Multiparametric magnetic resonance imaging (mpMRI) is a specific MRI test used to detect and evaluate prostate cancer. It requires additional radiologist training and sequences not routinely performed in anatomic imaging, such as diffusion-weighted and dynamic contrast-enhanced imaging.1 The National Comprehensive Cancer Network and the American Urological Association support using mpMRI for risk stratification before the first prostate biopsy, to perform targeted prostate biopsy, and in active surveillance.2,3

| Test | Indication | Population and frequency | Cost* |
|---|---|---|---|
| Multiparametric magnetic resonance imaging | Risk stratification for targeted biopsy; active surveillance of low-risk prostate cancer | Patients 50 years or older with suspected or known prostate cancer, frequency of test varies | $275 to $444 |
*—Payment rate according to the 2022 Centers for Medicare and Medicaid Services physician fee schedule (national payment amount for nonfacility cost).14
Accuracy
The prostate imaging reporting and data system, second revision (PI-RADSv2) provides standardized interpretation and reporting of mpMRI.1 A score between 1 and 5 is assigned by a radiologist (1 suggests that a clinically significant cancer is highly unlikely, and 5 suggests that it is highly likely). In a systematic review of 21 studies including 3,857 patients, the PI-RADSv2 tool (using a threshold score of 3 or 4) had a pooled sensitivity of 0.89 (95% CI, 0.86 to 0.92) and specificity of 0.73 (95% CI, 0.60 to 0.83) for the detection of prostate cancer.4
Inclusion of mpMRI information into a multivariate risk-prediction calculator improves the accuracy of cancer risk assessment and can assist in shared decision-making regarding management options.5
The strongest support for prebiopsy mpMRI has been derived from a systematic review of 15 studies (n = 2,293) in which mpMRI plus targeted biopsy was compared with transrectal ultrasound (TRUS)–guided systematic biopsy alone.6 By targeting suspicious areas of the prostate, the median rate of significant prostate cancer detection was 33% with mpMRI vs. 24% with biopsy alone. The targeted approach was also more efficient, requiring only nine core biopsies instead of 37. In addition, only 2% of clinically significant cancer was missed with mpMRI vs. 9% with TRUS-guided biopsy. Therefore, prebiopsy mpMRI can improve the diagnostic yield of a patient's first prostate biopsy.
Benefit
In a randomized controlled trial of 1,532 patients with prostate-specific antigen levels of 3 ng per mL (3 mcg per L) or greater, clinically significant prostate cancer was diagnosed in a similar percentage of patients who had mpMRI plus targeted biopsy as those who had TRUS-guided systematic biopsy alone (21% vs. 18%; risk difference = 3%; 95% CI, −1% to 7%). Additionally, clinically insignificant cancer was detected less often in the mpMRI plus targeted biopsy group compared with the TRUS-guided systematic biopsy group (4% vs. 12%; risk difference = −8%; 95% CI, −11% to −5%).7
A prospective cohort of 172 patients in whom cancer was suspected despite previous negative biopsies underwent mpMRI plus targeted biopsy and TRUS-guided systematic biopsy. Targeted biopsy detected clinically significant prostate cancer (Gleason score of 7 or more) more often than systematic biopsy (16% vs. 9%; P = .01).8
Based on improved risk assessment with the use of mpMRI and the test's high sensitivity, there is future potential to avoid unnecessary biopsies in patients with a low-risk result (PI-RADS score of 2 or less). Widespread adoption of this approach is not yet recommended because of dependence on operator experience and significant interobserver variability.9 Longer-term studies demonstrating the optimal biopsy threshold and safety are needed.
Harms
In 2017, the U.S. Food and Drug Administration started requiring a warning with gadolinium-based contrast agents because they may be partially retained in brain tissue for months to years after use.10 The only established complication of gadolinium-based contrast is nephrogenic systemic fibrosis, which affected up to 0.07% of patients with stage 4 or 5 chronic kidney disease in one large meta-analysis.11 The long-term consequences of gadolinium retention are otherwise unknown and require further safety studies.
Prostate cancer overdiagnosis remains a problem, but studies suggest that it is improved with the use of mpMRI.9
Cost
Studies suggest that mpMRI could be cost-effective at $23,483 per quality-adjusted life-year, although this conclusion depends on the assumption that a negative mpMRI result could be used to safely avoid biopsy.12
Coding for MRI-TRUS targeted biopsy is complicated. The test is considered investigational by many insurance carriers, and there is no specific Current Procedural Terminology (CPT) code for it.13 CPT codes for the MRI and 3D rendering are billed by a radiologist, whereas the codes that urologists use for a biopsy are not changed by the use of mpMRI. The added radiologic costs and related CPT codes are listed in Table 1.14
TABLE 1. Additional Costs Related to Multiparametric MRI of the Prostate
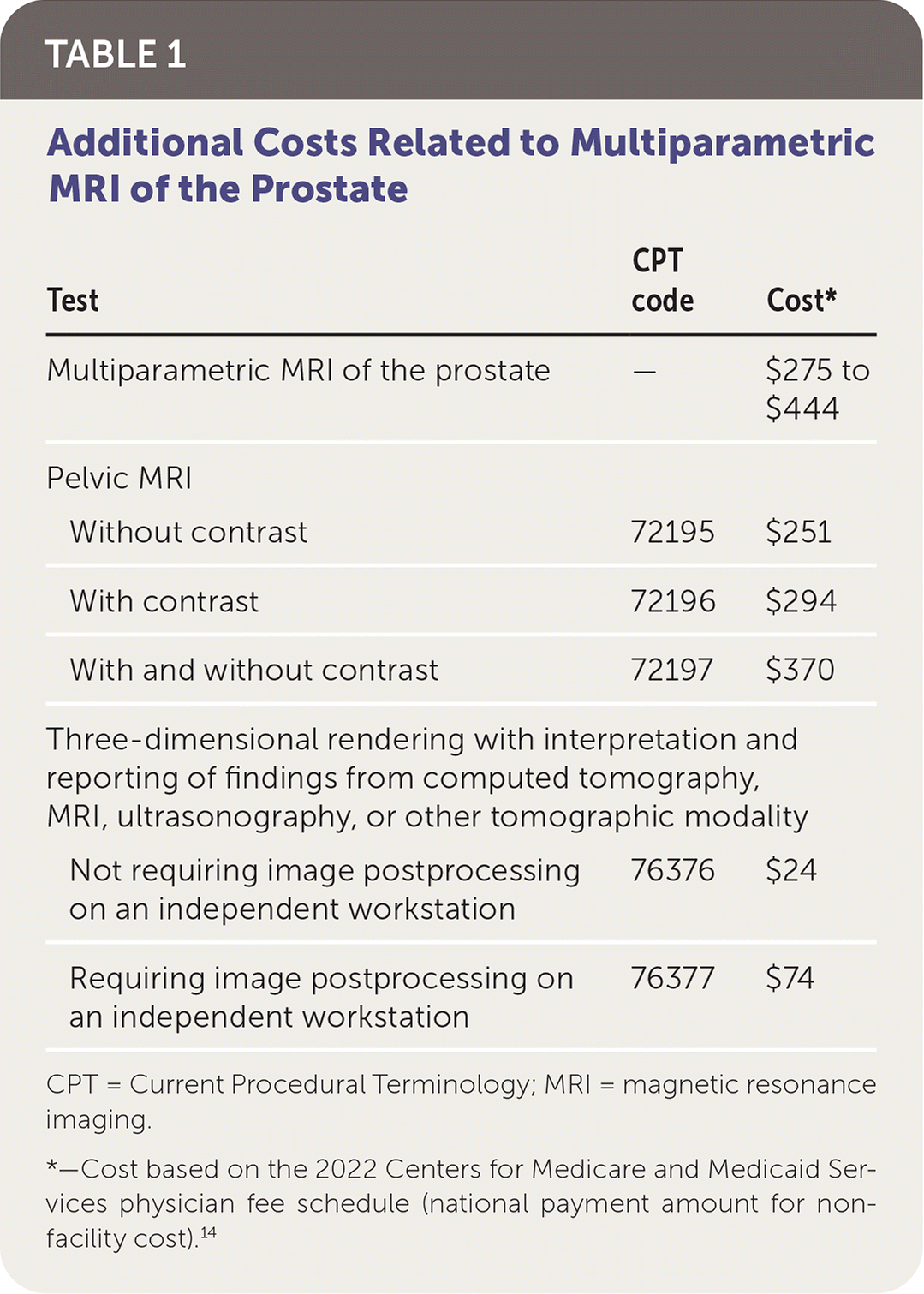
| Test | CPT code | Cost* |
|---|---|---|
| Multiparametric MRI of the prostate | — | $275 to $444 |
| Pelvic MRI | ||
| Without contrast | 72195 | $251 |
| With contrast | 72196 | $294 |
| With and without contrast | 72197 | $370 |
| Three-dimensional rendering with interpretation and reporting of findings from computed tomography, MRI, ultrasonography, or other tomographic modality | ||
| Not requiring image postprocessing on an independent workstation | 76376 | $24 |
| Requiring image postprocessing on an independent workstation | 76377 | $74 |
CPT = Current Procedural Terminology; MRI = magnetic resonance imaging.
*—Cost based on the 2022 Centers for Medicare and Medicaid Services physician fee schedule (national payment amount for non-facility cost).14
Bottom Line
In patients with known or suspected prostate cancer, mpMRI may improve care by providing an individualized assessment of clinically significant cancer risk and by improving the yield of prostate biopsy. However, no studies have evaluated the effects of mpMRI use on morbidity or mortality.
The authors thank Sanjay Ramakumar, MD, for his assistance in the preparation of this manuscript.

