Individuals with skin of color represent a diverse population of racial and ethnic backgrounds, including but not limited to Black or African American, American Indian or Alaska Native, Asian American or Pacific Islander, Hispanic or Latino, and Middle Eastern or North African. Dermatologic health disparities exist in part because of systemic racism and are exacerbated by inadequate physician training and a lack of high-quality research on skin diagnoses that disproportionately affect people with skin of color. These conditions, which include postinflammatory hyperpigmentation, keloids, dermatosis papulosa nigra, pseudofolliculitis barbae, and acne keloidalis nuchae, are usually diagnosed clinically and not associated with an underlying systemic disease. They can have significant impacts on mental health and quality of life and are often underdiagnosed or undertreated in skin of color. Hydroquinone 4% is considered the standard treatment for postinflammatory hyperpigmentation. Standard treatment for keloids includes combination intralesional therapy with triamcinolone and fluorouracil. If treatment is preferred for dermatosis papulosa nigra, options include scissor excision, cryotherapy, curettage, electrodesiccation, and laser therapies. Shaving cessation is the best initial treatment for pseudofolliculitis barbae. Individuals with acne keloidalis nuchae should avoid frequent close shaves or short haircuts on the nuchal area of the scalp.
Individuals with skin of color make up 40.9% of the U.S. population, and this is expected to increase to 59.5% by 2060.1 Skin conditions account for 12.4% of all diagnoses seen by family physicians,2–12 highlighting the importance of educating physicians on common dermatologic conditions in skin of color (Table 13–10,13,14 ). Although other skin conditions such as atopic dermatitis, psoriasis, and cellulitis can present differently in skin of color, this article focuses on five common diagnoses that disproportionately affect this population and can have a substantial impact on mental health and quality of life: postinflammatory hyperpigmentation, keloids, dermatosis papulosa nigra, pseudofolliculitis barbae, and acne keloidalis nuchae.
WHAT'S NEW ON THIS TOPIC

| Despite increased reporting of race and ethnicity in dermatology clinical trials, the percentage of non-White participants has not changed over the past 10 years. |
| An analysis of dermatologic textbooks showed that Fitzpatrick skin types V and VI are dramatically underrepresented compared with U.S. demographics. The one exception is sexually transmitted infections; skin of color represents 47% to 58% of images depicting these infections. |
| A systematic review of 36 articles evaluating cutaneous manifestations of COVID-19 showed that 120 out of 130 images (92%) represented patients with Fitzpatrick skin types I to III, with no images representing skin types V or VI. |
SORT: KEY RECOMMENDATIONS FOR PRACTICE
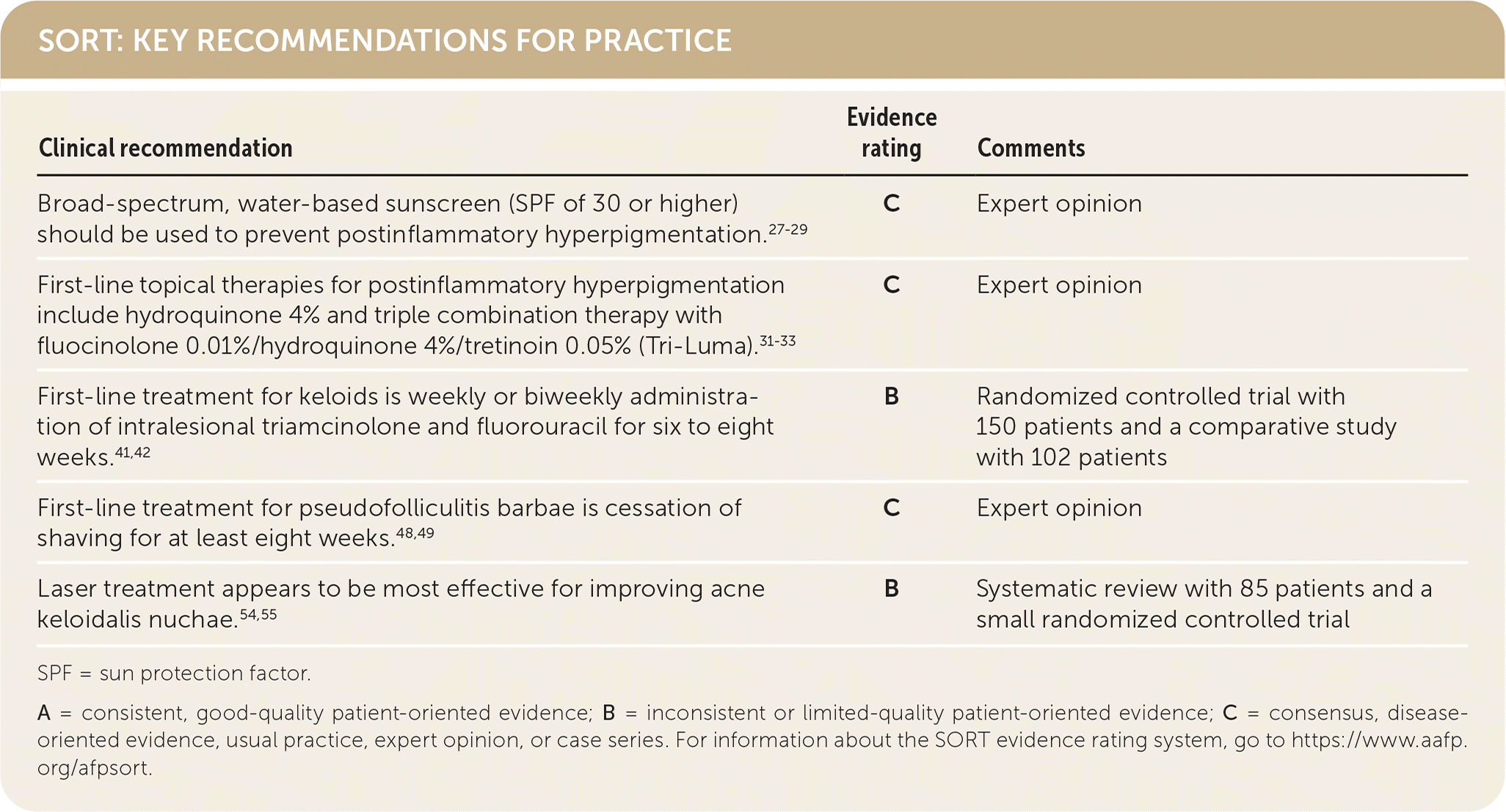
| Clinical recommendation | Evidence rating | Comments |
|---|---|---|
| Broad-spectrum, water-based sunscreen (SPF of 30 or higher) should be used to prevent postinflammatory hyperpigmentation.27–29 | C | Expert opinion |
| First-line topical therapies for postinflammatory hyperpigmentation include hydroquinone 4% and triple combination therapy with fluocinolone 0.01%/hydroquinone 4%/tretinoin 0.05% (Tri-Luma).31–33 | C | Expert opinion |
| First-line treatment for keloids is weekly or biweekly administration of intralesional triamcinolone and fluorouracil for six to eight weeks.41,42 | B | Randomized controlled trial with 150 patients and a comparative study with 102 patients |
| First-line treatment for pseudofolliculitis barbae is cessation of shaving for at least eight weeks.48,49 | C | Expert opinion |
| Laser treatment appears to be most effective for improving acne keloidalis nuchae.54,55 | B | Systematic review with 85 patients and a small randomized controlled trial |
SPF = sun protection factor.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
TABLE 1. Common Dermatologic Conditions in Skin of Color
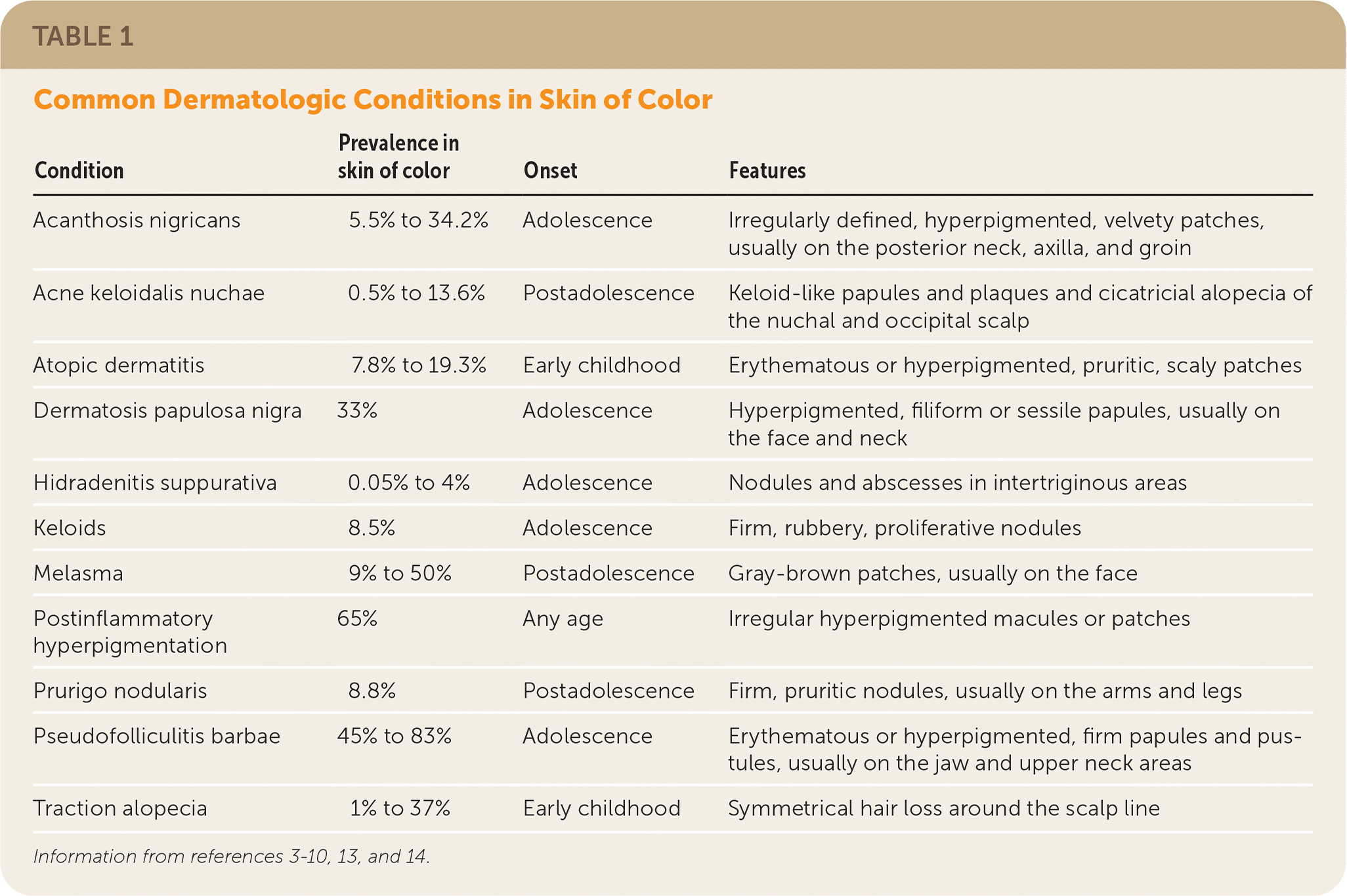
| Condition | Prevalence in skin of color | Onset | Features |
|---|---|---|---|
| Acanthosis nigricans | 5.5% to 34.2% | Adolescence | Irregularly defined, hyperpigmented, velvety patches, usually on the posterior neck, axilla, and groin |
| Acne keloidalis nuchae | 0.5% to 13.6% | Postadolescence | Keloid-like papules and plaques and cicatricial alopecia of the nuchal and occipital scalp |
| Atopic dermatitis | 7.8% to 19.3% | Early childhood | Erythematous or hyperpigmented, pruritic, scaly patches |
| Dermatosis papulosa nigra | 33% | Adolescence | Hyperpigmented, filiform or sessile papules, usually on the face and neck |
| Hidradenitis suppurativa | 0.05% to 4% | Adolescence | Nodules and abscesses in intertriginous areas |
| Keloids | 8.5% | Adolescence | Firm, rubbery, proliferative nodules |
| Melasma | 9% to 50% | Postadolescence | Gray-brown patches, usually on the face |
| Postinflammatory hyperpigmentation | 65% | Any age | Irregular hyperpigmented macules or patches |
| Prurigo nodularis | 8.8% | Postadolescence | Firm, pruritic nodules, usually on the arms and legs |
| Pseudofolliculitis barbae | 45% to 83% | Adolescence | Erythematous or hyperpigmented, firm papules and pustules, usually on the jaw and upper neck areas |
| Traction alopecia | 1% to 37% | Early childhood | Symmetrical hair loss around the scalp line |
The wide spectrum of skin color is commonly classified using Fitzpatrick skin phototypes I to VI (Table 2).2,3,13–18 This classification was first proposed in 1975 and relies on the subjective determination of an individual's propensity for photodermatitis (sunburn) based on skin color. Importantly, this classification system should not be used as a surrogate marker for race and ethnicity.15,16 In this article, skin of color refers to a diverse population of racial and ethnic backgrounds, including but not limited to those who identify as Black or African American, American Indian or Alaska Native, Asian American, Pacific Islander, Hispanic or Latino, and Middle Eastern or North African.
TABLE 2. Fitzpatrick Skin Phototypes

| Type | Example | Possible skin conditions |
|---|---|---|
| I | Melanoma | |
| II | Melanoma | |
| III | Dermatosis papulosa nigra Postinflammatory hyperpigmentation | |
| IV | Dermatosis papulosa nigra Postinflammatory hyperpigmentation | |
| V | Acne keloidalis nuchae Dermatosis papulosa nigra Keloids Postinflammatory hyperpigmentation Pseudofolliculitis barbae | |
| VI | Acne keloidalis nuchae Dermatosis papulosa nigra Keloids Postinflammatory hyperpigmentation Pseudofolliculitis barbae |
Dermatologic Health Disparities
Factors that contribute to dermatologic disparities include systemic racism, lack of high-quality evidence-based research on dermatologic conditions affecting skin of color, and lack of physician education on the treatment and diagnosis of these conditions.19 For example, melanoma and nonmelanoma skin cancers are less prevalent in patients with skin of color, but these patients clinically present with more advanced disease (16% vs. 5%) and have a lower five-year survival rate (66.2% vs. 90.1%) compared with White patients.20 Black patients are less likely to receive treatment for acne, atopic dermatitis, and psoriasis compared with White patients.21
Despite increased reporting of race and ethnicity in dermatology clinical trials, the percentage of non-White participants has not changed over the past 10 years.22 A 2020 systematic review evaluating cutaneous manifestations of COVID-19 showed that 120 out of 130 images (92%) from 36 articles represented patients with Fitzpatrick skin types I to III, with no images representing types V or VI.23 An analysis of dermatologic textbooks showed that skin types V and VI are dramatically underrepresented compared with U.S. demographics (with less than 14% of textbook images representing skin types V and VI).24 The one exception is sexually transmitted infections; skin of color represents 47% to 58% of images depicting these infections, compared with 28% of non–sexually transmitted infections.25
A 2011 survey showed that 47% of dermatologists report inadequate training on skin conditions common in Black patients.26 Medical students receive an average of only 16 to 22 total hours of dermatology training, and fewer than 40% of primary care residents feel that their medical school adequately prepared them to manage common skin conditions.26
Expanding dermatologic medical education and enhancing research funding directly related to common conditions in skin of color are key to reducing dermatologic health disparities.
Postinflammatory Hyperpigmentation
Postinflammatory hyperpigmentation is a reactive hypermelanosis that occurs after endogenous inflammation or external injury (Figure 1) and is most noticeable in Fitzpatrick skin types III to VI (90% of cases).14 The first step in treatment is identifying the underlying etiology of the injury or inflammation to prevent further damage.14 Endogenous causes include inflammatory conditions such as acne vulgaris (47.4% to 65.3% of cases), pseudofolliculitis barbae, atopic dermatitis, lichen planus, psoriasis, and contact dermatitis. Causative external injuries include insect bites, chemical peels, cryotherapy, and laser surgery. Use of a broad-spectrum (ultraviolet A and B protection), water-based sunscreen with a sun protection factor (SPF) of 30 or higher can reduce the incidence of postinflammatory hyperpigmentation.27–29 Sunscreen that blocks visible light, such as iron oxide sunscreen, can be especially helpful for patients with skin types III to VI.27,30
FIGURE 1.
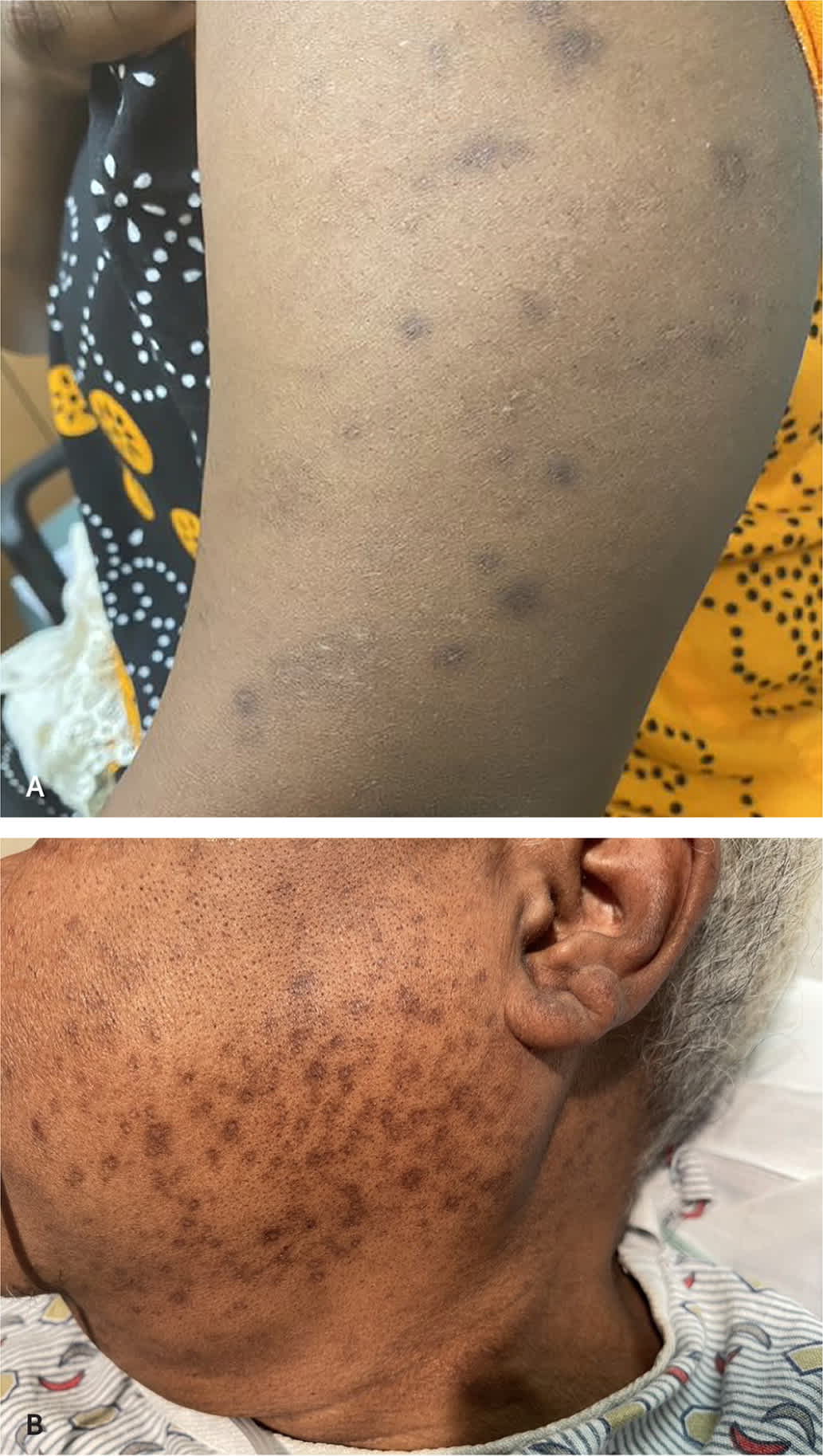
Postinflammatory hyperpigmentation caused by (A) insect bites and (B) acne.
The topical lightening agent hydroquinone 4% applied once or twice daily for three months is the standard treatment for postinflammatory hyperpigmentation31,32 (Table 3). Triple combination therapy with fluocinolone 0.01%/hydroquinone 4%/tretinoin 0.05% (Tri-Luma) is more effective than hydroquinone alone.32,33 Nightly use of tretinoin 0.1% (Retin-A) or third-generation retinoids (adapalene and tazarotene) is effective, particularly for acne-associated postinflammatory hyperpigmentation.32,34,35 Tazarotene 0.1% cream (Tazorac) has been shown to be more effective than adapalene 0.3% gel (Differin).35 Twice daily application of azelaic acid 15% gel (Finacea) over 16 weeks can significantly improve postinflammatory hyperpigmentation in patients with skin types IV to VI.36 Thiamidol (a tyrosinase inhibitor that prevents the first step in melanin production) showed a 40% reduction in postinflammatory hyperpigmentation compared with placebo.37 Common adverse effects of these topical treatments include desquamation (27.2%), burning/stinging (22.7%), dryness (15.7%), and pruritus (15.2%).14
TABLE 3. Common Topical Medications for Postinflammatory Hyperpigmentation and Pseudofolliculitis Barbae
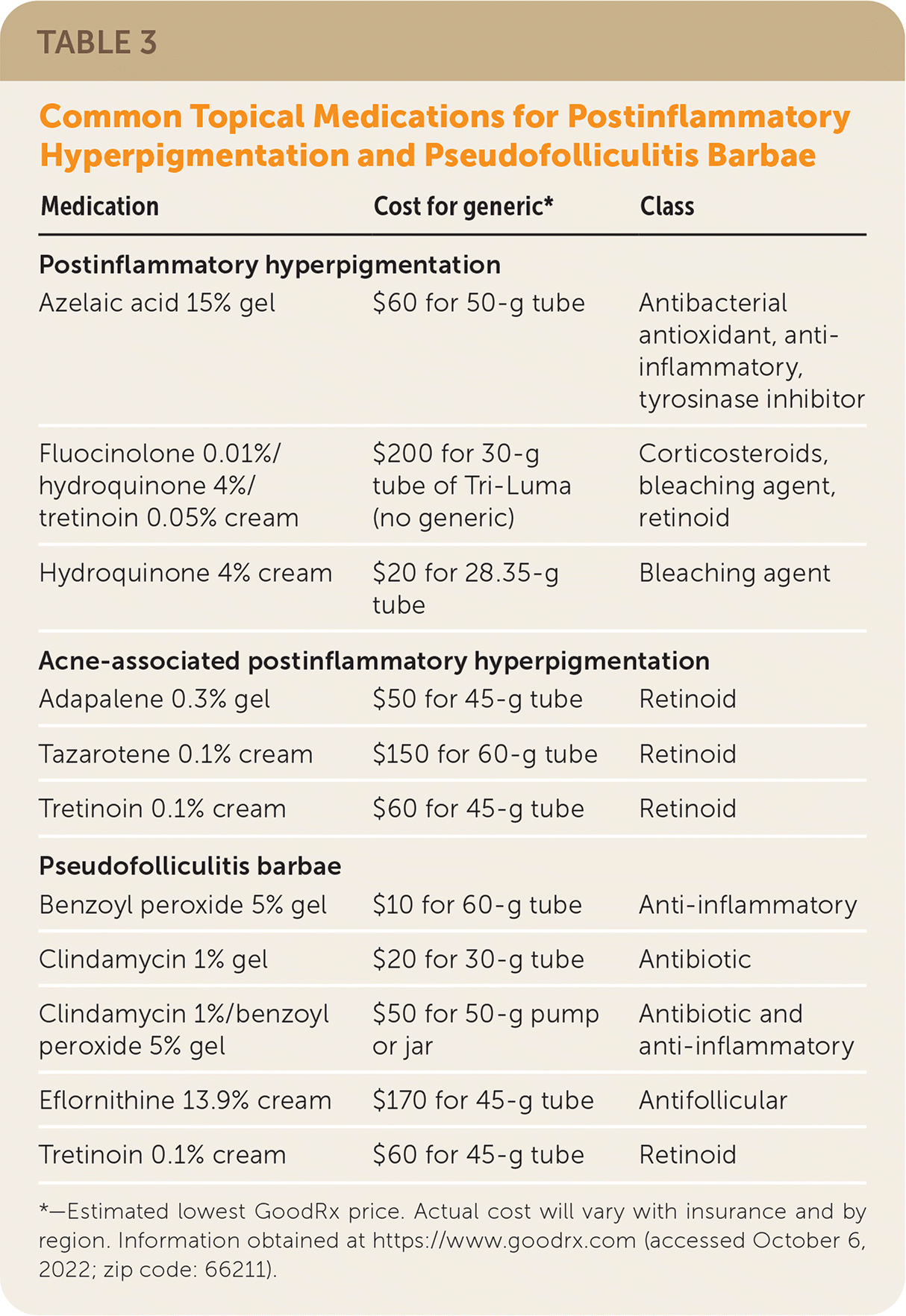
| Medication | Cost for generic* | Class |
|---|---|---|
| Postinflammatory hyperpigmentation | ||
| Azelaic acid 15% gel | $60 for 50-g tube | Antibacterial antioxidant, anti-inflammatory, tyrosinase inhibitor |
| Fluocinolone 0.01%/hydroquinone 4%/tretinoin 0.05% cream | $200 for 30-g tube of Tri-Luma (no generic) | Corticosteroids, bleaching agent, retinoid |
| Hydroquinone 4% cream | $20 for 28.35-g tube | Bleaching agent |
| Acne-associated postinflammatory hyperpigmentation | ||
| Adapalene 0.3% gel | $50 for 45-g tube | Retinoid |
| Tazarotene 0.1% cream | $150 for 60-g tube | Retinoid |
| Tretinoin 0.1% cream | $60 for 45-g tube | Retinoid |
| Pseudofolliculitis barbae | ||
| Benzoyl peroxide 5% gel | $10 for 60-g tube | Anti-inflammatory |
| Clindamycin 1% gel | $20 for 30-g tube | Antibiotic |
| Clindamycin 1%/benzoyl peroxide 5% gel | $50 for 50-g pump or jar | Antibiotic and anti-inflammatory |
| Eflornithine 13.9% cream | $170 for 45-g tube | Antifollicular |
| Tretinoin 0.1% cream | $60 for 45-g tube | Retinoid |
*—Estimated lowest GoodRx price. Actual cost will vary with insurance and by region. Information obtained at https://www.goodrx.com (accessed October 6, 2022; zip code: 66211).
Laser therapy can treat postinflammatory hyperpigmentation but must be used conservatively because skin of color is at high risk of dyspigmentation and scarring. Testing laser therapy on a small area before full application is preferred. Patients should be counseled to avoid excessive sun exposure and use broad-spectrum sunscreen before and after laser treatments.28
Keloids
Keloids are firm, rubbery, proliferative nodules that appear one to 12 months after a cutaneous injury (Figure 2). Keloids are diagnosed by clinical features and typically develop between 10 and 30 years of age.38 Keloids are distinct from hypertrophic scars because they extend beyond the boundary of the original injury and do not regress spontaneously. Keloids are caused by fibroproliferative overgrowth during wound healing, but the exact mechanism is unknown. They are more common in skin types III to VI (8.5% of cases) than types I and II (0.1% to 0.5% of cases).39 One-third of people with keloids have a first-degree relative with the condition.39 Keloids are more likely to develop in areas with high dermal tension (e.g., major joints, anterior chest, upper back, lower abdomen), earlobes, and cheeks.38
FIGURE 2.
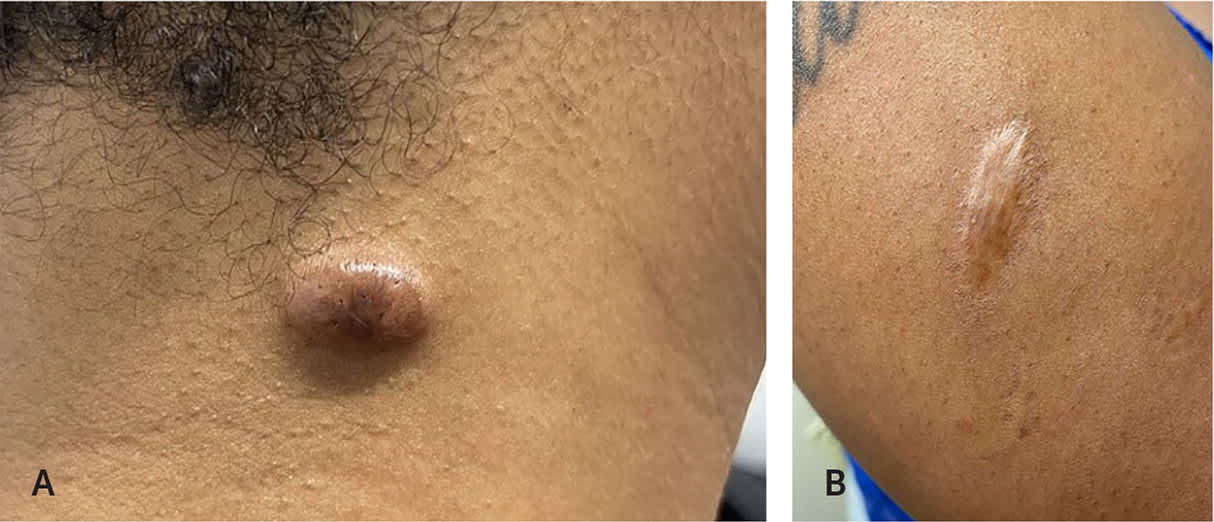
(A) Keloids are distinct from (B) hypertrophic scars because they extend beyond the boundary of the original wound.
Preoperative counseling should include patient education on risk factors for keloids and mitigation strategies such as compression therapy.40 Standard treatment for keloids includes combination intralesional therapy with triamcinolone and fluorouracil applied weekly or biweekly for six to eight weeks.41,42 A meta-analysis showed that triamcinolone plus fluorouracil leads to greater reduction in keloid scar height with fewer complications and lower rates of recurrence than triamcinolone monotherapy.43 Risks of intralesional steroids include atrophy, dyspigmentation, telangiectasias, and pain.38
Surgical excision is reserved for refractory cases because of the high risk of keloid recurrence (50% to 100%).44 Interventions that could decrease keloid formation in high-risk patients include postsurgical intralesional triamcinolone, postexcisional imiquimod 5% cream, and radiotherapy.45,46 Options with emerging evidence include bleomycin, intralesional verapamil, cryotherapy, electrical stimulation, onabotulinumtoxinA (Botox), and laser therapy.38
Dermatosis Papulosa Nigra
Dermatosis papulosa nigra presents as hyperpigmented filiform or sessile papules, ranging from 1 to 5 mm in diameter and 1 to 3 mm in elevation (Figure 3). The papules appear in a symmetrical pattern on the malar face (86%) and neck (58%).47 The lesions typically increase in number and size with age and can cause pruritus and pain. The etiology is multifactorial, including family history and cumulative ultraviolet exposure. If treatment is preferred, options include scissor excision, cryotherapy, curettage, electrodesiccation, and laser therapies.3 Pedunculated lesions can be excised using eye suture scissors. In a systematic review of 15 studies, laser therapy achieved higher rates of dermatosis papulosa nigra clearance and lower rates of postinflammatory hyperpigmentation than electrodesiccation and curettage.5
FIGURE 3.
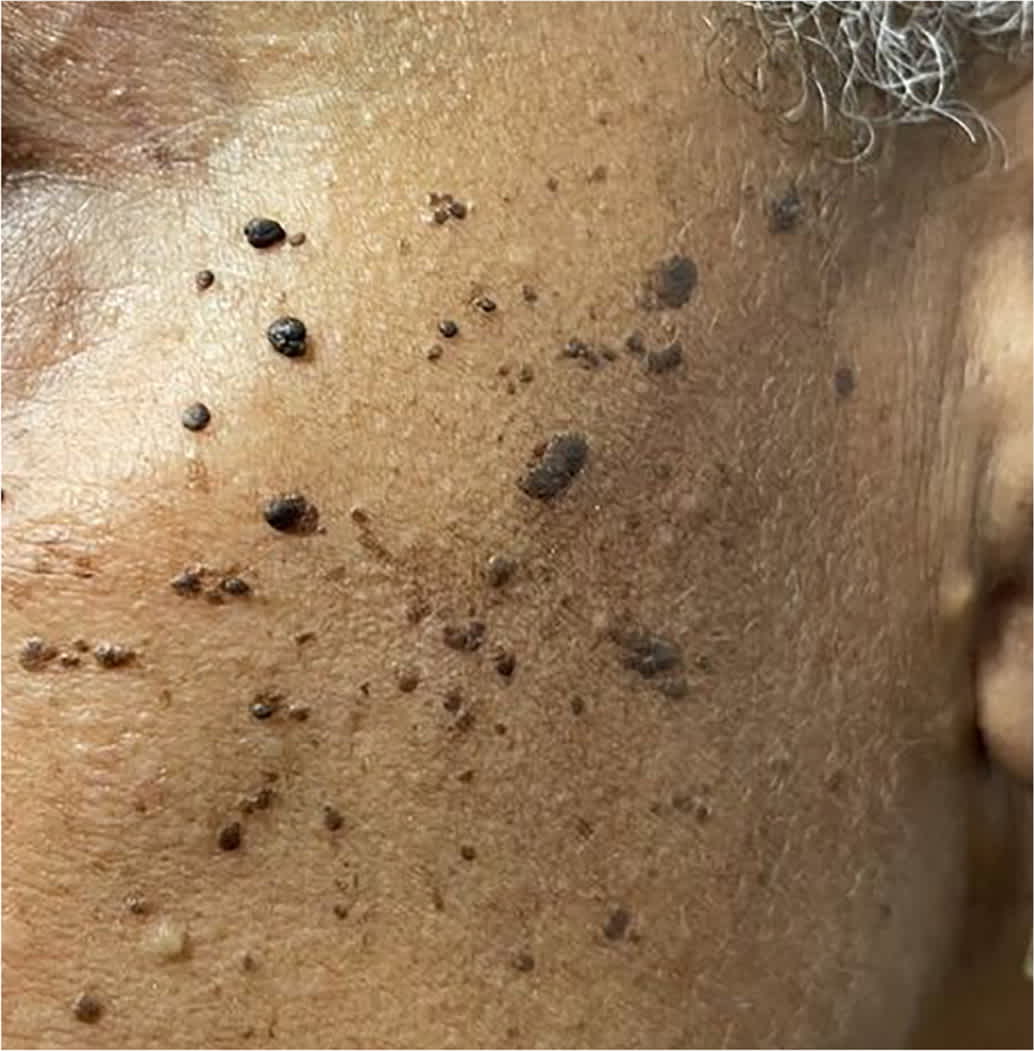
Dermatosis papulosa nigra on the malar face.
Pseudofolliculitis Barbae
Pseudofolliculitis barbae presents as erythematous or hyperpigmented, firm papules and pustules in a beard distribution (Figure 4). It is caused by follicular penetration of recently shaved curly hair. Close shaving can result in extrafollicular or transfollicular penetration within 24 to 48 hours, leading to a foreign body inflammatory response.3 The injured follicles are susceptible to infection (folliculitis barbae and sycosis barbae). The differential diagnosis of pseudofolliculitis barbae includes acne vulgaris, traumatic folliculitis, impetigo, and tinea barbae. The diagnosis is clinical; however, if unclear, it can be confirmed with visualization of follicular penetration on dermoscopy.
FIGURE 4.
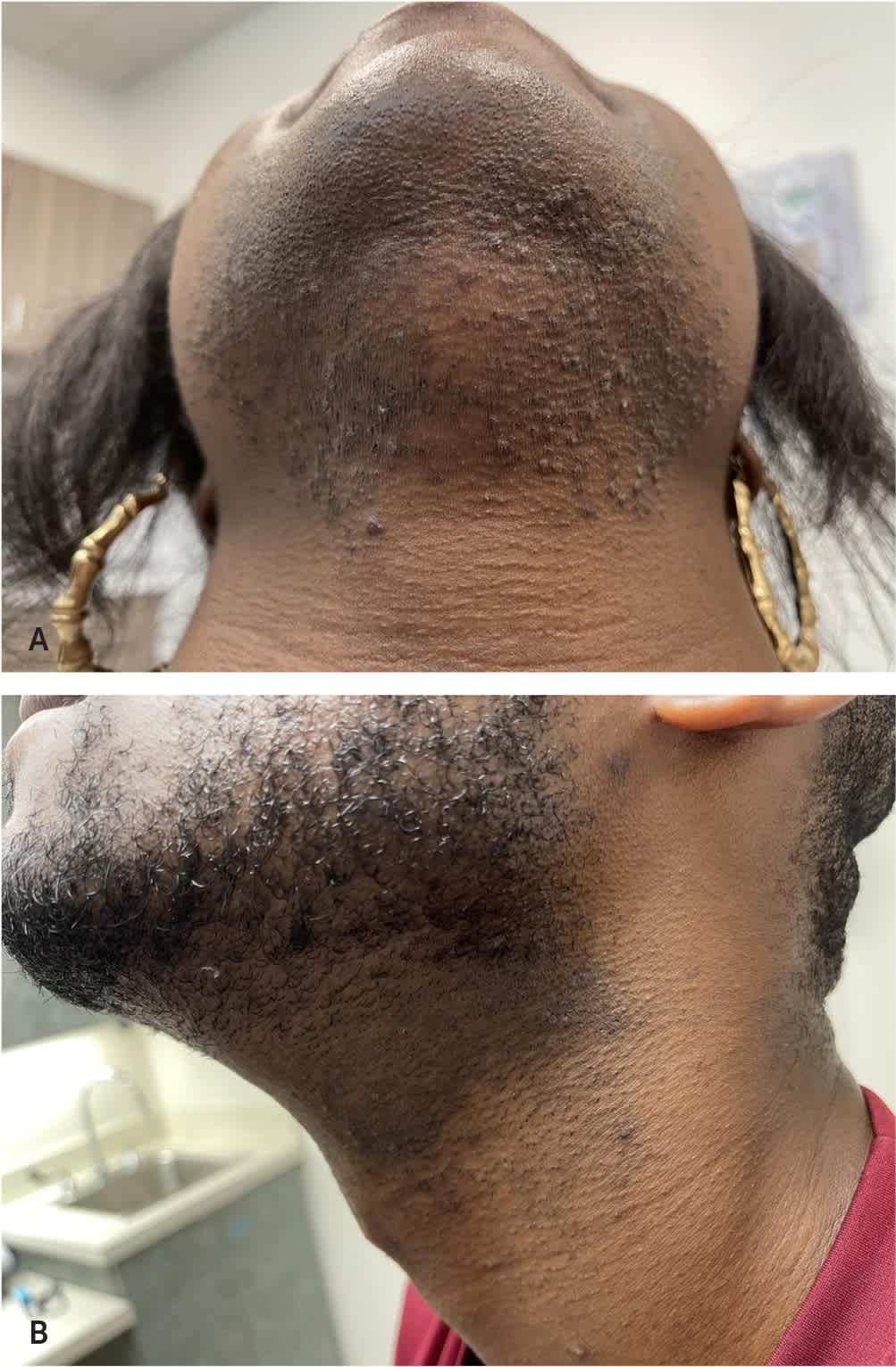
Pseudofolliculitis barbae (A) in a woman with hirsutism and (B) with associated postinflammatory hyperpigmentation.
Shaving cessation for at least eight weeks after diagnosis is the best initial treatment, and education on shaving best practices (Table 4) is important to prevent future inflammation.48,49 Studies evaluating shaving practices in predominantly self-identified African Americans found that shaving frequency, number of razor blades, and shaving products had no impact on the number of pseudofolliculitis barbae lesions.49 Depilatories (i.e., barium sulfide powder and calcium thioglycolate cream) produce fewer lesions than manual shaving but cause greater skin irritation.50
TABLE 4. Manual Shaving Best Practices to Prevent Pseudofolliculitis Barbae

| Electric razors are preferred, with settings to keep the hair at least 1 to 3 mm in length |
| Massage a warm compress in a circular motion over the beard area for five minutes to release embedded hair shafts and improve hair shaft hydration |
| Apply preshave oil and shaving cream (do not dry shave) |
| Shave with the grain, and do not pull the skin taut |
| Using short strokes, slowly and lightly shave the area in a single pass (avoid multiple passes with the razor) |
| Apply a cool compress to the area for five minutes after shaving |
| Replace razor blades after five uses (or use disposable razors once) |
| Shave every one to three days so that hair cannot grow long enough to penetrate the skin |
Recommended topical therapies, used alone or in combination, are low-potency corticosteroids, benzoyl peroxide, antibiotics, and retinoids3 (Table 3). Twice daily administration of combination clindamycin 1%/benzoyl peroxide 5% gel showed a greater reduction in pustules and papules than placebo after six weeks.51 Definitive treatment of pseudofolliculitis barbae involves permanent hair reduction primarily achieved through chemical peels or laser treatments. In a study comparing laser therapy with chemical peels (using glycolic acid 70% and trichloroacetic acid 20%), the laser therapy group had a lower number of pustules and papules and longer interval to recurrence.52 Laser hair removal combined with topical eflornithine 13.9% (Vaniqa) decreases hair loss with greater mean improvement of inflammatory papules compared with either therapy alone.48
Acne Keloidalis Nuchae
Acne keloidalis nuchae is a chronic inflammatory condition leading to scarring of the hair follicles, development of keloid-like papules and plaques, and cicatricial alopecia of the nuchal and occipital scalp 3 (Figure 5). Acne keloidalis nuchae presents after puberty with a 20:1 male to female ratio, suggesting that increasing androgen levels play a causative role along with skin injury and subsequent abnormal immune responses to the injury.2 People with acne keloidalis nuchae are more likely to also have metabolic syndrome and hidradenitis suppurativa.53
FIGURE 5.
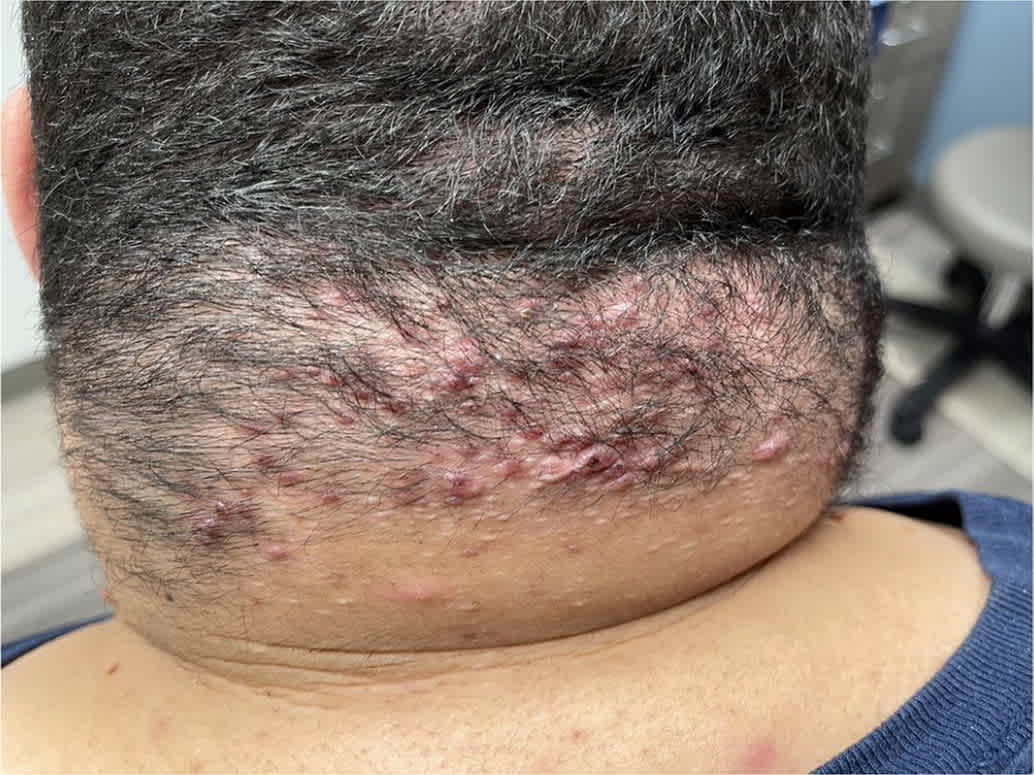
Acne keloidalis nuchae.
Individuals with acne keloidalis nuchae, particularly young men, should avoid frequent close shaves or short haircuts on the nuchal area of the scalp. Other preventive measures include minimizing irritation from closely fitting helmets, hats, or clothing such as high-collared shirts.2 Treatment options include topical agents, laser therapy, and surgical excision for refractory cases. Mild disease (papules smaller than 3 mm) can be treated with two to four weeks of triamcinolone combined with topical retinoids, or clindamycin 1% solution if pustules are present.54 Intralesional triamcinolone, 5 to 40 mg per mL, can treat mild to moderate disease (papules 3 mm or larger or plaques).54 Emerging treatment options include oral isotretinoin, cryotherapy, and oral doxycycline.54
Phototherapy and laser therapy, usually performed by dermatologists, are noninvasive and generally well-tolerated alternatives to medical management. Laser treatment appears to be most effective for improving acne keloidalis nuchae.54,55 Compared with placebo, eight monthly laser treatments with twice daily triamcinolone 0.1% cream have been shown to provide greater improvement in number and size of lesions.55 In small randomized controlled trials, phototherapy significantly decreased the mean number of lesions after 16 weeks.56 Surgical excision is reserved for extensive and refractory lesions with healing by secondary intention, although 41% of patients have mild recurrence.54
This article updates previous articles on this topic by Kundu and Patterson.3,57
Data Sources: A PubMed search was completed in Clinical Queries using the key terms keloids, acne keloidalis nuchae, dermatosis papulosa nigra, pseudofolliculitis barbae, and postinflammatory hyperpigmentation. Other key terms were skin of color, people of color, and dermatology health disparities. The search included meta-analyses, randomized controlled trials, clinical trials, and systematic reviews. Also searched were the Cochrane database, DynaMed, and Essential Evidence Plus. Search dates: July 2021 to August 2022.
The photos in Table 2 were provided by the University of Pittsburgh Medical Center St. Margaret Family Medicine Residency Program.

