Health care–associated infections (HAIs) are a significant cause of morbidity and mortality in the United States. Common examples include catheter-associated urinary tract infections, central line–associated bloodstream infections, ventilator-associated pneumonia, surgical site infections, and Clostridioides difficile infections. Standardized infection control processes and precautions have been shown to reduce the rate of HAIs, and targeted practices for HAIs have shown further reductions. Patient safety tools have been developed for various HAIs to help guide administrators and are free for public use through the Centers for Disease Control and Prevention STRIVE (States Targeting Reduction in Infections via Engagement) initiative. The Choosing Wisely initiative makes best practice recommendations for physicians to improve quality of care and reduce costs; targeted recommendations were developed to reduce the risk of HAIs. For example, using invasive devices only when indicated and for the shortest time possible reduces the risk of device-related HAIs. The goal of antibiotic stewardship is to reduce C. difficile infections and further development of multidrug-resistant organisms such as vancomycin-resistant Enterococcus and carbapenem-resistant Enterobacteriaceae. Antibiotic stewardship targets physician behaviors such as reviewing antibiotic therapy choices every 48 to 72 hours, reviewing culture results as soon as available, de-escalating antibiotic therapy when appropriate, and documenting the indications for initiating and continuing antibiotic therapy.
Health care–associated infections (HAIs) are a significant source of morbidity and mortality in the United States, with approximately 687,000 infections and 72,000 deaths annually, leading to costs in the billions of dollars.1 The Centers for Disease Control and Prevention (CDC) estimates that up to one in 25 patients will develop an HAI.1,2 The CDC tracks and reports data on the occurrence of catheter-associated urinary tract infections (UTIs), central line–associated bloodstream infections (CLABSIs), ventilator-associated pneumonia (VAP), and surgical site infections through the STRIVE (States Targeting Reduction in Infections via Engagement) initiative, with the goal of reducing HAIs.3 SARS-CoV-2 had a negative impact on rates of HAIs; between 2019 and 2020, CLABSIs, catheter-associated UTIs, and VAPs increased by 47%, 19%, and 45%, respectively, without a significant reduction in surgical site infections.4 Standard practices reduce HAIs, and more targeted practices instituted at the local level further reduce HAIs.3 Free tools are available through the STRIVE initiative and CDC that can be implemented and used to reduce HAIs.3
SORT: KEY RECOMMENDATIONS FOR PRACTICE
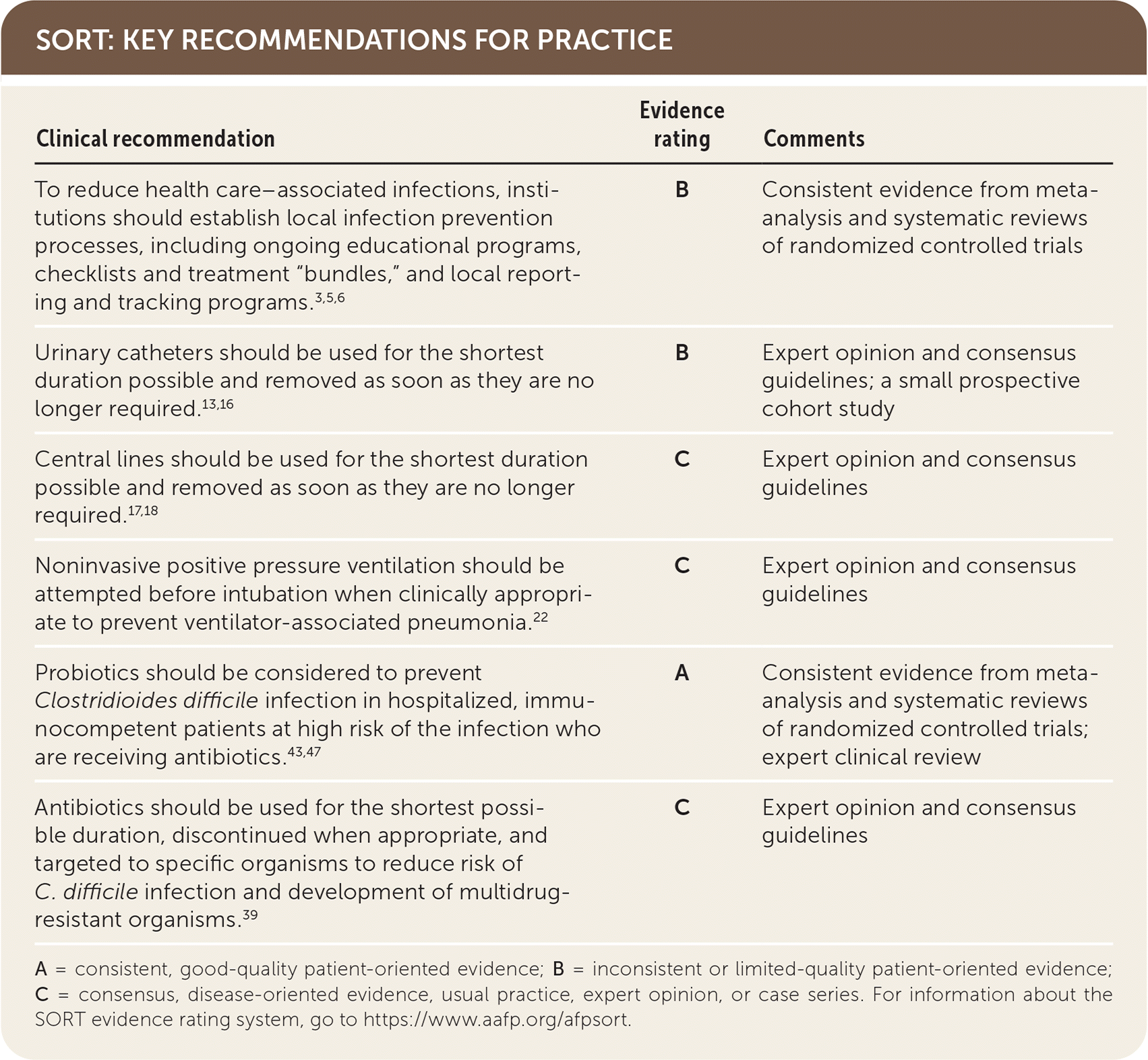
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
BEST PRACTICES IN PREVENTION OF HEALTH CARE–ASSOCIATED INFECTIONS

| Recommendation | Sponsoring organization |
|---|---|
| Catheter-associated urinary tract infection | |
| Do not place or maintain an indwelling urinary catheter in a patient unless there is a specific indication to do so. Do not place an indwelling urinary catheter to manage urinary incontinence. Do not place, or leave in place, urinary catheters for incontinence, convenience, or monitoring of output for noncritically ill patients (acceptable indications: critical illness, obstruction, hospice, in perioperative period for fewer than two days for urologic procedures); use daily weights instead to monitor diuresis. | American Academy of Nursing Society for Post-Acute and Long-Term Care Medicine Society of Hospital Medicine (Adult) |
| Central line–associated bloodstream infection | |
| Avoid invasive devices (including central lines, endotracheal tubes, and urinary catheters) and, if required, use them no longer than necessary. Do not place, or leave in place, peripherally inserted central lines for patient or clinician convenience. | Society for Healthcare Epidemiology of America Society of General Internal Medicine |
| Surgical site infection | |
| Do not continue antibiotics used for surgical prophylaxis after the patient has left the operating room. Do not routinely use topical antibiotics on a surgical wound. | Society for Healthcare Epidemiology of America American Academy of Dermatology |
| Clostridioides difficile infections/multidrug-resistant organisms | |
| Do not use antibiotics in patients without convincing evidence of need. Do not continue antibiotics beyond 72 hours in hospitalized patients unless patient has clear evidence of infection. | Society for Healthcare Epidemiology of America Society for Healthcare Epidemiology of America |
| C. difficile infections/ventilator-associated pneumonia | |
| Do not prescribe medications for stress ulcer prophylaxis to medical inpatients unless they are at high risk of gastrointestinal complications (e.g., high risk of gastrointestinal ulcer bleeding). Do not continue hospital-prescribed stress ulcer prophylaxis with proton pump inhibitor therapy in the absence of an appropriate diagnosis in the postacute and long-term care population. For pharmacologic treatment of patients with gastroesophageal reflux disease, long-term acid suppression therapy (proton pump inhibitors or histamine H2 blockers) should be titrated to the lowest effective dose to achieve therapeutic goals. | Society of Hospital Medicine (Adult) Society for Post-Acute and Long-Term Care Medicine American Gastroenterological Association |
Source: For more information on the Choosing Wisely Campaign, see https://www.choosingwisely.org. For supporting citations and to search Choosing Wisely recommendations relevant to primary care, see https://www.aafp.org/afp/recommendations/search.htm.
The health care environment increases the risk of the development and spread of multidrug-resistant bacteria, leading to increased difficulty in treating HAIs.3 Targeted practices should be aimed at reducing HAIs and the development of multidrug-resistant pathogens.2
General Recommendations
Health care institutions should establish local infection prevention processes, including ongoing educational programs, institutional checklists and treatment “bundles” (a combination of developed interventions that help reduce HAIs),3 and local reporting and tracking programs.5,6 Multiple professional organizations have made specific and targeted recommendations to reduce HAIs. Good hand hygiene and proper use of personal protective equipment are necessary.7 Guidelines for preventing HAIs are available from the CDC at https://www.cdc.gov/hai/prevent/prevention.html. The Society for Healthcare Epidemiology of America offers further recommendations and resources at https://shea-online.org/compendium-of-strategies-to-prevent-healthcare-associated-infections-in-acute-care-hospitals/.
SARS-CoV-2
Current evidence supports social distancing, masking, and vaccination to prevent or reduce HAIs related to SARS-CoV-2.8 The CDC recommends adhering to prevention practices for airborne diseases, which include having all individuals wear a face mask while in any indoor clinical space, placing the infected patient in an airborne infection isolation room if available, limiting visitation, and having health care workers use additional appropriate personal protective equipment (e.g., N95 respirator, goggles, gown, hair coverings, gloves).8 If an airborne infection isolation room is not available, private rooms can also reduce the spread of airborne diseases.8 Personal protective equipment should be donned and doffed using appropriate techniques before and after each patient encounter to reduce the risk of HAIs caused by SARS-CoV-2.8 A CDC educational video on a donning technique is available at https://youtu.be/H4jQUBAlBrI. Health care workers should also be immunized against COVID-19 and other vaccine-preventable communicable diseases to prevent or reduce spread.9
Mask wearing by health care workers has been demonstrated to reduce the spread of airborne diseases in health care environments.10,11 Effectiveness of cloth masks is unclear given the variability of material and construction of the masks, and they should not be worn during patient care when appropriate medical masks or N95 respirator masks are available.10,11
The CDC recommends screening (e.g., symptom and exposure questions, temperature checks) all patients entering a clinical building. Telemedicine should be used when available and appropriate.8 Following CDC recommendations and instituting local policies and practices are effective in preventing HAIs from SARS-CoV-2.12
Catheter-Associated UTIs
Catheter-associated UTIs are diagnosed in patients with symptomatic bacteriuria following bladder catheterization. UTIs account for more than 30% of nosocomial infections, of which most are associated with bladder instrumentation.13 More than 26,000 catheter-associated UTIs occurred in 2019 in acute care facilities, a decrease of 8% from 2018.1,14,15
Avoiding unnecessary bladder catheterization and using an appropriate placement technique are paramount in the prevention of catheter-associated UTIs. Bladder catheters should be used in hospitalized patients only for acute urinary retention, bladder outlet obstruction, accurate output measurement when critically ill, prolonged immobilization, and end-of-life care. The catheter should be placed using a sterile technique, and urine flow should be unobstructed with the collection system always remaining closed and below the level of the bladder.13 One study demonstrated that shorter duration of indwelling catheter use reduced the risk of catheter-associated UTI.16 No patients developed a catheter-associated UTI with less than three days of catheterization, whereas the rate of catheter-associated UTIs was 15% for three to six days and 68% for more than eight days of catheterization.16 Bladder catheters should be removed as soon as clinically indicated.16
Central Line–Associated Bloodstream Infections
CLABSIs are laboratory-confirmed nosocomial infections that can be diagnosed if occurring at least 48 hours after initial central line placement, the central line remains in place or was removed no more than one day before the date the positive blood culture was drawn, and there is no other source of infection. Central lines are the most common source of bloodstream HAIs, accounting for more than 250,000 infections in the United States annually.17 Risk of CLABSIs is lower with permanent central lines such as implanted ports and dialysis catheters than with nontunneled central lines, which are temporary and include peripherally inserted central catheters and central lines placed in subclavian, internal jugular, or femoral veins.
Nonmodifiable risk factors for CLABSIs include younger age (particularly neonates), male gender (guideline did not define how gender was determined), and underlying immunodeficiency.18 Modifiable risk factors include prolonged hospitalization before central-line insertion, use of multilumen catheters, receipt of parenteral nutrition, and placement and insertion techniques.18 If a central line is indicated, a subclavian vein site is associated with the lowest CLABSI risk.17 Central line placement can introduce infection from skin flora, either during insertion of the catheter or while handling the indwelling catheter with unclean hands18 (Figure 1).
FIGURE 1.
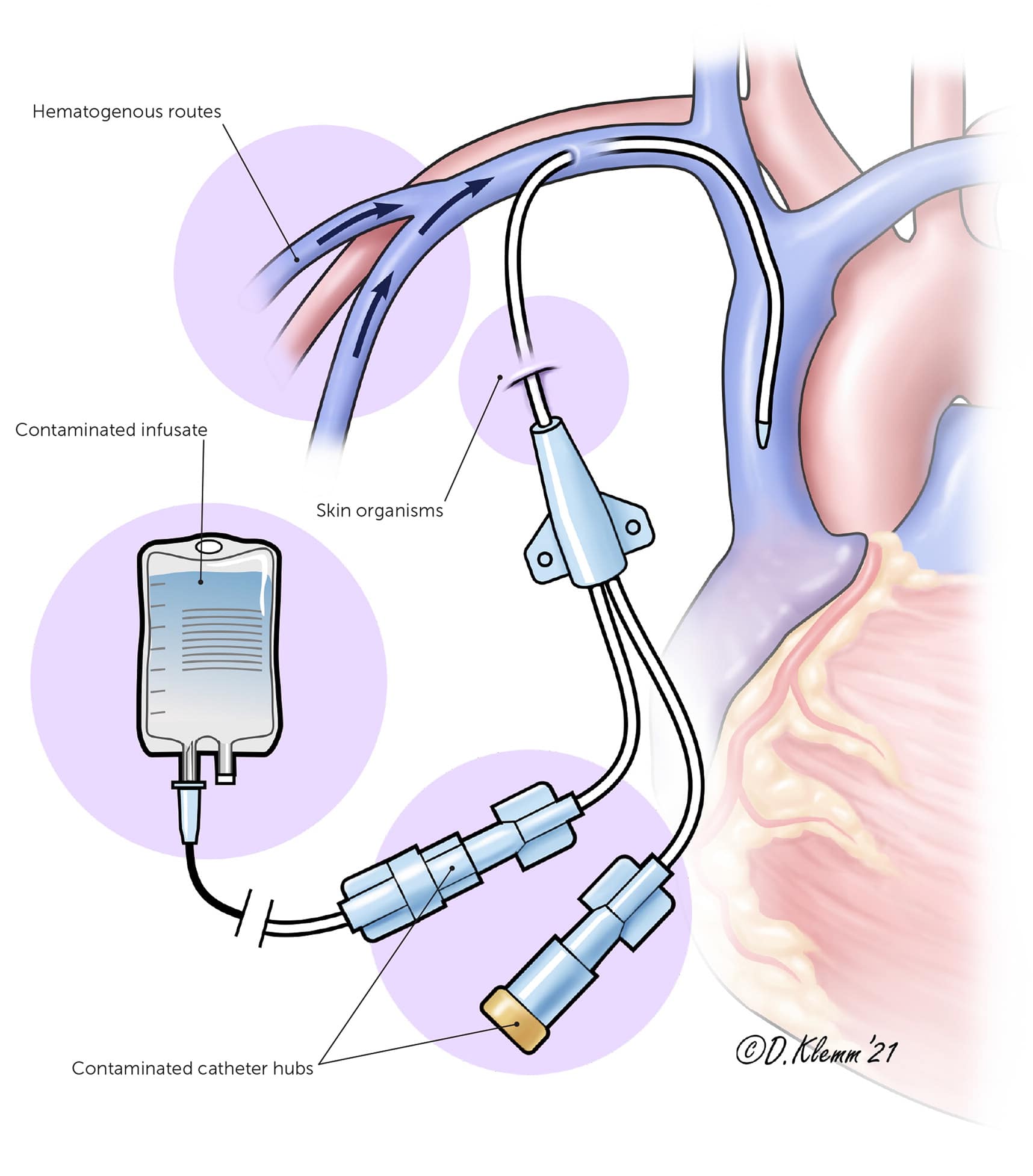
Common routes of contamination from central line placement.
Illustration by Dave Klemm
To reduce HAIs, the sterile barrier technique should be used, including full-body drape for the patient and cap, mask, sterile gown, and gloves for the physician. Skin preparation using greater than 0.5% chlorhexidine solution is preferred, followed by postprocedure chlorhexidine dressings.19
Central lines should be used for the shortest duration possible and removed as soon as they are no longer required.17,18 Administration of prophylactic antibiotics before central-line insertion has not been shown to reduce CLABSIs.20 Flushing or locking tunneled catheters with a combination of heparin and an antibiotic appears to reduce gram-positive catheter-related sepsis in patients at risk of neutropenia. However, this should be considered only in patients at institutions with an elevated baseline CLABSI rate of greater than 15%.20 There are insufficient data to recommend a specific frequency of postprocedure dressing changes to reduce the risk of CLABSIs.20,21
Ventilator-Associated Pneumonia
VAP refers to pneumonia that occurs after initiating mechanical ventilation and can be clinically challenging to diagnose because of issues with the accuracy and reproducibility of available testing. Historically, 10% to 20% of patients receiving mechanical ventilation have developed VAP, with a mortality rate of approximately 10%.22 Organisms commonly associated with these infections include Staphylococcus aureus and Pseudomonas aeruginosa; therefore, targeted antibiotics can improve treatment outcomes and decrease microbial resistance.22
The risk of VAP can be reduced by avoiding intubation and using noninvasive positive pressure ventilation when possible. Noninvasive positive pressure ventilation in acute respiratory failure caused by chronic obstructive pulmonary disease or congestive heart failure exacerbations can decrease the need or shorten the overall duration of mechanical ventilation, decrease length of hospital stay, and lower mortality rates from VAP. Minimizing sedation in patients receiving mechanical ventilation also reduces HAIs because this practice reduces the average duration of ventilator use by two to four days.22
Using local treatment bundles or checklists (e.g., for daily assessment of readiness for extubation) can help reduce HAIs.22 There is weak evidence that elevating the head of the bed to 30 to 45 degrees for intubated patients reduces VAP risk and that enteral feeding in the supine position should be avoided.22 A 2020 Cochrane review demonstrated that use of chlorhexidine (Peridex) mouthwash or gel in critically ill patients reduces VAP but not the duration of mechanical ventilation, intensive care unit (ICU) stay, or overall mortality.23 Given the association between histamine H2 blockers and proton pump inhibitors and an increased risk of VAP, routine use should be reserved for patients at high risk of gastrointestinal ulcer bleeding.24
Surgical Site Infections
Surgical site infections are those that occur following a surgical procedure and involve the corresponding incision, organ, or space.25 It is estimated that up to 55% of surgical site infections are preventable. 26,27 Recommendations for preventing these infections include surgery-specific antibiotic prophylaxis based on available guidelines, perioperative glycemic control (goal of less than 180 to 200 mg per dL [9.99 to 11.10 mmol per L] for patients with or without diabetes mellitus), full-body bathing no earlier than the night before the operation (with or without antiseptics), and intraoperative skin preparation with antiseptics.27–29
Methicillin-resistant S. aureus (MRSA) is the most common organism identified in surgical site infections, and nares colonization with S. aureus increases the rate of surgical site infections five- to 10-fold.30,31 Studies have shown that patients who screen positive for S. aureus have lower rates of surgical site infections when decolonization protocols are followed before orthopedic and cardiothoracic surgery.28,29,32 However, this practice should not be used universally for other surgeries because of concern about the development of mupirocin- or chlorhexidine-resistant bacteria.32 Policies regarding screening for S. aureus can be institution-specific based on local rates of S. aureus colonization and surgical site infections.33
Studies have not demonstrated superiority of one type of postsurgical dressing in preventing surgical site infections.34,35 Guidelines recommend using antimicrobial sutures, especially when there is high risk of surgical site infection, such as with intracolorectal surgeries, and when there is lower risk but infection may be devastating, such as with cardiothoracic surgery.27,28
Clostridioides difficile Infections
Clostridioides difficile (formerly Clostridium difficile) is the bacteria most commonly associated with HAIs.26 Although the incidence of community-acquired C. difficile infections is rising, most cases continue to be HAIs.1 Additionally, the virulence of C. difficile is increasing, particularly among strains that are resistant to metronidazole (Flagyl), which has led to the addition of oral vancomycin or fidaxomicin (Dificid) as first-line agents in treatment guidelines.36,37
The main strategies for reducing C. difficile HAI include patient isolation with initiation of contact precautions and appropriate hand hygiene when infection is suspected or confirmed. Environmental cleaning performed daily and terminally with C. difficile sporicidal agents is recommended and has been shown to decrease risk of transmission in the environment and by health care workers.38
Health care workers and surfaces in rooms that previously contained a patient with C. difficile are known vectors and increase the risk of subsequent C. difficile infections.39,40 There is increasing evidence that the use of disinfecting ultraviolet light systems, along with standard sporicidal practices, may decrease the risk of subsequent infections in these high-risk rooms.35,39,41 Guidelines recommend handwashing with nonantimicrobial or antimicrobial soap rather than alcohol-based hand rubs, but superiority has not been demonstrated in clinical trials.42,43
Several antibiotics have been associated with a high risk of C. difficile infection, with clindamycin demonstrating the highest risk compared with no antibiotics.44,45 If C. difficile infection is diagnosed while a patient is taking an antibiotic, the medication should be discontinued as soon as the clinical situation allows.39 Antibiotic stewardship is estimated to reduce rates of C. difficile infections by as much as 50% to 77%.43,46
Emerging evidence suggests using probiotics when prescribing antibiotics for prevention of C. difficile infections.43,47 A systematic review of 31 randomized controlled trials demonstrated a number needed to treat of 12 to prevent one case of C. difficile–associated diarrhea in those considered at high risk of C. difficile infection.47 Given their relative safety profile, probiotics could be considered in an immunocompetent patient at high risk of C. difficile infection.47
Antibiotic Stewardship
Antibiotic stewardship is a cornerstone of HAI prevention (Table 148 ). The CDC estimates that nearly 30% of all antibiotics prescribed for inpatients are unnecessary or suboptimal, thereby increasing the risk of C. difficile infection and multidrug-resistant organisms. 44 Appropriate antibiotic prescribing can improve outcomes while minimizing patient harms.
TABLE 1. Physician and Pharmacy Interventions for Improving Hospital Antibiotic Stewardship

| Review all antibiotic therapy for appropriateness every 48 to 72 hours following initiation. |
| Document indications for initiating and continuing antibiotic therapy. |
| Order appropriate diagnostic tests for suspected infection, including imaging and cultures. |
| Review culture results often, and when culture results are available to guide therapy, use the narrowest-spectrum antibiotics for the shortest duration possible based on local resistance markers and susceptibilities; consider pathologist or laboratory comments regarding whether colonization or contamination is possible. |
| When culture results are not available to guide therapy, consider use of society guidelines to direct antibiotic choice, frequency, and duration. |
| Use electronic health record–enabled antibiotic stop dates to ensure appropriate duration of therapy. |
| If the patient reports a penicillin allergy, consider assessing the nature of the allergy and reaction. |
| Transition from intravenous to oral antibiotics when clinically appropriate. |
Information from reference 48.
The CDC recommends using proper diagnostic testing when evaluating suspected infection to narrow therapy when appropriate and documenting the indications for initiating and continuing antibiotic therapy.44 Guidelines, such as those from the Infectious Diseases Society of America (https://www.idsociety.org/practice-guideline/implementing-an-ASP/), should be used to guide choice of antibiotic, dosing, frequency, and duration.44 Given the uncertainty surrounding many patient-reported penicillin allergies, physicians can use tools such as PEN-FAST or formal allergy testing to assess true risk, potentially allowing for future use of beta-lactam antibiotics.49
Multidrug-Resistant Organisms
Multidrug-resistant organisms are those that are resistant to one or more commonly used antibiotics. The most common of these organisms include MRSA and vancomycin-resistant Enterococcus. More recently, carbapenem-resistant Enterobacteriaceae have been declared an urgent public health threat by the CDC.50 Although development of carbapenem-resistant Enterobacteriaceae highlights the dangerous problem of antibiotic resistance, the national incidence of this infection has remained low at approximately 0.3 to 2.93 cases per 100,000 person-years.51
Strategies for preventing infections with multidrug-resistant organisms are similar to those for preventing C. difficile infections: patient isolation when possible, contact precautions and appropriate hand hygiene for patients colonized or infected with a multidrug-resistant organism, environmental cleaning daily and terminally, and antibiotic stewardship.52 The CDC recommends decolonization protocols for ICU patients who are found to be colonized with MRSA, but evidence is lacking regarding protocols for other multidrug-resistant organisms.52
There is evidence that bathing ICU patients daily with chlorhexidine decreases the rate of MRSA colonization and infection and the rate of vancomycin-resistant Enterococcus colonization compared with usual bathing protocols.30 However, the benefit of this practice for the general inpatient population is unclear because the meta-analysis included only studies performed in the ICU.30,53 Strains of MRSA that are resistant to chlorhexidine and mupirocin have been reported, but clinical relevance is unclear because rates of MRSA infection have decreased as resistance has increased.30 Development of disinfectant-resistant organisms is possible with repeated exposure but is thought to be unlikely in the clinical setting where higher concentrations are used.54
Data Sources: PubMed searches were completed using the key terms cross infection/prevention and control, drug resistance, multiple bacterial, Clostridium difficile, Clostridioides difficile, pneumonia, ventilator-associated, urinary catheterization, central venous catheter, SARS-CoV-2, COVID-19, and hospital. We also searched Essential Evidence Plus and Clinical Evidence, the Cochrane Library, the Centers for Disease Control and Prevention, and the Society for Healthcare Epidemiology of America. Search dates: October to December 2020; January 2021.
The views expressed are those of the authors and do not reflect the official policy or position of the Departments of the Army or Air Force, the Department of Defense, or the U.S. government.

