Adenomyosis is a clinical condition where endometrial glands are found in the myometrium of the uterus. One in three patients with adenomyosis is asymptomatic, but the rest may present with heavy menstrual bleeding, pelvic pain, or infertility. Heavy menstrual bleeding is the most common symptom. Adenomyosis is distinct from endometriosis (the presence of endometrial glands outside of the uterus), but the two conditions often occur simultaneously. Risk factors for developing adenomyosis include increasing age, parity, and history of uterine procedures. Most patients are diagnosed from 40 to 50 years of age, but younger patients with infertility are increasingly being diagnosed with adenomyosis as imaging modalities improve. Diagnosis of adenomyosis begins with clinical suspicion and is confirmed with transvaginal ultrasonography and pelvic magnetic resonance imaging. Treatment of adenomyosis typically starts with hormonal menstrual suppression. Levonorgestrel-releasing intrauterine systems have shown some effectiveness. Patients with adenomyosis may ultimately have a hysterectomy if symptoms are not controlled with medical therapy.
Adenomyosis is a benign uterine disorder in which endometrial glands are found in the myometrium of the uterus. Adenomyosis is distinct from endometriosis, which is the presence of endometrial glands outside of the uterus. Adenomyosis is a poorly understood condition.
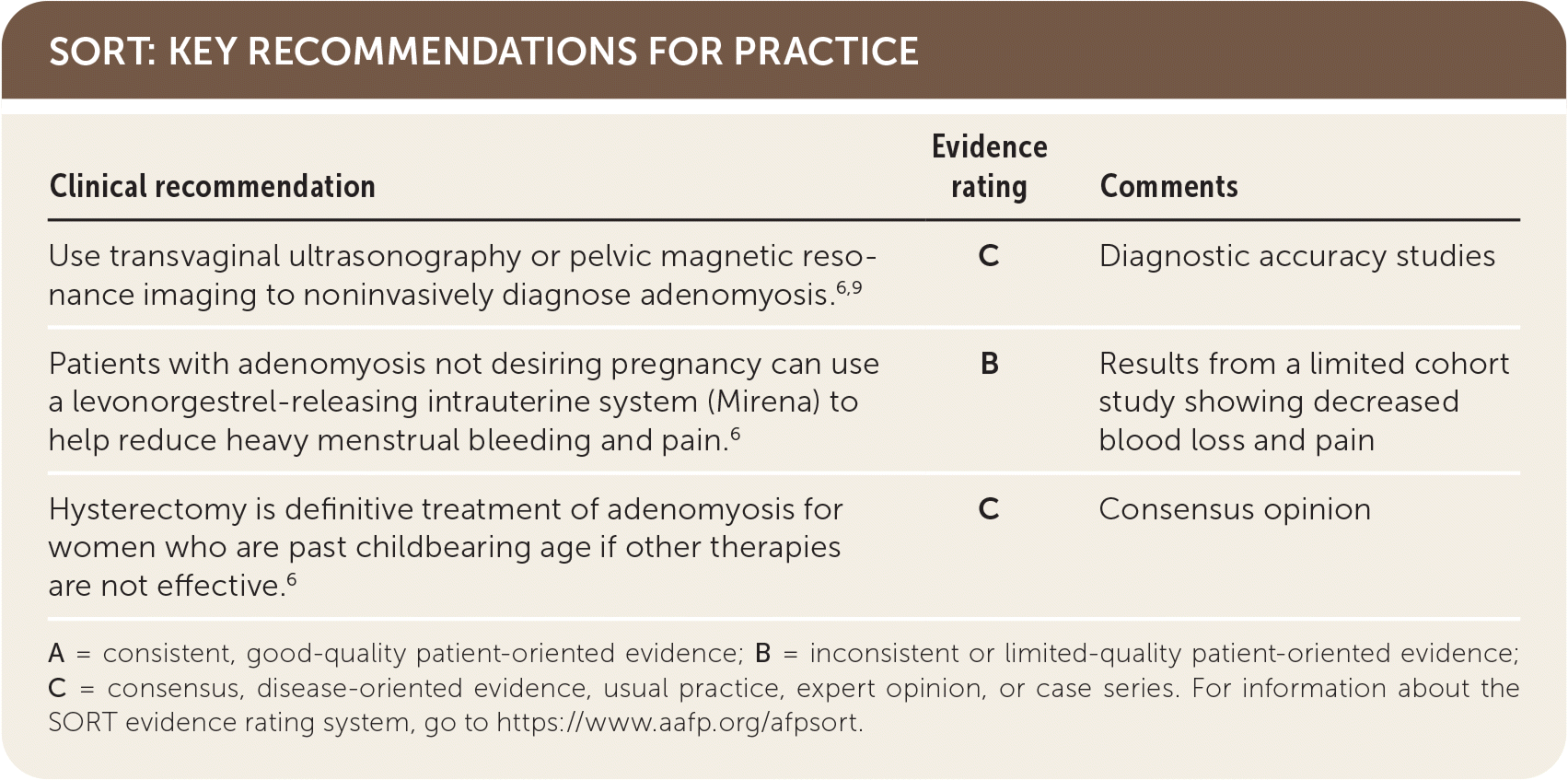
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | Comments |
|---|---|---|
| Use transvaginal ultrasonography or pelvic magnetic resonance imaging to noninvasively diagnose adenomyosis.6,9 | C | Diagnostic accuracy studies |
| Patients with adenomyosis not desiring pregnancy can use a levonorgestrel-releasing intrauterine system (Mirena) to help reduce heavy menstrual bleeding and pain.6 | B | Results from a limited cohort study showing decreased blood loss and pain |
| Hysterectomy is definitive treatment of adenomyosis for women who are past childbearing age if other therapies are not effective.6 | C | Consensus opinion |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Two theories prevail regarding the pathogenesis of adenomyosis.1,2 The first theory suggests that with injury of the endometrium, the basalis endometrium invaginates into the myometrium through an altered or interrupted junctional zone creating adenomyotic lesions. The tissue injury and repair theory may help explain why having a previous uterine procedure (e.g., cesarean delivery, dilation and curettage) increases the risk of subsequent adenomyosis. The second theory suggests that adenomyotic lesions arise from metaplasia of embryonic pluripotent Müllerian remnants. Adenomyosis can be classified as diffuse (involving a large area of endometrium) or focal.1
Epidemiology
The diagnosis of adenomyosis was previously confirmed in only post-hysterectomy cases, and it was thought to predominate in patients older than 40 years. Improved imaging makes it clear that younger patients also have adenomyosis.1 In one study of 985 symptomatic patients seen in a gynecology clinic using specific ultrasound diagnostic criteria, adenomyosis had a 20.9% prevalence in the study population (including pre- and postmenopausal, nulligravid, and multiparous patients), with a range of 10% to 35% in histology reports after hysterectomy.3
A population-based study of 650,000 patients estimated the overall incidence of adenomyosis at 1%, or 29 per 10,000 person-years, over a 10-year period based on International Classification of Diseases, 10th ed. (ICD-10) coding.4 Of those with adenomyosis, 90.8% had associated clinical symptoms based on chart review.4 The incidence of adenomyosis in the study was highest among patients 41 to 45 years of age.4 The overall prevalence of adenomyosis in 2015 was 0.8%, with a high of 1.5% among patients 41 to 45 years of age.4 The large variations in estimating incidence and prevalence of adenomyosis could be because there are no standard histologic criteria for diagnosis, as well as the recent advent of laparoscopic surgery creating morcellating specimens that alter the arrangement of tissue, leading to a more difficult diagnosis.5 Table 1 lists populations in which adenomyosis is commonly diagnosed.1
TABLE 1. Patients in Whom Adenomyosis Is Commonly Diagnosed

| Most common Multiparous Older than 40 years Prior cesarean delivery Prior uterine surgery Increasingly diagnosed Infertile Younger than 40 years With dysmenorrhea, abnormal uterine bleeding, or both |
Information from reference 1.
CASE
A 42-year-old patient presents with a six-month history of chronic pelvic pain. The patient describes the pain as aching and deep in the pelvis. The patient has regular menses but notes that it has been getting progressively heavier with more dysmenorrhea for the past few years. The patient has a history of infertility, and the uterus feels mildly enlarged and tender on bimanual examination. Pelvic ultrasonography suggests diffuse adenomyosis.
Clinical Presentation
Up to one-third of patients with adenomyosis can be asymptomatic.5 Symptoms typically arise between 40 and 50 years of age.5 There is no pathognomonic sign or symptom of adenomyosis. Common symptoms include abnormal uterine bleeding (heavy menstrual bleeding and irregular menses) and dysmenorrhea (Table 2). Less common symptoms include dyspareunia and chronic pelvic pain.6,7
TABLE 2. Symptoms and Signs of Adenomyosis

| Symptoms Abnormal uterine bleeding Chronic pelvic pain Dysmenorrhea Dyspareunia Signs Infertility Uterine enlargement and boggy consistency Uterine tenderness |
Heavy menstrual bleeding occurs in 40% to 60% of patients with adenomyosis.6 Heavy bleeding is likely caused by the increased surface area of the endometrium, subsequent increase in total volume of the endometrium and endometrial glands, or the increased vascularization of the lining of the endometrium and is directly correlated with the extent of myometrial invasion.5,6 Adenomyosis is part of the American College of Obstetricians and Gynecologists PALM-COEIN acronym for the evaluation of abnormal uterine bleeding in reproductive-aged patients (Table 3).8
TABLE 3. American College of Obstetricians and Gynecologists PALM-COEIN Acronym for Etiology of Abnormal Uterine Bleeding

| Structural (PALM) Polyps (endometrial or cervical) Adenomyosis Leiomyoma Malignancy and hyperplasia Nonstructural (COEIN) Coagulopathy Ovulatory dysfunction Endometrial Iatrogenic Not yet classified |
Information from reference 8.
Dysmenorrhea occurs in 15% to 30% of patients with adenomyosis.6 It is postulated that dysmenorrhea is related to the increased number of oxytocin receptors in the endometrium and increased prostaglandin production contributing to uterine contractions causing dysmenorrhea.5,9 Adenomyosis in a patient with fibroids can cause more severe dysmenorrhea, dyspareunia, or chronic pelvic pain. If a patient with fibroids is having significant dysmenorrhea, dyspareunia, or chronic pelvic pain, an evaluation for adenomyosis may be warranted.
Common signs of adenomyosis include uterine enlargement, uterine tenderness with boggy consistency, and infertility. Coexisting conditions such as leiomyomas could contribute to an enlarged uterus. As many as 50% of patients with adenomyosis have leiomyomas; 11% have endometriosis.6 In patients undergoing hysterectomy for fibroids, adenomyosis was reported in 15% to 57% of specimens.10 In 25% to 70% of patients with endometriosis, adenomyosis was also reported; in patients with deep endometriosis, the prevalence of adenomyosis was found to be 49% to 66%.10
Impaired fertility in adenomyosis is thought to be attributable to abnormal thickening of the junctional zone of the myometrium, abnormal uterine peristalsis, and altered sperm transport.10 Because patients are typically diagnosed with adenomyosis after childbearing age, the incidence of adenomyosis in patients with infertility is unclear. But, a meta-analysis found that patients with adenomyosis had a 28% decreased probability of clinical pregnancy using in-vitro fertilization/intracytoplasmic sperm injection vs. patients without adenomyosis.11
Diagnosis
Diagnosis of adenomyosis is based on clinical suspicion as well as imaging. With improvements in transvaginal ultrasonography and pelvic magnetic resonance imaging (MRI), more cases of adenomyosis are being diagnosed.6,9 Similar to endometriosis, adenomyosis does not have any classic physical examination findings or laboratory studies that identify it as a likely diagnosis. On transvaginal ultrasonography, diffuse adenomyosis has a variable appearance. An attempt by gynecologists to define the various features resulted in the Morphological Uterus Sonographic Assessment criteria.12 These criteria have low reproducibility between imagers and unknown predictive value in the diagnosis of adenomyosis.13,14 Adenomyosis is considered when the uterus is globular in configuration and multiple areas of shadowing are visible, sometimes described as fan shaped, with difficulty differentiating the outer myometrium from the junctional zone and cystic changes in the junctional zone and myometrium12,13 (Figure 1). Additional features that may be seen include an irregular, or interrupted, junctional zone with hyperechoic islands. When multiple signs are present, the diagnosis is more certain; however, adenomyosis can be focal rather than diffuse. When focal adenomyosis occurs, only a few focal areas of shadowing might be visible, which can be confused with fibroids. In these cases, MRI can differentiate adenomyosis from fibroids with greater certainty (Figure 2).
FIGURE 1.
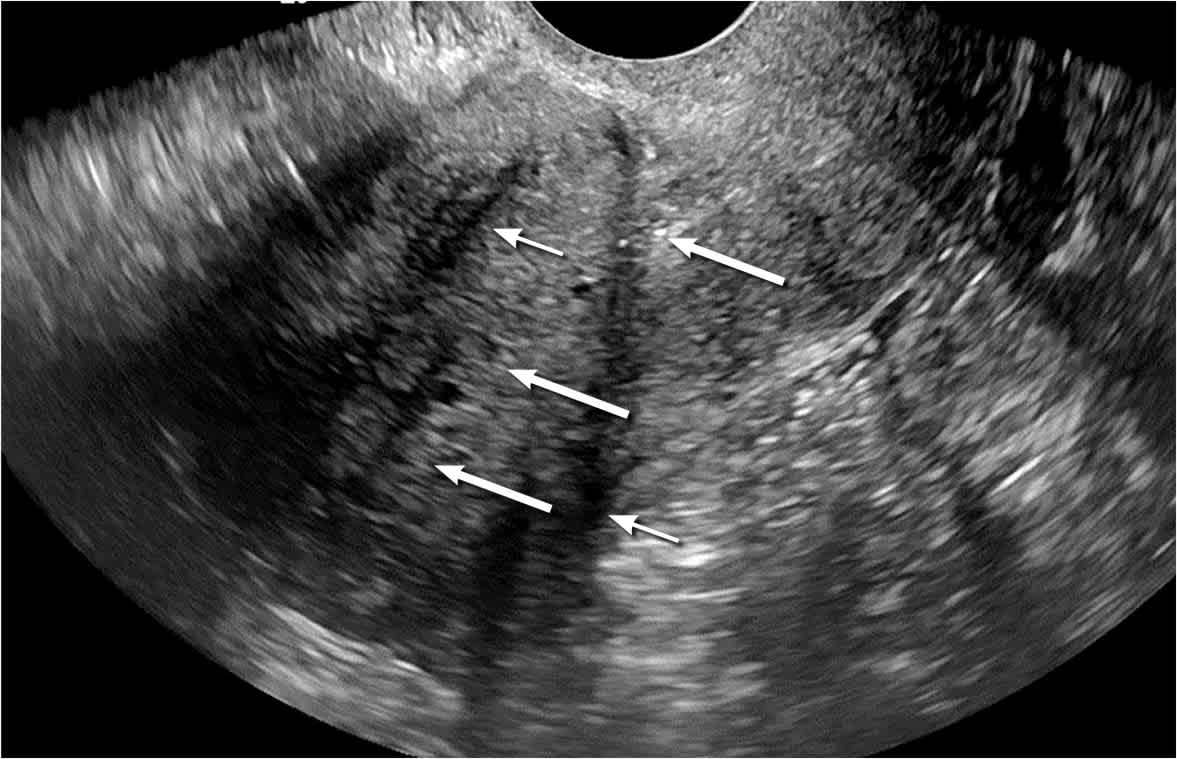
Sagittal gray-scale transvaginal ultrasound image with two focal areas of shadowing (thin arrows), rather than the typical fan-shaped shadowing seen in adenomyosis. The endometrium is distinct from the myometrium (thick arrows); however, the junctional zone is difficult to identify. These imaging findings may represent adenomyosis, although the lack of fan-shaped shadowing and other diagnostic features makes the diagnosis less certain.
FIGURE 2.
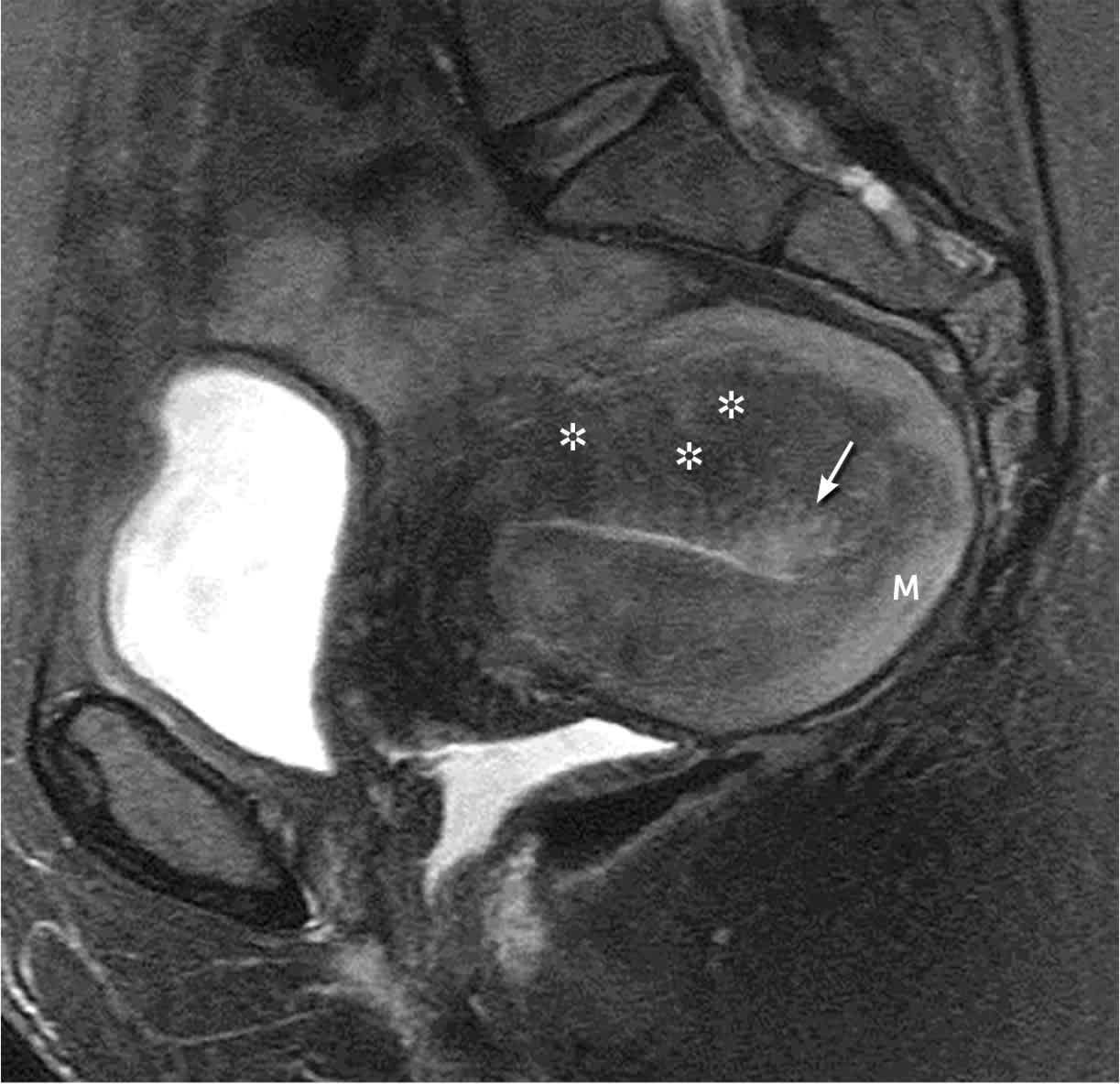
Sagittal T2-weighted magnetic resonance image with signs of adenomyosis, including a widened junctional zone (asterisks) causing thickening of the anterior and posterior myometrium and high signal related to cystic changes and edema (arrow). The normal myometrium (M) is higher in signal intensity than the darker junctional zone (asterisks).
Both a systematic review and meta-analysis demonstrated that transvaginal ultrasonography has a sensitivity for diagnosing adenomyosis of 83.8% and a specificity of 63.9%.15 MRI has a sensitivity of 77% with a specificity of 89%, making it a better confirmatory diagnostic test than transvaginal ultrasonography.16,17 However, because of cost, transvaginal ultrasonography is the first-line imaging technique used in most patients with suspected adenomyosis. In cases where the diagnosis is in question, hysteroscopy may be a helpful adjunct tool. Several changes in the endometrium such as hypervascularization, endometrial defects, and submucosal hemorrhagic cysts can suggest a diagnosis of adenomyosis.10
Treatment
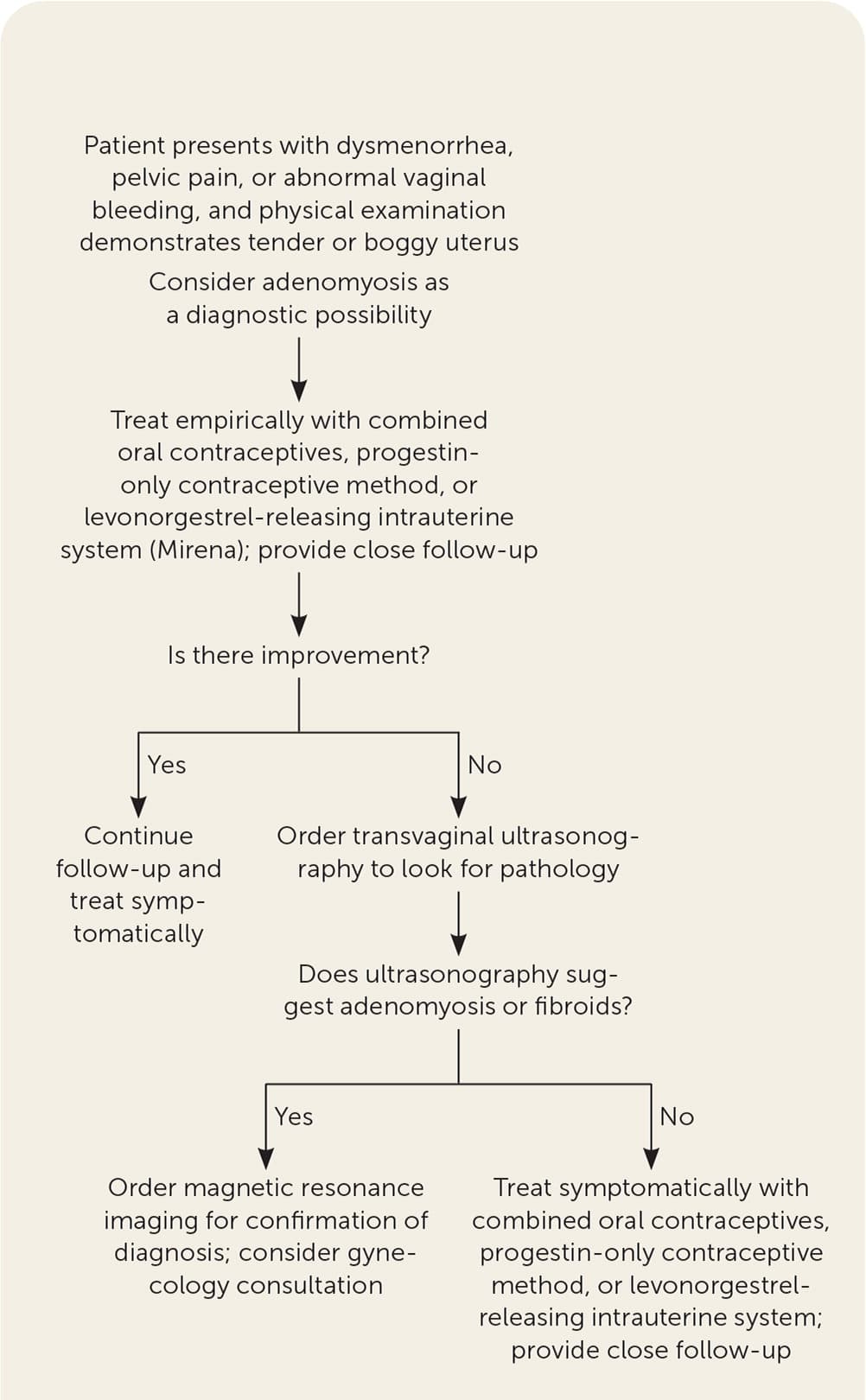
Treatment of adenomyosis focuses on symptom control. There are no medical treatments approved by the U.S. Food and Drug Administration for adenomyosis; however, many medical therapies that are successful in treating endometriosis are also used off label for adenomyosis18 (Table 41,6,18,19 ). Definitive therapy for adenomyosis is hysterectomy if other therapies are not effective. In a large population-based study, more than 80% of women with adenomyosis had a hysterectomy, and almost 40% used chronic pain medications.4 Uterine artery embolization is a potential minimally invasive option to treat focal adenomyosis.20 Figure 3 shows an approach to diagnosis and treatment of adenomyosis.18–22
TABLE 4. Nonsurgical Treatment of Adenomyosis
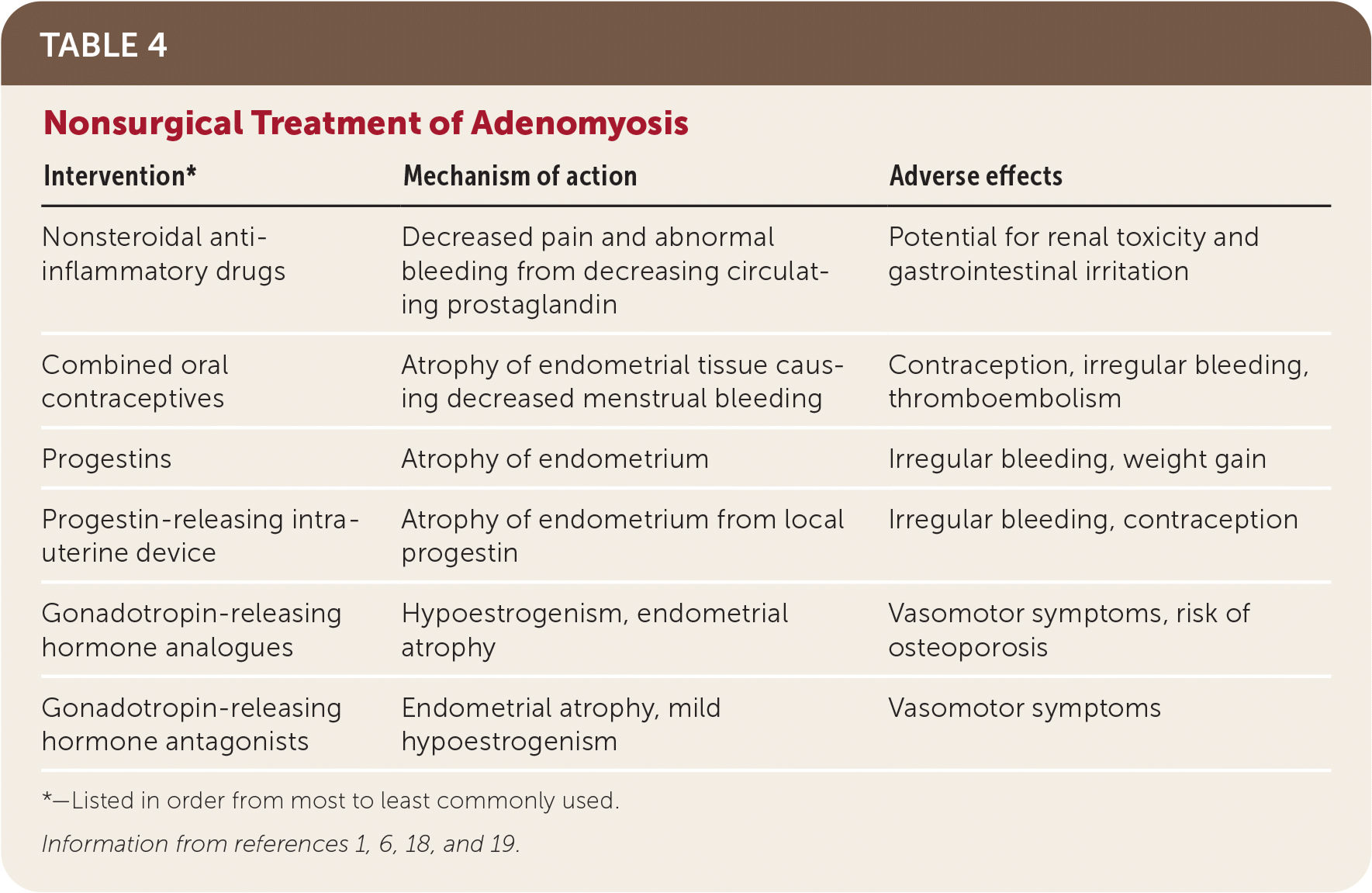
| Intervention* | Mechanism of action | Adverse effects |
|---|---|---|
| Nonsteroidal anti-inflammatory drugs | Decreased pain and abnormal bleeding from decreasing circulating prostaglandin | Potential for renal toxicity and gastrointestinal irritation |
| Combined oral contraceptives | Atrophy of endometrial tissue causing decreased menstrual bleeding | Contraception, irregular bleeding, thromboembolism |
| Progestins | Atrophy of endometrium | Irregular bleeding, weight gain |
| Progestin-releasing intrauterine device | Atrophy of endometrium from local progestin | Irregular bleeding, contraception |
| Gonadotropin-releasing hormone analogues | Hypoestrogenism, endometrial atrophy | Vasomotor symptoms, risk of osteoporosis |
| Gonadotropin-releasing hormone antagonists | Endometrial atrophy, mild hypoestrogenism | Vasomotor symptoms |
*—Listed in order from most to least commonly used.
CASE RESOLUTION
The use of MRI vs. treating symptoms empirically with a progestin-releasing intrauterine device is discussed with your patient. The patient elects to try the device and returns in six months reporting that pain is 50% better; however, one year later the patient reports worsening pain. After gynecology consultation and MRI confirmation of the diagnosis, the patient elects to have a hysterectomy.
Data Sources: A PubMed search was completed in Clinical Queries using the key terms adenomyosis, diagnosis, and treatment. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. The Agency for Healthcare Research and Quality evidence reports, Clinical Evidence, Cochrane database, Essential Evidence Plus, Institute for Clinical Systems Improvement, and DynaMed were also searched. Search dates: November 20, 2020, and November 7, 2021.