from surgery can lead to unreliable measurements of conventional markers.36 Latex agglutination is another potentially useful adjunctive test for detecting bacterial antibodies or antigens in CSF. However, its sensitivity varies depending on the organism, and false positives have been reported.26,39 Latex agglutination is not recommended by the European Society of Clinical Microbiology and Infectious Diseases, which notes that this method is no more useful than Gram stain testing.29
CSF cultures can provide a definitive diagnosis and antimicrobial sensitivities. If antibiotic treatment is initiated before a CSF sample is obtained, the culture's yield will be decreased by 10% to 20%.29
VIRAL MENINGITIS
Viral meningitis is the most common type of CNS infection.40,41 Opening pressure is typically normal; the WBC count is generally less than 250 per μL; and lymphocytes are typically predominant, although polymorphonuclear leukocytes may be predominant in the early phase.42 The CSF protein level can be normal to mildly elevated (less than 150 mg per dL [1,500 mg per L]).31 The CSF glucose level is usually normal but can be mildly decreased; mumps infection can lower the CSF glucose level by 25%.8,31 CSF and serum immunoglobulin M antibody testing are the preferred diagnostic tests for arboviruses such as West Nile virus.15
IMMUNOCOMPROMISE
Immunocompromised patients are at increased risk of opportunistic infection. Adults older than 60 years and people with deficiencies in cell-mediated immunity have a higher incidence of Listeria monocytogenes meningitis.43 Elevated CSF protein and WBC measurements are significantly less likely in these patients, and Gram stain is negative in 66% of cases.44
Patients with acute retroviral syndrome due to early or acute HIV infection can present with an aseptic CSF profile (mild lymphocyte elevation, normal to mild protein elevation, and a normal to slightly low glucose level).8 Cryptococcal meningitis, toxoplasmosis, and neurosyphilis should be considered in patients with HIV and low CD4 cell counts.45 Table 2 summarizes characteristics of CSF in patients with common infections.1,8,14–25
Subarachnoid Hemorrhage
Subarachnoid hemorrhage has a mortality rate of 25% to 50% and requires urgent diagnosis.46 Symptoms include sudden onset of severe headache, neck stiffness, photophobia, nausea and vomiting, seizure, and loss of consciousness. Most clinically significant subarachnoid hemorrhages are apparent on computed tomography of the head, which is nearly 100% sensitive in the first six hours of symptoms46,47; after six to 12 hours, the sensitivity wanes and CSF analysis may be required for diagnosis.48
CSF is usually colorless in patients with subarachnoid hemorrhage, but the presence of as few as 400 RBCs can change its appearance.47 Enzymatic degradation of RBCs to produce bilirubin contributes to xanthochromia, a yellowish appearance of CSF. Xanthochromia begins to emerge after six hours in approximately 20% of subarachnoid hemorrhages and is present in 90% of patients after 12 hours.46 At least 50% of patients who present with a subarachnoid hemorrhage had an initial subclinical sentinel bleed two to three weeks earlier, which increases the likelihood of xanthochromia.46 Xanthochromia can persist for up to two weeks after a bleeding event, and its presence significantly decreases the probability of blood being present due to traumatic lumbar puncture.46 A CSF RBC count of less than 2,000 per μL in the absence of xanthochromia effectively rules out subarachnoid hemorrhage.42 Other causes of xanthochromia include a serum bilirubin level greater than 10 to 15 mg per dL (171.04 to 256.56 μmol per L), CSF protein level greater than 150 mg per dL, or a traumatic lumbar puncture with more than 100,000 RBCs per μL.1 Decreasing RBC counts in successive CSF collection tubes strongly suggest a traumatic lumbar puncture.49
Spontaneous Intracranial Hypotension
Intracranial hypotension, defined as an opening pressure lower than 60 mm H2O in an adult, is an uncommon finding. It can be spontaneous or associated with a CSF leak, usually after a head injury.50 Spontaneous intracranial hypotension has emerged as a significant cause of secondary headache syndrome, and its symptoms usually include a positional component.50 Case studies suggest that it may contribute to reversible dementia.51 Although evidence shows that many patients with spontaneous intracranial hypotension have low-normal opening pressures, less than 60 mm H2O is considered diagnostic.50
Idiopathic Intracranial Hypertension
Intracranial hypertension, defined as an opening pressure higher than 250 mm H2O in an adult, can be idiopathic or associated with infectious meningitis or intracranial masses. Idiopathic intracranial hypertension, also called pseudotumor cerebri, should be considered in cisgender women of childbearing age who have obesity and chronic headaches; it is unclear whether this risk also pertains to trans men and nonbinary persons with XX chromosomes. It has an annual incidence of 19.3 cases per 100,000 women 20 to 44 years of age who have obesity and can result in permanent vision loss if untreated.52 Diagnosis is based on the modified Dandy criteria (eTable A), which include an elevated opening pressure and normal CSF composition.53 When performing a lumbar puncture in a patient with an elevated opening pressure, the manometer should be used throughout the procedure to continuously monitor CSF pressure, and no fluid should be drained after the pressure decreases by 50%.54
eTABLE A Modified Dandy Criteria for the Diagnosis of Idiopathic Intracranial Hypertension

| CSF opening pressure > 250 mm H2O |
| Neuroimaging shows no etiology for intracranial hypertension |
| No impaired consciousness |
| No other cause of intracranial hypertension |
| No other neurologic abnormalities (except abducens nerve palsy) |
| Normal CSF composition |
| Signs and symptoms of increased intracranial pressure (e.g., headache, transient visual obscurations, pulsatile tinnitus, papilledema, vision loss) |
CSF = cerebrospinal fluid.
Information from Wall M, Corbett JJ. The modified Dandy criteria for idiopathic intracranial hypertension, no need to fix what is not broken. Neurology. January 10, 2014. Accessed May 10, 2020. https://n.neurology.org/content/modified-dandy-criteria-idiopathic-intracranial-hypertension-no-need-fix-what-not-broken
Demyelinating Disease
CSF analysis can assist in the diagnosis of neurologic conditions such as multiple sclerosis (MS) and Guillain-Barré syndrome. Patients with these conditions typically have an elevated CSF protein level but otherwise normal CSF composition. However, CSF analysis is only one component of these diagnoses; clinical and radiographic findings are also important considerations.55–58
CSF-restricted oligoclonal bands are the classic CSF finding in patients with MS. Their high sensitivity contributed to recent revisions in diagnostic criteria that allow the presence of CSF-restricted oligoclonal bands to substitute for the traditional dissemination-in-time criterion, particularly in patients younger than 50 years who have a clinical presentation otherwise typical of MS.55 However, oligoclonal bands are not necessarily specific for MS and can occur in other inflammatory and infectious conditions. Elevations in CSF neurofilament light proteins correlate with relapsing MS.56
The classic CSF finding in patients with Guillain-Barré syndrome is an elevated protein level with a normal WBC count in the setting of acute ascending areflexic paralysis. It is present in 50% of patients in the first week after presentation, increasing to approximately 75% by three weeks.57
Malignancy
Leptomeningeal carcinomatosis is a metastatic invasion of the leptomeninges that is difficult to treat and has a poor prognosis.59 It can present with headache, confusion, and nuchal rigidity, mimicking a CNS infection. CSF findings may also suggest infection: a decreased glucose level and elevated opening pressure, WBC count, and protein level are common.59 Magnetic resonance imaging has limited diagnostic specificity, and single-sample CSF cytology has poor sensitivity.60 Obtaining three high-volume lumbar punctures (approximately 10 mL) improves the sensitivity to approximately 90%, and flow cytometry can improve specificity.59 Recent evidence suggests that tumor cell–associated biomarkers in CSF (circulating tumor DNA, microRNA, and their respective metabolites) can assist in the diagnosis and treatment of leptomeningeal carcinomatosis; research into their potential use is ongoing.60
This article updates a previous article on this topic by Seehusen, et al.42
Data Sources: A literature search was performed in PubMed using the terms cerebrospinal fluid and meningitis. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. We also searched Essential Evidence Plus, the Cochrane database, and evidence reports from the Agency for Healthcare Research and Quality using the same terms. Search dates: August 2019 to July 2020.
The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the Department of the Army, Department of Defense, or the U.S. government.

