Polymyalgia rheumatica and giant cell arteritis are inflammatory conditions that occur predominantly in people 50 years and older, with peak incidence at 70 to 75 years of age. Polymyalgia rheumatica is more common and typically presents with constitutional symptoms, proximal muscle pain, and elevated inflammatory markers. Diagnosis of polymyalgia rheumatica is clinical, consisting of at least two weeks of proximal muscle pain, constitutional symptoms, and elevated erythrocyte sedimentation rate or C-reactive protein. Treatment of polymyalgia rheumatica includes moderate-dose glucocorticoids with a prolonged taper. Giant cell arteritis, also known as temporal arteritis, usually presents with new-onset headache, visual disturbances or changes, constitutional symptoms, scalp tenderness, and temporal artery symptoms. Inflammatory markers are markedly elevated. Temporal arterial biopsy should be used for diagnosis. However, color duplex ultrasonography, magnetic resonance imaging, and fluorodeoxyglucose positron emission tomography may be helpful when biopsy is negative or unavailable. All patients with suspected giant cell arteritis should receive empiric high-dose glucocorticoids because the condition may lead to blindness if untreated. Tocilizumab is approved by the U.S. Food and Drug Administration for giant cell arteritis and should be considered in addition to glucocorticoids for initial therapy. Polymyalgia rheumatica and giant cell arteritis respond quickly to appropriate dosing of glucocorticoids but typically require prolonged treatment and have high rates of relapse; therefore, monitoring for glucocorticoid-related adverse effects and symptoms of relapse is necessary. Methotrexate may be considered as an adjunct to glucocorticoids in patients with polymyalgia rheumatica or giant cell arteritis who are at high risk of relapse.
Polymyalgia rheumatica (PMR) and giant cell arteritis (GCA) are related inflammatory conditions, which typically affect people 50 years and older. This article provides a summary and review of the best available patient-oriented evidence for each condition.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
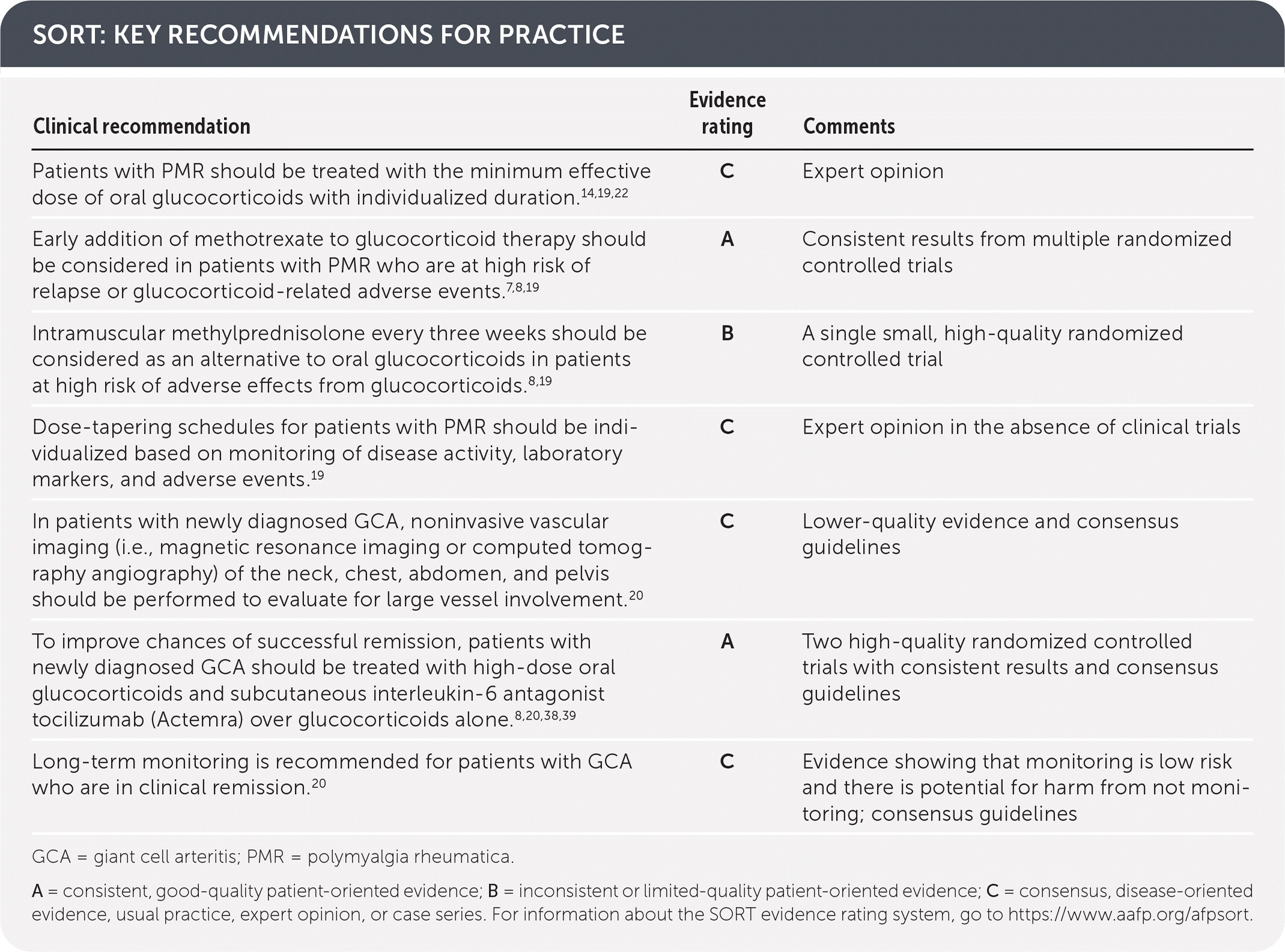
GCA = giant cell arteritis; PMR = polymyalgia rheumatica.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Epidemiology
- Incidences of PMR and GCA vary among people 50 years and older depending on geographic location. The incidence per 100,000 people 50 years and older ranges from 2 (Korea) to 113 (Norway) for PMR and 1 (Turkey) to 44 (Iceland) for GCA.1
- Approximately 20% of patients with PMR have GCA, and about 50% of patients with GCA have PMR.2,3
- Risk factors for PMR and GCA include female sex, Northern European ancestry, and age.4–6
- The age of onset for both conditions is typically 50 years and older, with peak incidence at 70 to 75 years of age.2,3
Polymyalgia Rheumatica
DIAGNOSIS
TABLE 1. Typical Clinical Presentation of Polymyalgia Rheumatica
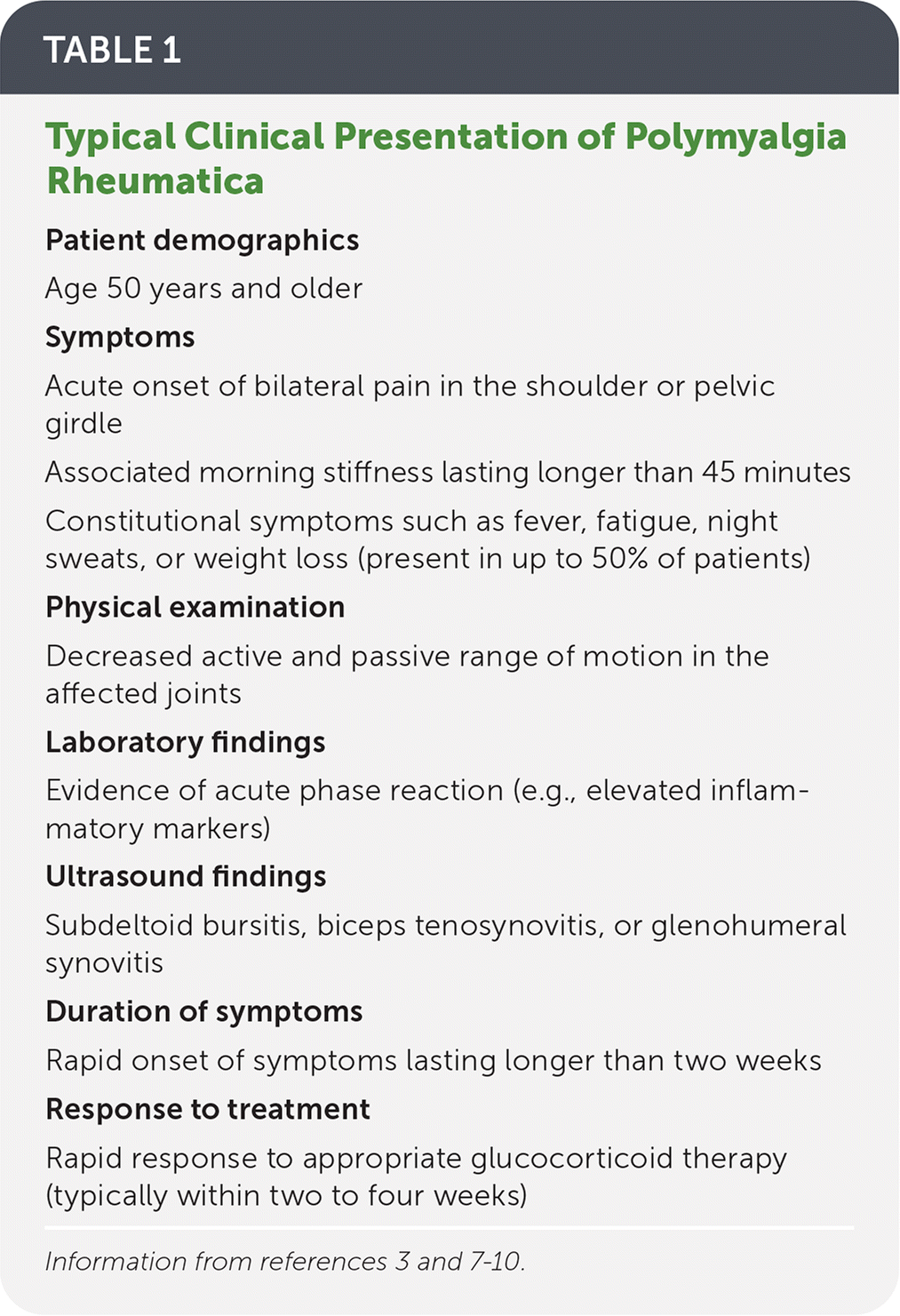
| Patient demographics |
| Age 50 years and older |
| Symptoms |
| Acute onset of bilateral pain in the shoulder or pelvic girdle |
| Associated morning stiffness lasting longer than 45 minutes |
| Constitutional symptoms such as fever, fatigue, night sweats, or weight loss (present in up to 50% of patients) |
| Physical examination |
| Decreased active and passive range of motion in the affected joints |
| Laboratory findings |
| Evidence of acute phase reaction (e.g., elevated inflammatory markers) |
| Ultrasound findings |
| Subdeltoid bursitis, biceps tenosynovitis, or glenohumeral synovitis |
| Duration of symptoms |
| Rapid onset of symptoms lasting longer than two weeks |
| Response to treatment |
| Rapid response to appropriate glucocorticoid therapy (typically within two to four weeks) |
TABLE 2. Differential Diagnosis for Polymyalgia Rheumatica and Giant Cell Arteritis
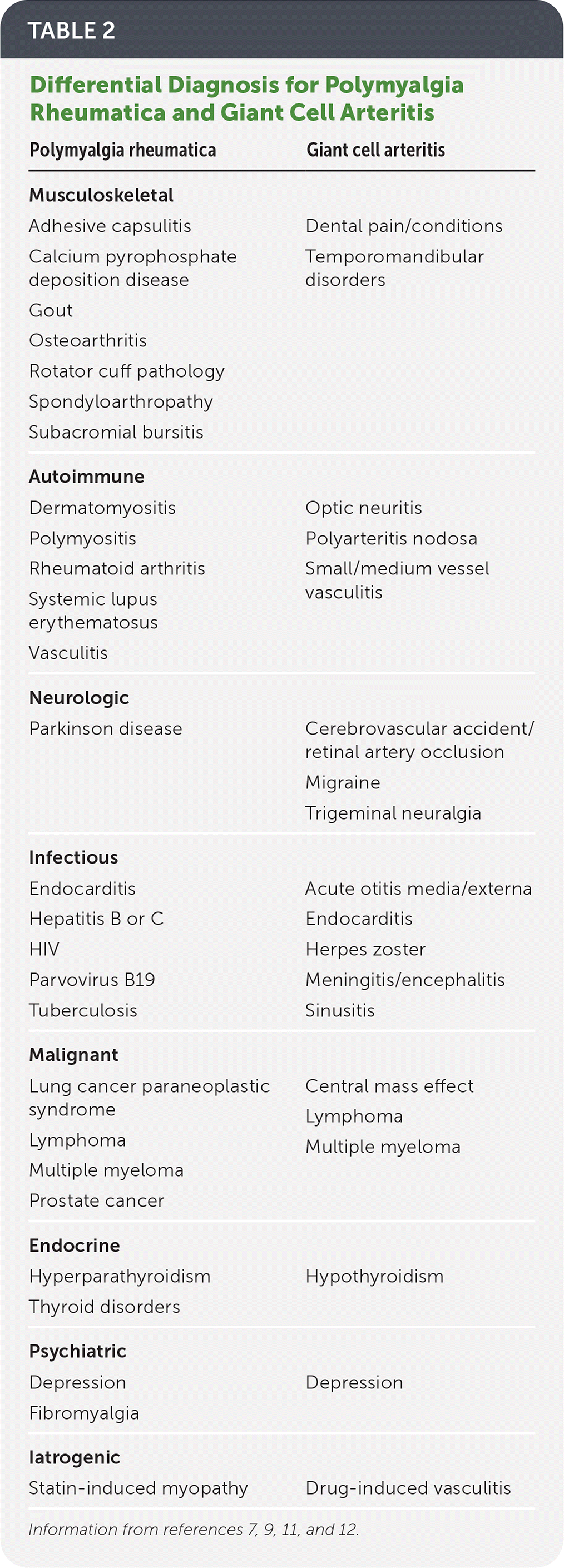
| Polymyalgia rheumatica | Giant cell arteritis |
|---|---|
| Musculoskeletal | |
| Adhesive capsulitis Calcium pyrophosphate deposition disease Gout Osteoarthritis Rotator cuff pathology Spondyloarthropathy Subacromial bursitis | Dental pain/conditions Temporomandibular disorders |
| Autoimmune | |
| Dermatomyositis Polymyositis Rheumatoid arthritis Systemic lupus erythematosus Vasculitis | Optic neuritis Polyarteritis nodosa Small/medium vessel vasculitis |
| Neurologic | |
| Parkinson disease | Cerebrovascular accident/retinal artery occlusion Migraine Trigeminal neuralgia |
| Infectious | |
| Endocarditis Hepatitis B or C HIV Parvovirus B19 Tuberculosis | Acute otitis media/externa Endocarditis Herpes zoster Meningitis/encephalitis Sinusitis |
| Malignant | |
| Lung cancer paraneoplastic syndrome Lymphoma Multiple myeloma Prostate cancer | Central mass effect Lymphoma Multiple myeloma |
| Endocrine | |
| Hyperparathyroidism Thyroid disorders | Hypothyroidism |
| Psychiatric | |
| Depression Fibromyalgia | Depression |
| Iatrogenic | |
| Statin-induced myopathy | Drug-induced vasculitis |
History and Physical Examination
- The patient history reveals predominantly symmetrical shoulder and thigh stiffness, which may cause difficulties in rising from a seated position and raising arms overhead.3
- Up to 50% of patients report constitutional symptoms such as fever, fatigue, night sweats, or weight loss.3
- Physical examination may show restricted active range of motion and weakness or tenderness in the hip and shoulder muscles.
Laboratory Studies
- An erythrocyte sedimentation rate (ESR) of more than 30 to 40 mm per hour or elevated C-reactive protein (CRP) levels are present in approximately 90% of patients with PMR.13,14
- Complete blood count often shows associated normochromic anemia and thrombocytosis.4 Liver function tests may show mildly elevated alkaline phosphatase.15
- Muscle enzyme levels, including creatinine kinase, are normal in patients with PMR.16
- Positive antinuclear antibodies, antineutrophil cytoplasmic antibodies, rheumatoid factor, and anti-citrullinated peptide antibodies are not found in patients with PMR and indicate an alternative diagnosis if present.7
Imaging
- Ultrasonography of the shoulder shows subdeltoid bursitis in up to 79% of patients with PMR and may be considered as an adjunct diagnostic study.17,18
- If clinically indicated, chest radiography may be considered because lung cancer paraneoplastic syndrome can mimic PMR.14 An exhaustive evaluation for underlying occult malignancy or infection is not recommended.4
- Plain radiography of the involved joints may show concomitant degenerative joint disease but does not add diagnostic value.7
TREATMENT
TABLE 3. Treatment of Polymyalgia Rheumatica and Giant Cell Arteritis
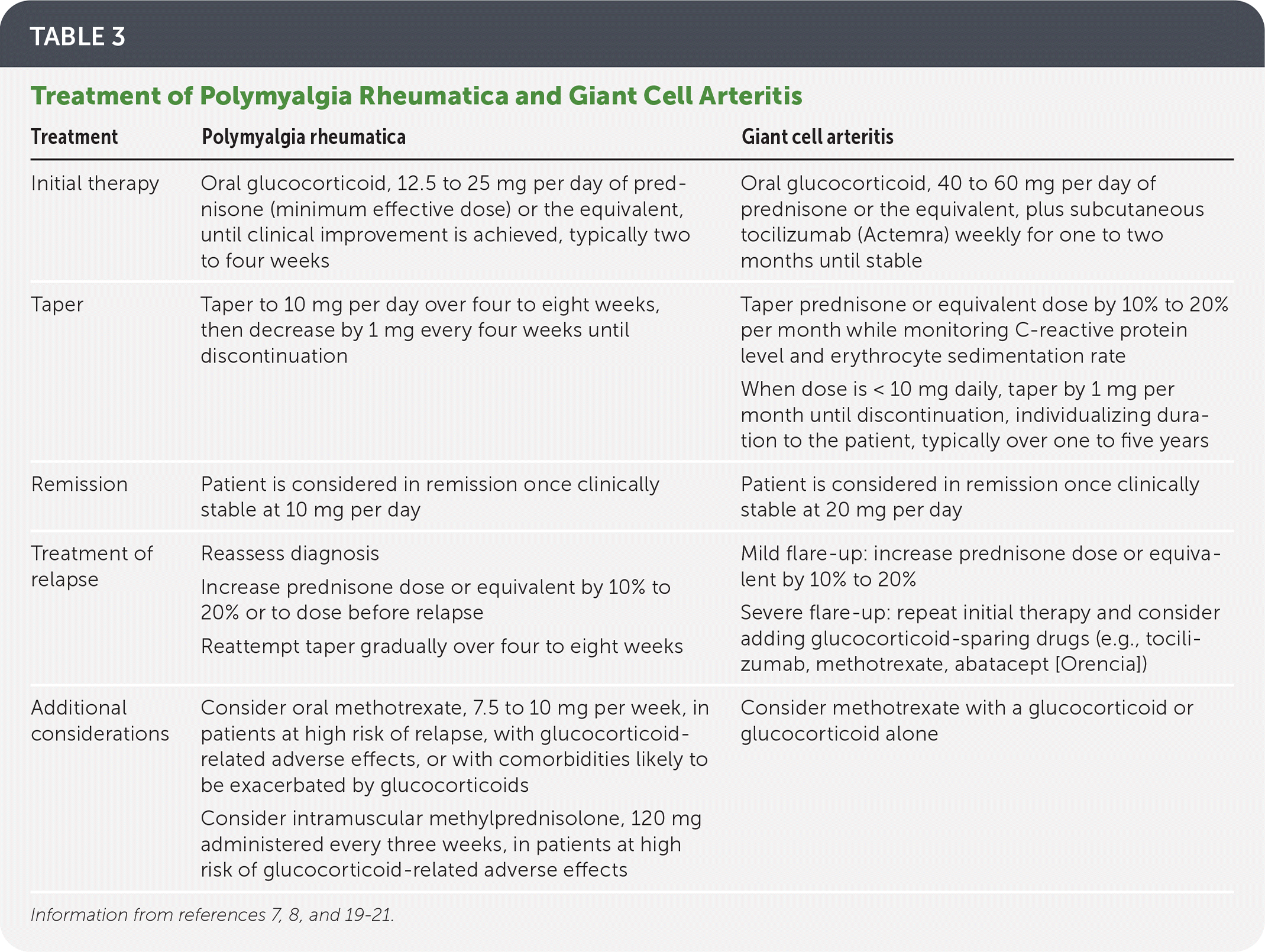
| Treatment | Polymyalgia rheumatica | Giant cell arteritis |
|---|---|---|
| Initial therapy | Oral glucocorticoid, 12.5 to 25 mg per day of prednisone (minimum effective dose) or the equivalent, until clinical improvement is achieved, typically two to four weeks | Oral glucocorticoid, 40 to 60 mg per day of prednisone or the equivalent, plus subcutaneous tocilizumab (Actemra) weekly for one to two months until stable |
| Taper | Taper to 10 mg per day over four to eight weeks, then decrease by 1 mg every four weeks until discontinuation | Taper prednisone or equivalent dose by 10% to 20% per month while monitoring C-reactive protein level and erythrocyte sedimentation rate When dose is < 10 mg daily, taper by 1 mg per month until discontinuation, individualizing duration to the patient, typically over one to five years |
| Remission | Patient is considered in remission once clinically stable at 10 mg per day | Patient is considered in remission once clinically stable at 20 mg per day |
| Treatment of relapse | Reassess diagnosis Increase prednisone dose or equivalent by 10% to 20% or to dose before relapse Reattempt taper gradually over four to eight weeks | Mild flare-up: increase prednisone dose or equivalent by 10% to 20% Severe flare-up: repeat initial therapy and consider adding glucocorticoid-sparing drugs (e.g., tocilizumab, methotrexate, abatacept [Orencia]) |
| Additional considerations | Consider oral methotrexate, 7.5 to 10 mg per week, in patients at high risk of relapse, with glucocorticoid-related adverse effects, or with comorbidities likely to be exacerbated by glucocorticoids Consider intramuscular methylprednisolone, 120 mg administered every three weeks, in patients at high risk of glucocorticoid-related adverse effects | Consider methotrexate with a glucocorticoid or glucocorticoid alone |
Medical Management
- The consensus primary treatment of PMR is oral glucocorticoid therapy, starting at 12.5 to 25 mg per day of prednisone (the minimum effective dose) or the equivalent. Once clinical improvement is achieved, typically in two to four weeks, an initial taper to 10 mg per day over four to eight weeks is recommended. Remission is likely if the patient remains clinically stable at 10 mg per day, and glucocorticoid dose may be slowly tapered by 1 mg every four weeks until discontinuation.7,8,14,19,22
- If there is a relapse or flare-up, the glucocorticoid dose should be increased by 10% to 20% or to the dose before relapse. Once the patient is stable on the new dose, the glucocorticoid dose should be tapered over four to eight weeks to where it was when the relapse occurred.8
- The addition of methotrexate may be warranted in patients at high risk of relapse (e.g., females, those with high baseline ESR), glucocorticoid-related adverse effects, or comorbidities that are likely to be exacerbated by glucocorticoids (e.g., diabetes mellitus, osteoporosis).7,8,19 Multiple high-quality randomized controlled trials have shown that the early addition of methotrexate therapy is associated with lower relapse rates and lower cumulative glucocorticoid dose. The dosage of methotrexate used in these trials ranged from 7.5 to 10 mg per week.23–25
- One small but high-quality randomized controlled trial of intramuscular methylprednisolone, 120 mg administered every three weeks, demonstrated comparable remission rates but lower cumulative glucocorticoid dose and less weight gain compared with standard oral therapy.26 Therefore, intramuscular methylprednisolone may be considered as an alternative to oral prednisone equivalents in patients at high risk of glucocorticoid-related adverse effects.8,19
- Infliximab (Remicade) and etanercept (Enbrel) have not shown benefit in the treatment of PMR.27,28
Follow-up and Monitoring
- Patients should be assessed for response to therapy, glucocorticoid-related adverse effects, relapse, and development of GCA.14
- Dose-tapering schedules for patients with PMR should be individualized based on monitoring of disease activity, laboratory markers, and adverse events.19
- An alternative diagnosis should be considered in patients who do not respond to glucocorticoid therapy or who have recurrent relapses.19
- Assessment of bone mineral density with dual-energy x-ray absorptiometry and treatment of osteoporosis should be considered in patients at high risk of fracture (e.g., those 65 years and older or with a history of fracture) while undergoing glucocorticoid therapy.14,29
- Rheumatology consultation should be considered for patients who respond poorly to initial treatment, are at high risk of relapse or treatment-induced adverse effects, or have contraindications to glucocorticoid therapy.19
Lifestyle and Alternative Therapies
- An individualized exercise program to maintain muscle mass and prevent falls, especially in older or frail patients with PMR, is recommended based on expert opinion.7,19
- The use of herbal preparations for the treatment of PMR is not recommended given a paucity of supporting evidence and unclear adverse effect profiles.19
PROGNOSIS
- Mortality in patients with PMR is comparable to that in an age-matched control group.2
- Given the nonerosive nature of PMR-associated synovitis, permanent tissue damage to the involved joints is rare.30
- Relapse may occur in up to 50% of patients.31 Risk factors for relapse and the development of concomitant GCA include female sex, high baseline ESR, initial glucocorticoid dosing of less than 10 mg per day or more than 20 mg per day, and a faster than recommended glucocorticoid taper.22,32,33
Giant Cell Arteritis
DIAGNOSIS
History and Physical Examination
- Diagnosis of GCA relies on a thorough history focused on vascular, ocular, musculoskeletal, and neurologic examinations.
- Classic clinical findings of GCA include new-onset headache, visual disturbances or changes, constitutional symptoms (e.g., weight loss, fatigue, anorexia), scalp tenderness, and temporal artery symptoms (i.e., enlargement, loss of pulses, nodularity, or tenderness).8,11,20
- Additional clinical findings include limb claudication, stroke or transient ischemic attack, dysphagia, peripheral pulse disturbances, or aortic aneurysm (Table 4).8,11,20
TABLE 4. Clinical Findings in Giant Cell Arteritis
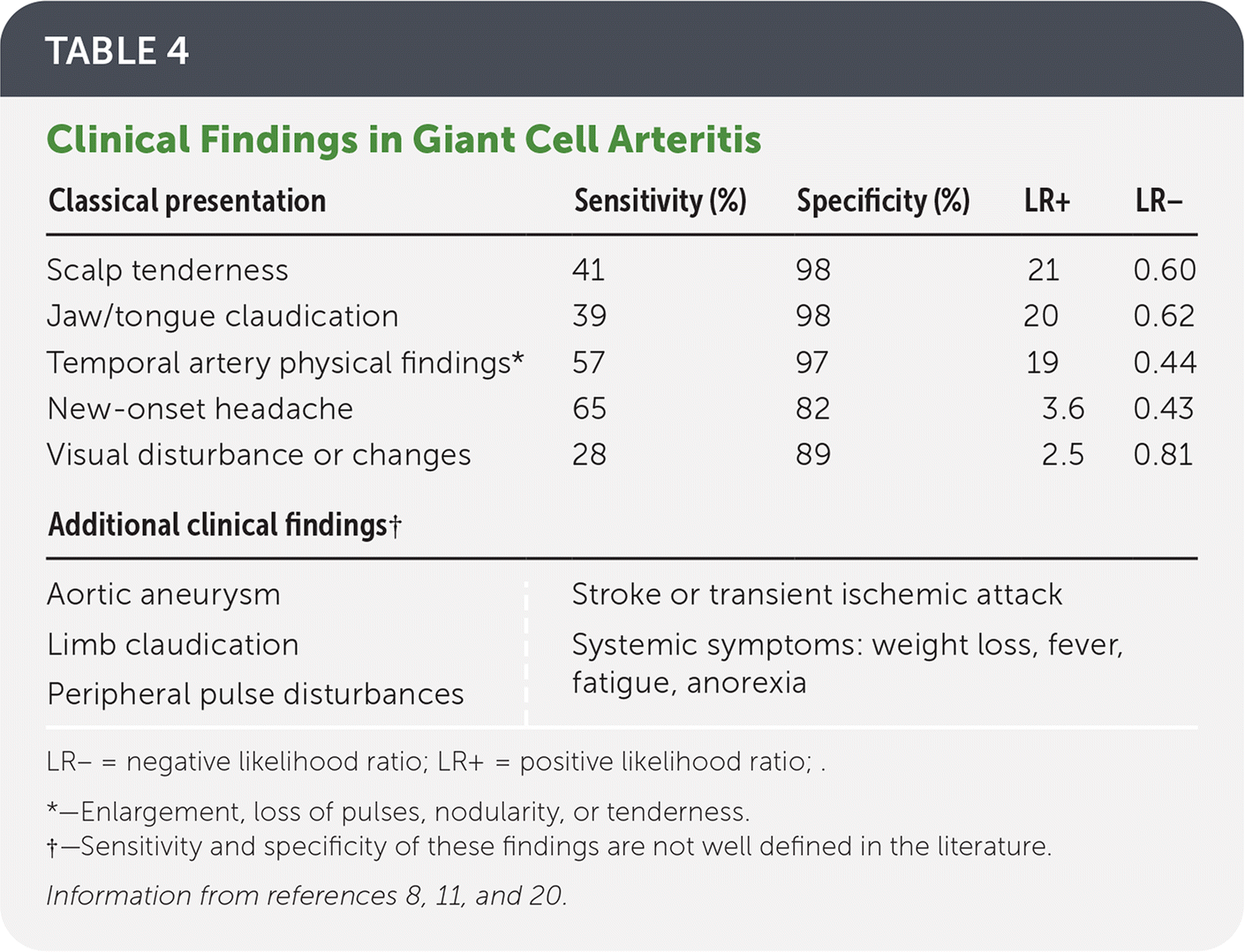
| Classical presentation | Sensitivity (%) | Specificity (%) | LR+ | LR− |
|---|---|---|---|---|
| Scalp tenderness | 41 | 98 | 21 | 0.60 |
| Jaw/tongue claudication | 39 | 98 | 20 | 0.62 |
| Temporal artery physical findings* | 57 | 97 | 19 | 0.44 |
| New-onset headache | 65 | 82 | 3.6 | 0.43 |
| Visual disturbance or changes | 28 | 89 | 2.5 | 0.81 |
| Additional clinical findings† | ||||
| Aortic aneurysm | Stroke or transient ischemic attack | |||
| Limb claudication | Systemic symptoms: weight loss, fever, fatigue, anorexia | |||
| Peripheral pulse disturbances | ||||
LR− = negative likelihood ratio; LR+ = positive likelihood ratio ; .
*—Enlargement, loss of pulses, nodularity, or tenderness.
†—Sensitivity and specificity of these findings are not well defined in the literature.
Laboratory Testing
- Temporal artery biopsy can confirm the diagnosis and should be performed in all patients with suspected GCA (Table 5).20
- ESR, CRP, complete blood count, and comprehensive metabolic panel should be obtained at presentation and while monitoring medical management.
- Marked elevation of ESR (40 to 60 mm per hour) or CRP is present in most cases. Disease severity may be reflected in the degree of ESR elevation.8
- Measurement of interleukin-6 should be considered when the patient does not have increased ESR or CRP but the index of suspicion is high. Interleukin-6 may be elevated in new and recurrent cases of GCA, returning to baseline on remission.34
TABLE 5. Recommendations for Performing a Temporal Artery Biopsy
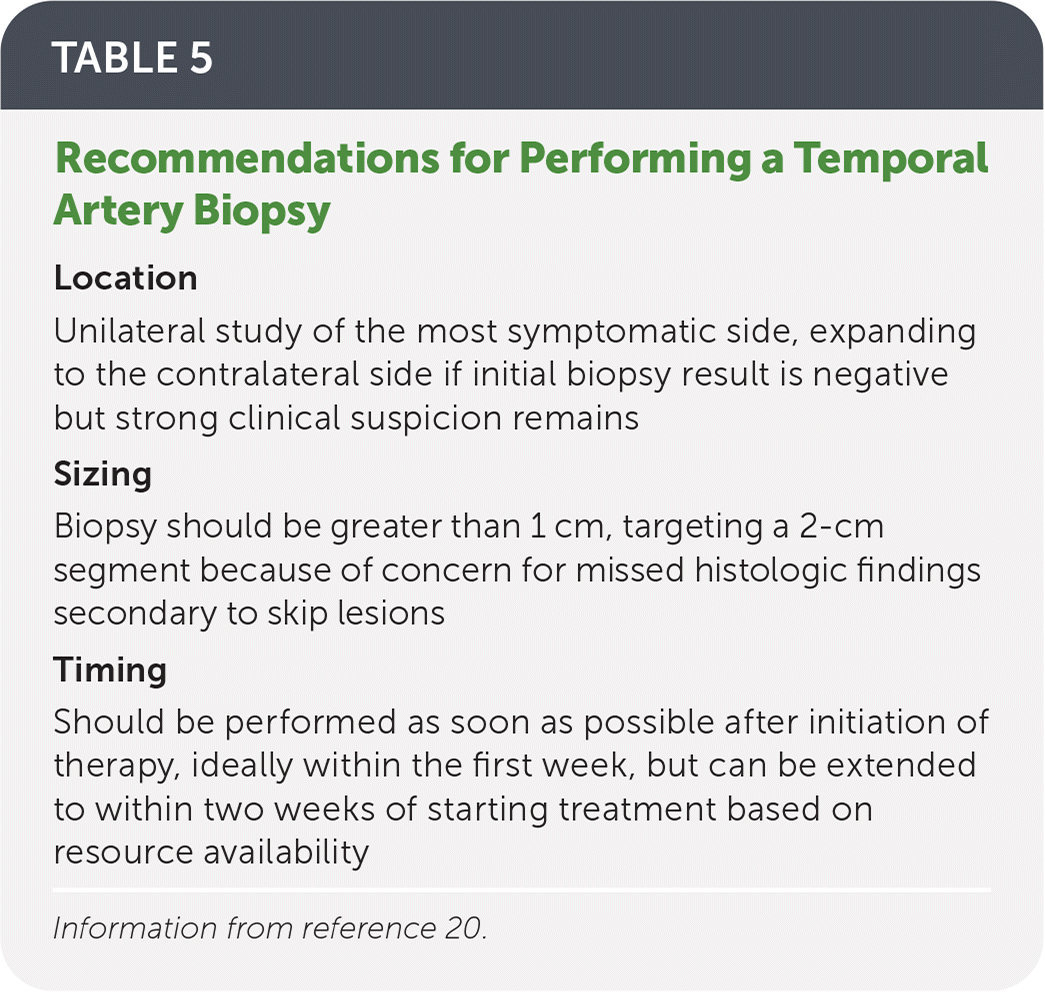
| Location |
| Unilateral study of the most symptomatic side, expanding to the contralateral side if initial biopsy result is negative but strong clinical suspicion remains |
| Sizing |
| Biopsy should be greater than 1 cm, targeting a 2-cm segment because of concern for missed histologic findings secondary to skip lesions |
| Timing |
| Should be performed as soon as possible after initiation of therapy, ideally within the first week, but can be extended to within two weeks of starting treatment based on resource availability |
Information from reference 20.
Imaging Studies
- Imaging studies may be indicated if there is strong clinical concern for GCA but temporal artery biopsy results are negative. Characteristic findings on color duplex ultrasonography, magnetic resonance imaging, or fluorodeoxyglucose positron emission tomography can support the diagnosis. Availability and technician experience may limit these modalities.8,20,35,36
- In patients with newly diagnosed GCA, noninvasive vascular imaging (i.e., magnetic resonance imaging or computed tomography angiography) of the neck, chest, abdomen, and pelvis should be performed to evaluate for large vessel involvement. Patients with hypertension at the time of GCA diagnosis are at higher risk of thoracic or aortic aneurysm and should be evaluated for large vessel involvement with angiography or ultrasonography.37
TREATMENT
- Table 3 includes consensus treatment recommendations for GCA.7,8,19–21
- Suspected GCA should be treated as a medical emergency, and initiation of treatment should not be delayed for diagnostic studies.20,21
- Treatment should be followed by urgent referral to a rheumatologist.12,20
- High-dose oral glucocorticoids are the primary treatment for GCA without cranial ischemia, starting at 40 to 60 mg per day of prednisone or equivalent for one to two months, followed by individualized tapering regimens over one to five years.8,20,21
- Two high-quality randomized controlled trials showed that adding weekly or every-other-week administration of subcutaneous tocilizumab (Actemra) to a 26-week prednisone taper was superior to prednisone taper plus placebo for achievement of sustained GCA remission.38,39
- High-dose intravenous glucocorticoids should be initiated in patients with symptoms of critical cranial ischemia, including visual disturbances.20
- Patients undergoing high-dose glucocorticoid therapy require bone-conserving and gastroprotective therapy, including calcium and vitamin D supplementation and consideration of bisphosphonates and proton pump inhibitors.29
- Glucocorticoid-sparing therapies such as tocilizumab, methotrexate, abatacept (Orencia), and other biologics can be considered in all patients based on symptom severity, patient goals and preferences, and physician experience.20
PROGNOSIS
- Early recognition and treatment are associated with decreased morbidity.8,20
- Although GCA does not affect overall survival, patients with the condition are more likely to die of circulatory diseases than their peers because of the risk of progressive vasculitis involving large vessels elsewhere in the body.12
- Morbidity is usually related to adverse effects of glucocorticoids.21
- Long-term clinical and laboratory monitoring is associated with decreased frequency and severity of recurrence.12,20
This article updates previous articles on this topic by Meskimen, et al.40; Epperly, et al.41; Unwin, et al.42; and Caylor and Perkins.9
Data Sources: This article was based on literature cited in Essential Evidence Plus, the Cochrane database, and a PubMed search using the Clinical Queries database for the terms polymyalgia rheumatica, giant cell arteritis, and temporal arteritis. Search dates: September and November 2021 and July 2022.
The views expressed in this material are those of the authors and do not reflect the official policy or position of the U.S. government, the U.S. Department of Defense, the U.S. Department of the Army, or the Uniformed Services University of the Health Sciences.

