Hypokalemia and hyperkalemia occur when serum potassium levels are less than 3.5 mEq per L or greater than 5.0 mEq per L, respectively. The World Health Organization recommends a potassium intake of at least 3,510 mg per day for optimal cardiovascular health. Hypokalemia is caused by decreased intake, renal losses, gastrointestinal losses, or transcellular shifts. Severe features of hypokalemia that require urgent treatment include a serum potassium level of 2.5 mEq per L or less, electrocardiography abnormalities, or neuromuscular symptoms. The underlying cause should be addressed, and potassium levels replenished. An oral route is preferred if the patient has a functioning gastrointestinal tract and a serum potassium level greater than 2.5 mEq per L. Hyperkalemia is caused by impaired renal excretion, transcellular shifts, or increased potassium intake. Electrocardiography identifies cardiac conduction disturbances but may not correlate with serum potassium levels. Emergent treatment is recommended for patients with clinical signs and symptoms (e.g., muscle weakness, paralysis) or if electrocardiography abnormalities are present. Acute treatment may include intravenous calcium, insulin, sodium bicarbonate, diuretics, and beta agonists. Dialysis may be considered in the presence of end-stage renal disease, severe renal impairment, or ongoing potassium release. Patiromer and sodium zirconium cyclosilicate are newer potassium binders and may be used in chronic or acute hyperkalemia. Sodium polystyrene sulfonate is associated with serious gastrointestinal adverse effects. Long-term management of potassium disturbances includes correcting underlying conditions, dietary counseling, and adjusting causative medications.
Homeostasis maintains a normal range of serum potassium defined as 3.5 to 5.0 mEq per L.1–3 The prevalence of hyperkalemia in the general population is 3.3%, and hypokalemia is 1.9%; however, in the emergency department, the prevalence is 3.6% and 5.5%, respectively.4,5 In patients with chronic kidney disease (CKD), the prevalence of hyperkalemia is 18%.5 The inpatient prevalence of hyperkalemia and hypokalemia ranges from 6.9% to 12.3% and 2.9% to 7.4%, respectively.6,7 Patients with an abnormal potassium level on admission (adjusted odds ratio [OR] = 1.49; 95% CI, 1.26 to 1.75) and patients with hyperkalemia (adjusted OR = 1.44; 95% CI, 1.11 to 1.87) requiring admission to a cardiac intensive care unit have an elevated risk of mortality.6,8 Serum potassium levels have a U-shaped curve association with morbidity and mortality.5,6 The best outcomes in observational studies are associated with a serum potassium level between 4 and 5 mEq per L, specifically for patients with heart failure or CKD.5,9–11 In the general population, moderate hyperkalemia (greater than 5.5 mEq per L; adjusted hazard ratio [HR] = 1.22; 95% CI, 1.15 to 1.29) and hypokalemia (less than 3.0 mEq per L; adjusted HR = 1.49; 95% CI, 1.26 to 1.76) are associated with an increased risk of all-cause mortality.5
WHAT'S NEW ON THIS TOPIC

| Patiromer (Veltassa) and sodium zirconium cyclosilicate (Lokelma) are preferred over sodium polystyrene sulfonate for the treatment of chronic hyperkalemia due to higher efficacy and lower risk of serious adverse effects. |
SORT: KEY RECOMMENDATIONS FOR PRACTICE
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease- oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Dietary Recommendations
The World Health Organization recommends a potassium intake of at least 3,510 mg per day for adults for optimal cardiovascular health.12–14 High dietary potassium intake has been found to lower blood pressure in patients with hypertension, although it may precipitate hyperkalemia if renal function is impaired.3,13–15 An increased dietary potassium intake is associated with a lower risk of stroke.12 Potassium-rich foods are common to a healthy diet (Table 1).12,16,17 A low-potassium diet is generally recommended for patients with advanced CKD (Table 2).12,16,17 More research is needed for dietary potassium restriction in CKD.18
TABLE 1. Potassium-Rich Foods
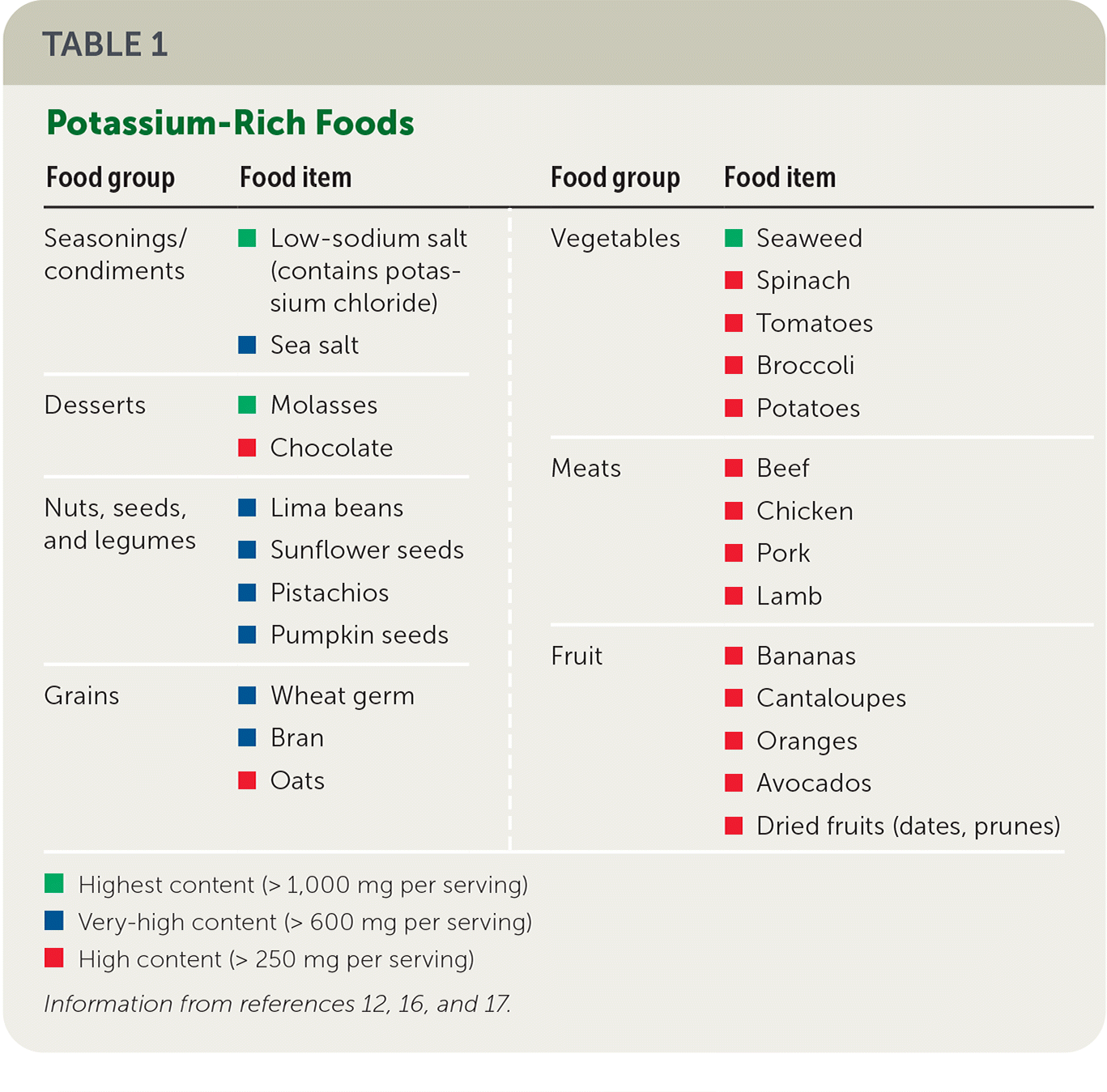
| Food group | Food item | Food group | Food item |
|---|---|---|---|
| Seasonings/condiments Desserts Nuts, seeds, and legumes Grains | Low-sodium salt (contains potassium chloride) Sea salt Molasses Chocolate Lima beans Sunflower seeds Pistachios Pumpkin seeds Wheat germ Bran Oats | Vegetables Meats Fruit | Seaweed Spinach Tomatoes Broccoli Potatoes Beef Chicken Pork Lamb Bananas Cantaloupes Oranges Avocados Dried fruits (dates, prunes) |
Highest content (> 1,000 mg per serving)—green box
Very-high content (> 600 mg per serving)—blue box
High content (> 250 mg per serving)—red box
TABLE 2. Dietary Recommendations Based on Chronic Kidney Disease Stage
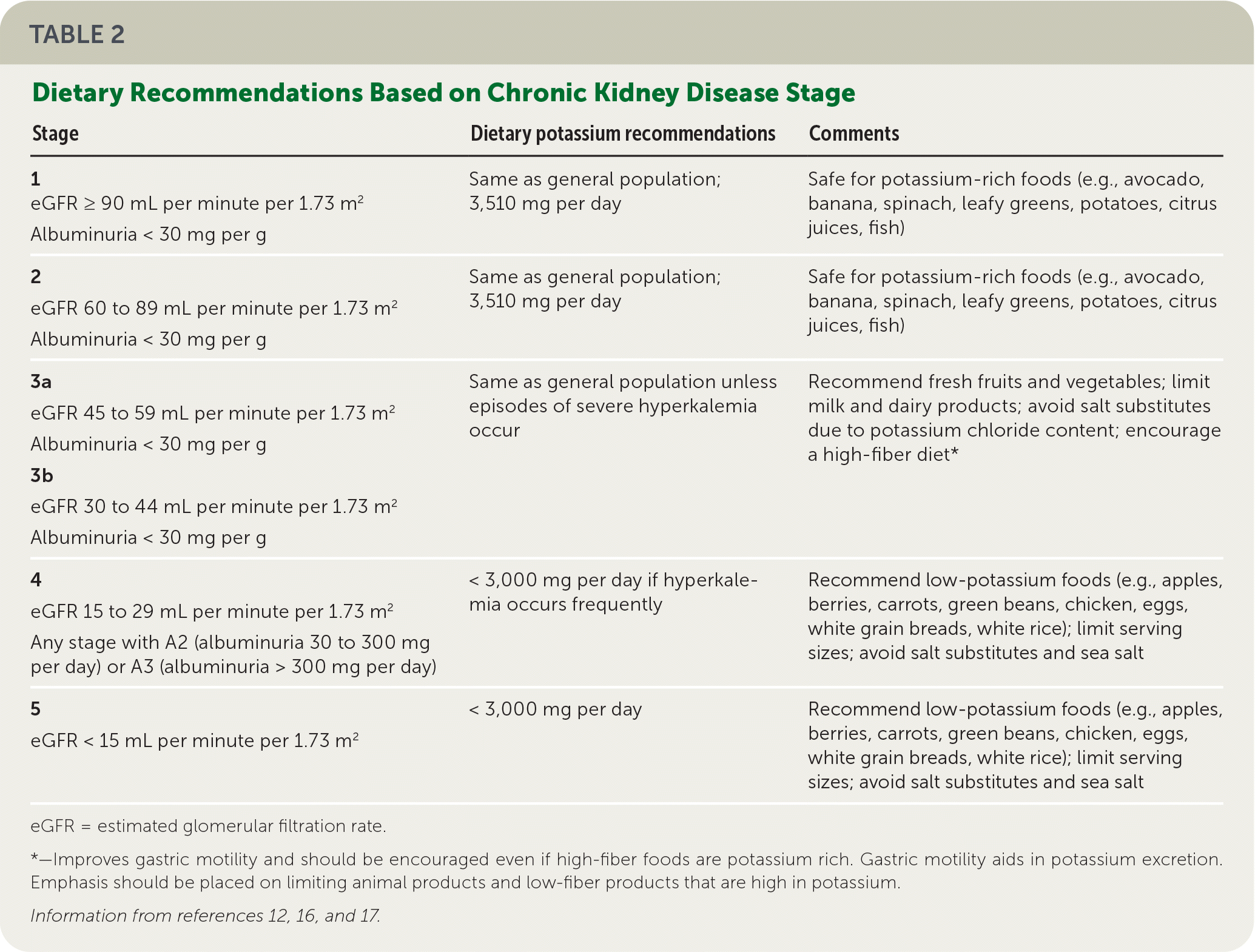
| Stage | Dietary potassium recommendations | Comments |
|---|---|---|
| 1 eGFR ≥ 90 mL per minute per 1.73 m2 Albuminuria < 30 mg per g | Same as general population; 3,510 mg per day | Safe for potassium-rich foods (e.g., avocado, banana, spinach, leafy greens, potatoes, citrus juices, fish) |
| 2 eGFR 60 to 89 mL per minute per 1.73 m2 Albuminuria < 30 mg per g | Same as general population; 3,510 mg per day | Safe for potassium-rich foods (e.g., avocado, banana, spinach, leafy greens, potatoes, citrus juices, fish) |
| 3a eGFR 45 to 59 mL per minute per 1.73 m2 Albuminuria < 30 mg per g 3b eGFR 30 to 44 mL per minute per 1.73 m2 Albuminuria < 30 mg per g | Same as general population unless episodes of severe hyperkalemia occur | Recommend fresh fruits and vegetables; limit milk and dairy products; avoid salt substitutes due to potassium chloride content; encourage a high-fiber diet* |
| 4 eGFR 15 to 29 mL per minute per 1.73 m2 Any stage with A2 (albuminuria 30 to 300 mg per day) or A3 (albuminuria > 300 mg per day) | < 3,000 mg per day if hyperkalemia occurs frequently | Recommend low-potassium foods (e.g., apples, berries, carrots, green beans, chicken, eggs, white grain breads, white rice); limit serving sizes; avoid salt substitutes and sea salt |
| 5 eGFR < 15 mL per minute per 1.73 m2 | < 3,000 mg per day | Recommend low-potassium foods (e.g., apples, berries, carrots, green beans, chicken, eggs, white grain breads, white rice); limit serving sizes; avoid salt substitutes and sea salt |
eGFR = estimated glomerular filtration rate.
*—Improves gastric motility and should be encouraged even if high-fiber foods are potassium rich. Gastric motility aids in potassium excretion. Emphasis should be placed on limiting animal products and low-fiber products that are high in potassium.
Hypokalemia
CAUSES
Hypokalemia is caused by decreased intake, renal losses, gastrointestinal losses, or transcellular shifts (Table 3).19–21 Diuretics are among the most common causes of hypokalemia, especially at higher doses.18,22 Thiazide diuretics are associated with an 11-fold increased risk of hypokalemia and a fivefold increased risk of moderate hypokalemia (less than 3.0 mEq per L).23 Gastrointestinal losses are most often caused by acute or chronic diarrhea. Hypokalemia can occur with low-volume colonoscopy preparation in patients taking diuretics or patients who are hospitalized.24 Female sex and oral laxative use have been associated with preoperative hypokalemia in older patients.25
TABLE 3. Causes of Hypokalemia
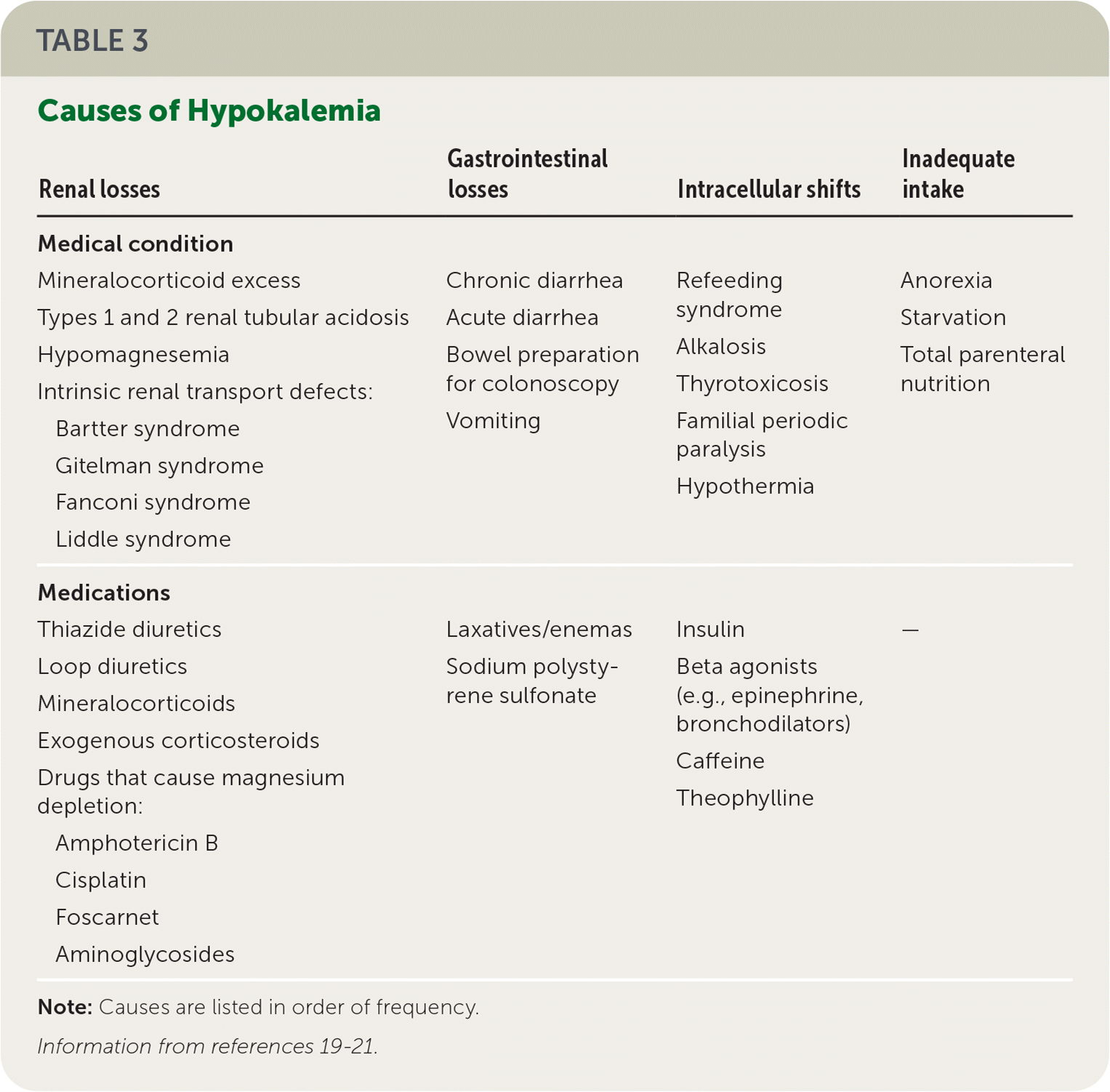
| Renal losses | Gastrointestinal losses | Intracellular shifts | Inadequate intake |
|---|---|---|---|
| Medical condition Mineralocorticoid excess Types 1 and 2 renal tubular acidosis Hypomagnesemia Intrinsic renal transport defects: Bartter syndrome Gitelman syndrome Fanconi syndrome Liddle syndrome | Chronic diarrhea Acute diarrhea Bowel preparation for colonoscopy Vomiting | Refeeding syndrome Alkalosis Thyrotoxicosis Familial periodic paralysis Hypothermia | Anorexia Starvation Total parenteral nutrition |
| Medications Thiazide diuretics Loop diuretics Mineralocorticoids Exogenous corticosteroids Drugs that cause magnesium depletion: Amphotericin B Cisplatin Foscarnet Aminoglycosides | Laxatives/enemas Sodium polystyrene sulfonate | Insulin Beta agonists (e.g., epinephrine, bronchodilators) Caffeine Theophylline | — |
Note: Causes are listed in order of frequency.
HISTORY AND PHYSICAL EXAMINATION
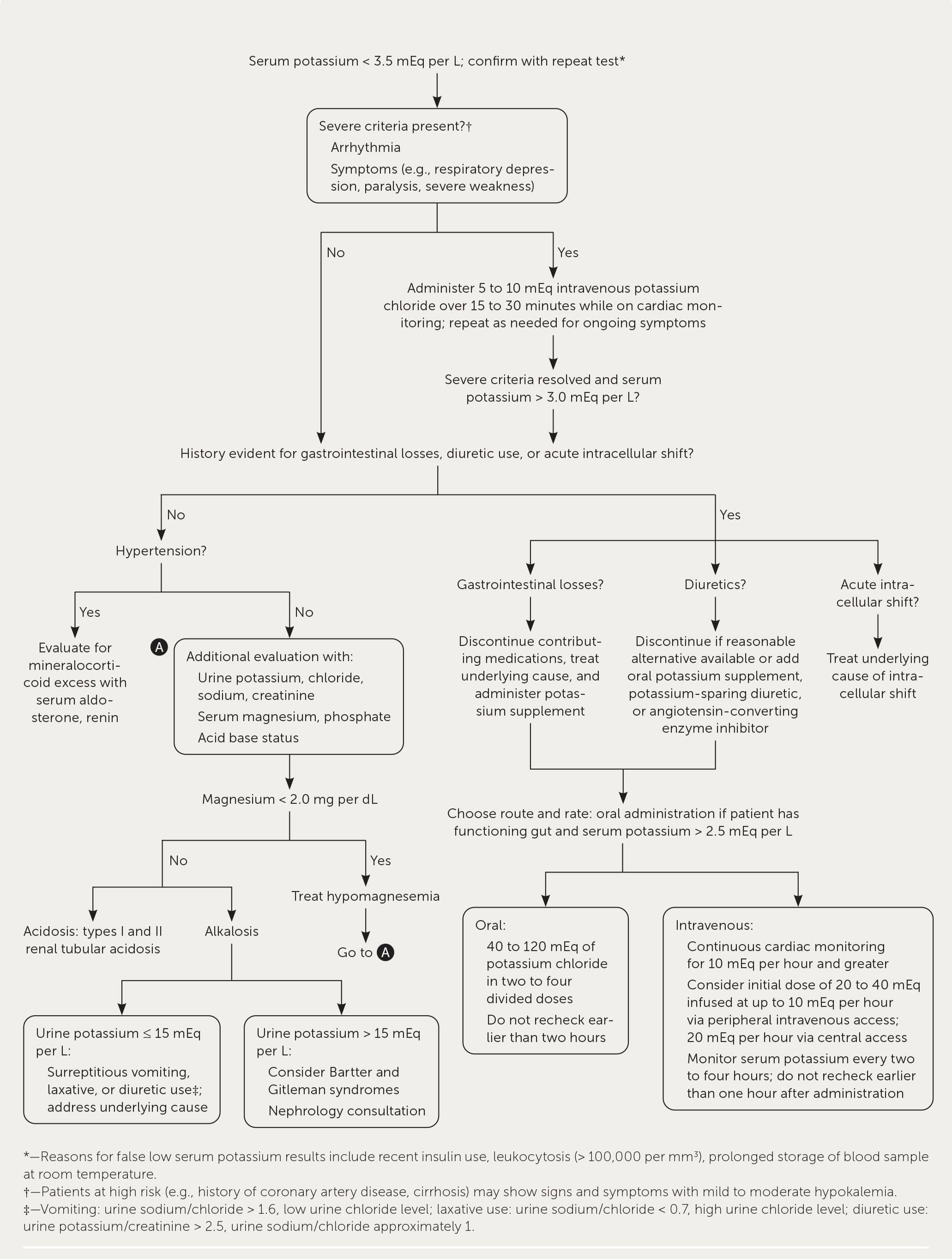
Figure 1 provides an approach to the evaluation and management of hypokalemia.18,26–28 The history should include gastrointestinal losses (e.g., vomiting, diarrhea); medications; high-risk conditions such as heart, kidney, or liver disease; and family history.20 The degree of hypokalemia does not always correlate with the severity of symptoms. Patients with underlying heart disease and hypokalemia are at an elevated risk of developing arrhythmias, especially patients taking digoxin.29 Patients with a known history of cirrhosis are at an increased risk of hepatic encephalopathy because hypokalemia causes the kidney to exchange potassium for hydrogen, which stimulates ammoniagenesis.26 There is a low likelihood of symptoms with serum potassium levels greater than 2.5 mEq per L, but it is more likely with a rapid onset of hypokalemia. Symptoms occur in approximately one-half of patients with severe hypokalemia (2.5 mEq per L or less) and consist of weakness, pain, and cramps.20,30 The physical examination should focus on the cardiac, neurologic, and respiratory systems to evaluate for arrhythmias, severe muscle weakness, paralysis, or respiratory failure.
DIAGNOSTIC TESTING
The diagnosis of hypokalemia should be confirmed with a repeat serum potassium test to exclude pseudohypokalemia.18,26 Pseudohypokalemia results from intracellular potassium uptake when the sample analysis is delayed in the setting of a large number of abnormal white blood cells (greater than 100,000 per mm3) or recent insulin administration.28 If the etiology of hypokalemia cannot be determined based on history and physical examination, a continued evaluation should include serum renin, aldosterone, urine potassium, sodium, chloride, and creatinine. Further testing for thyroid-stimulating hormone or adrenal abnormalities (e.g., morning cortisol, adrenocorticotropic hormone) can be considered if there is diagnostic uncertainty after the initial laboratory evaluation. Renal potassium wasting is indicated by a 24-hour urine potassium level exceeding 15 to 30 mEq per L. A random urine potassium-to-creatinine ratio of greater than 13 may indicate renal potassium wasting; however, recent evidence suggests limited use of spot testing due to intraindividual variability and low specificity.18,31
Electrocardiography (ECG) should be performed when hypokalemia is identified to determine the urgency of treatment; however, ECG changes are not always found in patients with hypokalemia. The earliest change is a decreased T-wave amplitude (eTable A). ECG results may demonstrate progressive changes when potassium levels continue to decline. There are no universally accepted thresholds for specific ECG abnormalities.32–34
eTABLE A Electrocardiography Changes of Potassium Derangements
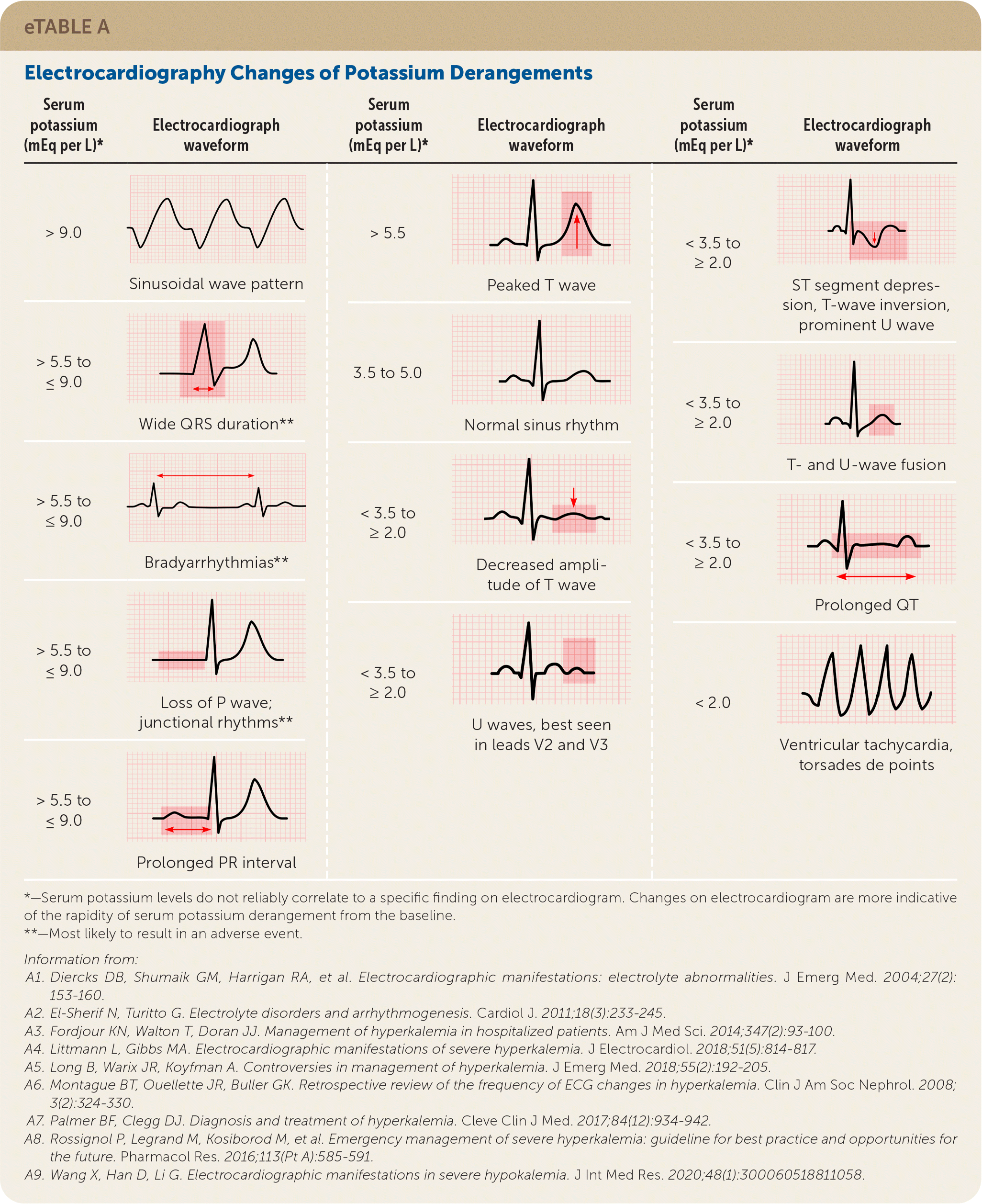
| Serum potassium (mEq per L)* | Electrocardiograph waveform | Serum potassium (mEq per L)* | Electrocardiograph waveform | Serum potassium (mEq per L)* | Electrocardiograph waveform |
|---|---|---|---|---|---|
| > 9.0 | Sinusoidal wave pattern | > 5.5 | Peaked T wave | < 3.5 to ≥ 2.0 | ST segment depression, T-wave inversion, prominent U wave |
| > 5.5 to ≤ 9.0 | Wide QRS duration** | 3.5 to 5.0 | Normal sinus rhythm | < 3.5 to ≥ 2.0 | T- and U-wave fusion |
| > 5.5 to ≤ 9.0 | Bradyarrhythmias** | < 3.5 to ≥ 2.0 | Decreased amplitude of T wave | < 3.5 to ≥ 2.0 | Prolonged QT |
| > 5.5 to ≤ 9.0 | Loss of P wave; junctional rhythms** | < 3.5 to ≥ 2.0 | U waves, best seen in leads V2 and V3 | < 2.0 | Ventricular tachycardia, torsades de points |
| > 5.5 to ≤ 9.0 | Prolonged PR interval |
*—Serum potassium levels do not reliably correlate to a specific finding on electrocardiogram. Changes on electrocardiogram are more indicative of the rapidity of serum potassium derangement from the baseline.
**—Most likely to result in an adverse event.
Information from:
A1. Diercks DB, Shumaik GM, Harrigan RA, et al. Electrocardiographic manifestations: electrolyte abnormalities. J Emerg Med. 2004;27(2):153–160.
A2. El-Sherif N, Turitto G. Electrolyte disorders and arrhythmogenesis. Cardiol J. 2011;18(3):233–245.
A3. Fordjour KN, Walton T, Doran JJ. Management of hyperkalemia in hospitalized patients. Am J Med Sci. 2014;347(2):93–100.
A4. Littmann L, Gibbs MA. Electrocardiographic manifestations of severe hyperkalemia. J Electrocardiol. 2018;51(5):814–817.
A5. Long B, Warix JR, Koyfman A. Controversies in management of hyperkalemia. J Emerg Med. 2018;55(2):192–205.
A6. Montague BT, Ouellette JR, Buller GK. Retrospective review of the frequency of ECG changes in hyperkalemia. Clin J Am Soc Nephrol. 2008;3(2):324–330.
A7. Palmer BF, Clegg DJ. Diagnosis and treatment of hyperkalemia. Cleve Clin J Med. 2017;84(12):934–942.
A8. Rossignol P, Legrand M, Kosiborod M, et al. Emergency management of severe hyperkalemia: guideline for best practice and opportunities for the future. Pharmacol Res. 2016;113(Pt A):585–591.
A9. Wang X, Han D, Li G. Electrocardiographic manifestations in severe hypokalemia. J Int Med Res. 2020;48(1):300060518811058.
Hypokalemia in patients with congestive heart failure or myocardial infarction is associated with an increased likelihood of ventricular tachycardia or fibrillation.10,29 Patients with coronary heart disease or congestive heart failure should maintain a serum potassium level of 4 to 5 mEq per L, because even mild hypokalemia is associated with adverse events, including ventricular arrhythmia and death.5,10
ACUTE TREATMENT
The management of hypokalemia begins with the recognition and treatment of life-threatening sequelae. Patients with severe signs and symptoms (e.g., arrhythmia, paralysis, respiratory failure, severe weakness) should be treated promptly with 5 to 10 mEq of intravenous potassium chloride over 15 to 30 minutes and placed on cardiac monitoring.27,35 This treatment can be repeated until the patient is hemodynamically stable with a resolution of ECG changes and serum potassium level greater than 3 mEq per L; potassium levels should be monitored every two to four hours. Intravenous potassium should be administered in glucose-free fluids. Glucose may worsen symptoms by stimulating insulin release and causing potassium to shift into cells.27 Further intravenous treatment may be administered with 20 to 40 mEq of isotonic fluids and infused up to 10 mEq per hour. The rate can be increased up to 20 mEq per hour if needed but requires cardiac monitoring and central access to avoid pain or venous sclerosis.18
In a patient with a serum potassium level greater than 2.5 mEq per L, no severe signs or symptoms, and a functioning gastrointestinal tract, it is appropriate to start with oral potassium administration instead of intravenous because it has a lower risk of rebound hyperkalemia.27 Potassium chloride is the preferred formulation because it is the most effective for raising serum levels (Table 4).18,21,26,27,35–41 However, potassium phosphate may be appropriate for patients who require phosphate replacement, such as for refeeding syndrome. Potassium bicarbonate is preferred in patients with mild hypokalemia and metabolic acidosis as found in renal tubular acidosis; therefore, consider a nephrology consultation before administration.35 Generally, 20 mEq increases the serum concentration by 0.2 mEq per L. Clinicians should monitor the patient for ongoing losses and intracellular potassium shift during the replacement of the total body deficit.27,35
TABLE 4. Medications for the Treatment of Hypokalemia
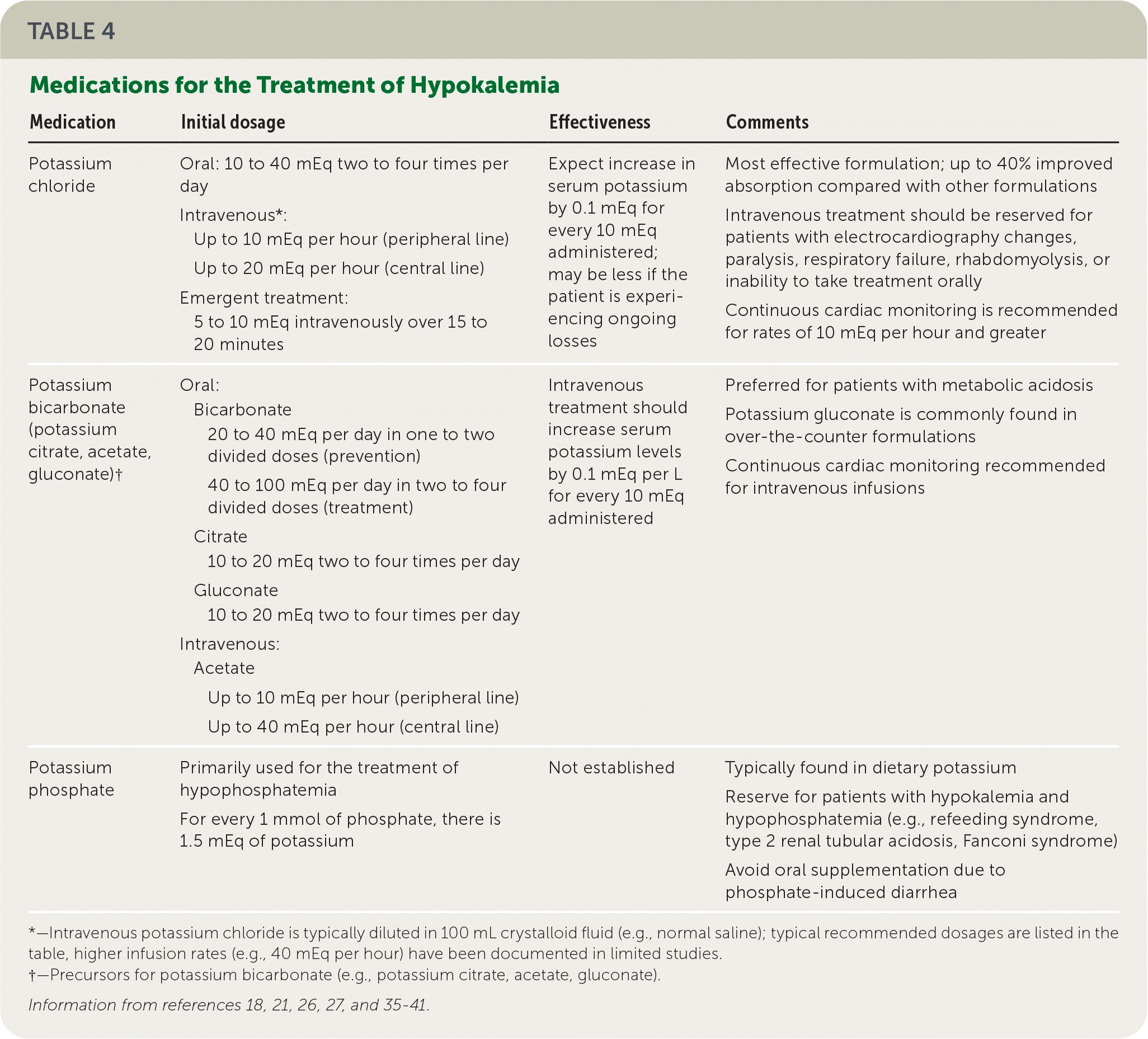
| Medication | Initial dosage | Effectiveness | Comments |
|---|---|---|---|
| Potassium chloride | Oral: 10 to 40 mEq two to four times per day Intravenous*: Up to 10 mEq per hour (peripheral line) Up to 20 mEq per hour (central line) Emergent treatment: 5 to 10 mEq intravenously over 15 to 20 minutes | Expect increase in serum potassium by 0.1 mEq for every 10 mEq administered; may be less if the patient is experiencing ongoing losses | Most effective formulation; up to 40% improved absorption compared with other formulations Intravenous treatment should be reserved for patients with electrocardiography changes, paralysis, respiratory failure, rhabdomyolysis, or inability to take treatment orally Continuous cardiac monitoring is recommended for rates of 10 mEq per hour and greater |
| Potassium bicarbonate (potassium citrate, acetate, gluconate)† | Oral: Bicarbonate 20 to 40 mEq per day in one to two divided doses (prevention) 40 to 100 mEq per day in two to four divided doses (treatment) Citrate 10 to 20 mEq two to four times per day Gluconate 10 to 20 mEq two to four times per day Intravenous: Acetate Up to 10 mEq per hour (peripheral line) Up to 40 mEq per hour (central line) | Intravenous treatment should increase serum potassium levels by 0.1 mEq per L for every 10 mEq administered | Preferred for patients with metabolic acidosis Potassium gluconate is commonly found in over-the-counter formulations Continuous cardiac monitoring recommended for intravenous infusions |
| Potassium phosphate | Primarily used for the treatment of hypophosphatemia For every 1 mmol of phosphate, there is 1.5 mEq of potassium | Not established | Typically found in dietary potassium Reserve for patients with hypokalemia and hypophosphatemia (e.g., refeeding syndrome, type 2 renal tubular acidosis, Fanconi syndrome) Avoid oral supplementation due to phosphate-induced diarrhea |
*—Intravenous potassium chloride is typically diluted in 100 mL crystalloid fluid (e.g., normal saline); typical recommended dosages are listed in the table, higher infusion rates (e.g., 40 mEq per hour) have been documented in limited studies.
†—Precursors for potassium bicarbonate (e.g., potassium citrate, acetate, gluconate).
LONG-TERM TREATMENT
The underlying etiology should be identified and addressed for all patients. For patients taking diuretics, physicians should determine if the patient can discontinue the medication. If that is not an option, the patient should adhere to a low-salt diet. Physicians can add a potassium-sparing diuretic, an angiotensin-converting enzyme (ACE) inhibitor, an angiotensin receptor blocker (ARB), or a beta blocker.18,27,35 Increasing dietary potassium is inadequate because most potassium in foods is coupled with phosphate and is not effective in replacing potassium losses associated with chloride loss, such as in diuretic use, vomiting, or nasogastric suction.20,27 Oral supplementation with potassium chloride (50 to 75 mEq per day) has been shown to increase the serum potassium by approximately 0.14 mEq per L, and this effect is increased with the addition of an ACE inhibitor or an ARB.42
Hyperkalemia
CAUSES
Hyperkalemia results from impaired urinary potassium excretion (e.g., CKD, renin-angiotensin-aldosterone system inhibitors), transcellular shifts, or increased potassium intake such as salt substitutes (Table 5).3,7,43–46 Patients with cardiovascular disease, CKD, and diabetes mellitus are at an increased risk of hyperkalemia and its sequelae.3,7,43–46 Pseudohyperkalemia should be excluded if the serum potassium level does not fit the clinical presentation. A repeat serum potassium test should be performed using a sample collected without a tourniquet and with an unclenched fist.18,28,47
TABLE 5. Causes of Hyperkalemia
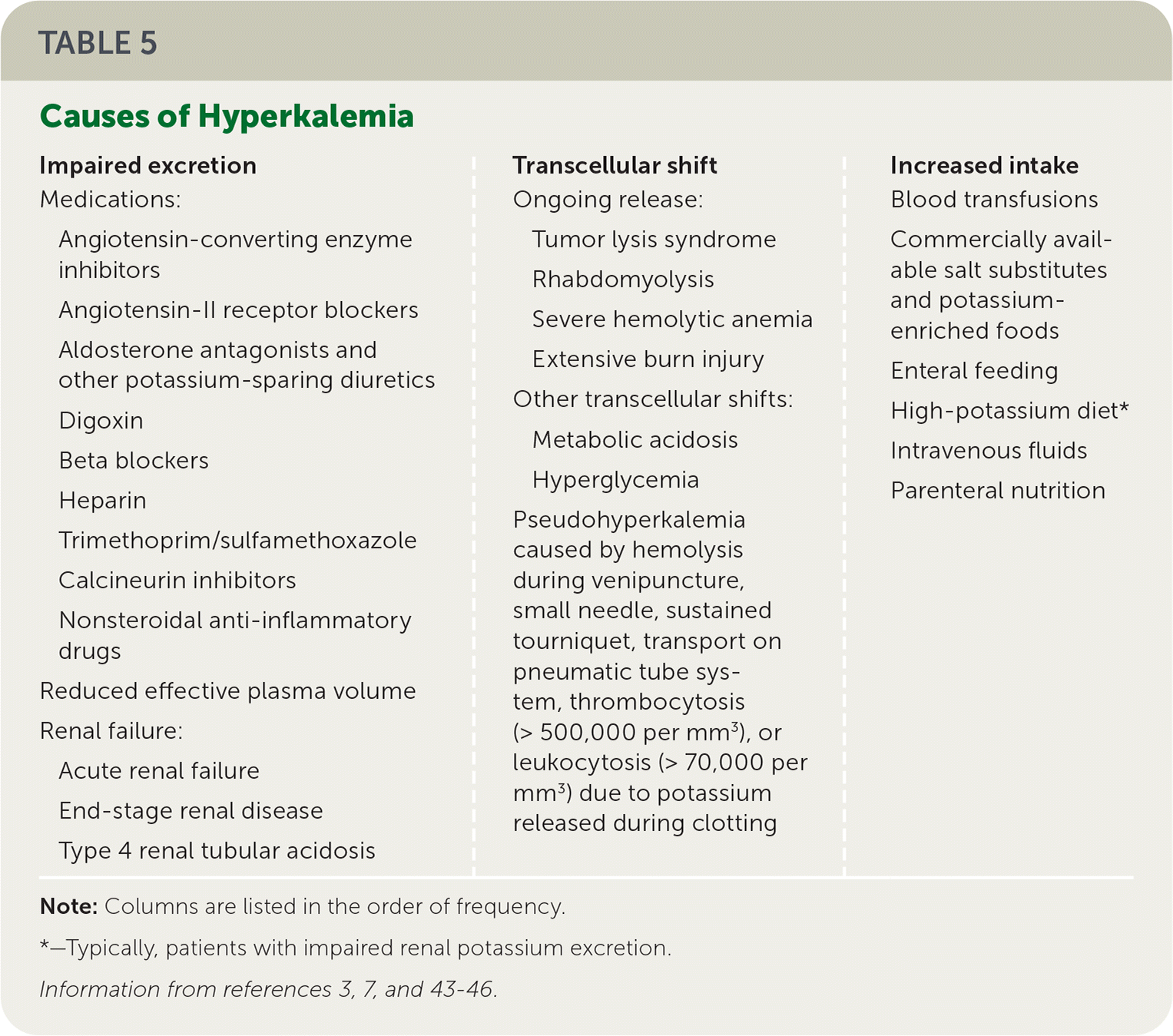
| Impaired excretion Medications: Angiotensin-converting enzyme inhibitors Angiotensin-II receptor blockers Aldosterone antagonists and other potassium-sparing diuretics Digoxin Beta blockers Heparin Trimethoprim/sulfamethoxazole Calcineurin inhibitors Nonsteroidal anti-inflammatory drugs Reduced effective plasma volume Renal failure: Acute renal failure End-stage renal disease Type 4 renal tubular acidosis | Transcellular shift Ongoing release: Tumor lysis syndrome Rhabdomyolysis Severe hemolytic anemia Extensive burn injury Other transcellular shifts: Metabolic acidosis Hyperglycemia Pseudohyperkalemia caused by hemolysis during venipuncture, small needle, sustained tourniquet, transport on pneumatic tube system, thrombocytosis (> 500,000 per mm3), or leukocytosis (> 70,000 per mm3) due to potassium released during clotting | Increased intake Blood transfusions Commercially available salt substitutes and potassium-enriched foods Enteral feeding High-potassium diet* Intravenous fluids Parenteral nutrition |
Note: Columns are listed in the order of frequency.
*—Typically, patients with impaired renal potassium excretion.
Acute or chronic renal dysfunction impairs potassium excretion. Medications such as renin-angiotensin-aldosterone system inhibitors (e.g., ACE inhibitors, ARBs), nonsteroidal anti-inflammatory drugs, and potassium-sparing diuretics impair the excretion of potassium from the body.18,47,48 Selective cyclooxygenase-2 inhibitors are associated with a greater risk of hyperkalemia than nonselective nonsteroidal anti-inflammatory drugs.49 Transcellular shifts can occur from cell breakdown in disease processes such as tumor lysis syndrome, rhabdomyolysis, severe hemolytic anemia, extensive burn injury, acidosis, or decreased insulin. Hyperkalemia attributed to increased dietary intake alone is rare if renal function is normal.47 Salt substitutes, which often contain a mixture of sodium and potassium chloride, may contribute to hyperkalemia.18,43 Other causes include enteral feeding, parenteral nutrition, intravenous fluids, and blood transfusions.48,50
HISTORY AND PHYSICAL EXAMINATION
The severity of hyperkalemia and the need for rapid correction should be determined. Evaluation should focus on a review of medical conditions and medication reconciliation with attention to medications that interfere with the renin-angiotensin-aldosterone system.18,47,48 Physical examinations should evaluate volume status because hypovolemia can lead to hyperkalemia. Signs and symptoms of severe hyperkalemia include muscle weakness, paralysis, and arrhythmias.18,51
DIAGNOSTIC TESTING
ECG should be performed to identify cardiac conduction disturbances in patients with severe hyperkalemia (e.g., greater than 6.5 mEq per L based on expert opinion), rapid serum potassium elevations, symptoms suggesting hyperkalemia, or high-risk conditions (e.g., CKD, heart failure, myocardial infarction).47,52 ECG changes have a low sensitivity for detecting hyperkalemia and do not correlate reliably with serum potassium concentration levels.47,53–57 ECG abnormalities may be a marker of a rapid increase in serum potassium rather than a specific elevated potassium level; therefore, the decision for urgent treatment should not be based on ECG findings alone.46,47,56,58 A prolonged QRS interval (relative risk [RR] = 4.7; 95% CI, 2.0 to 11.6), bradycardia (RR = 12.3; 95% CI, 6.7 to 22.6), and junctional rhythm (RR = 7.5; 95% CI, 5.3 to 11.1) were the most likely to be associated with a serious adverse event such as ventricular fibrillation, cardiopulmonary resuscitation, or death within six hours59 (eTable A). However, the threshold of serum potassium that consistently produces ECG findings predictive of clinical outcomes such as arrhythmia or death has not been established.18,54
ACUTE TREATMENT
Emergent treatment for hyperkalemia is indicated for patients with clinical signs or symptoms of hyperkalemia (e.g., muscle weakness, paralysis), including ECG changes.47,52 Chronic elevations, defined as recurrent elevations in a patient requiring ongoing maintenance therapy, can be lowered over days to weeks.52 Figure 2 outlines an approach to hyperkalemia.28,46–48,52 Acute treatment involves stabilizing the cardiac cell membranes using intravenous calcium, shifting extracellular potassium into cells using intravenous insulin and beta agonists, and eliminating potassium from the body using loop diuretics, potassium binders, and sodium bicarbonate if clinically indicated (Table 6).47,52,60–65 Dialysis is the most effective method of rapidly removing potassium and should be considered early in patients with end-stage renal disease, severe renal impairment, or ongoing potassium release.47
TABLE 6. Medications for the Treatment of Hyperkalemia
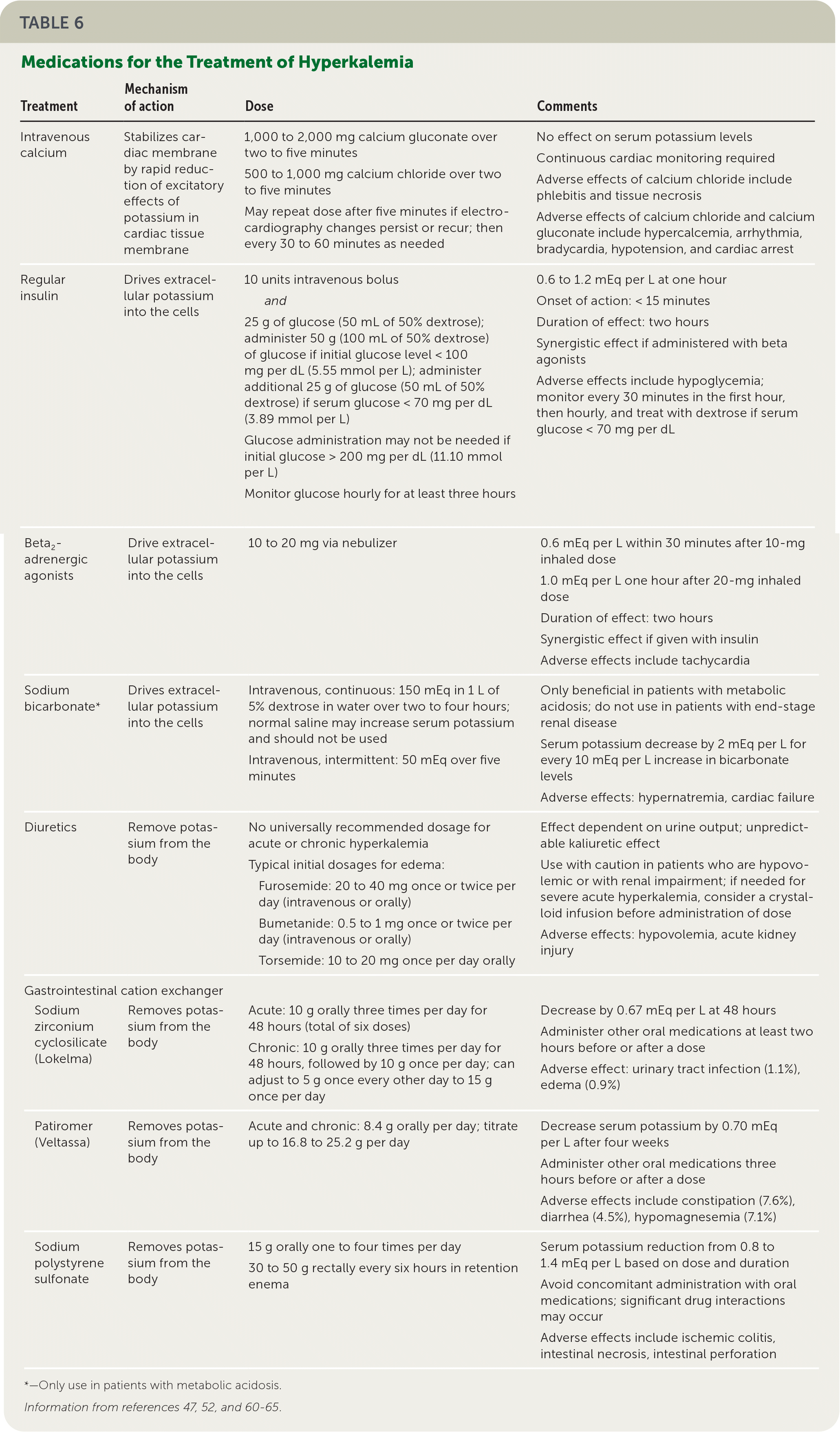
| Treatment | Mechanism of action | Dose | Comments |
|---|---|---|---|
| Intravenous calcium | Stabilizes cardiac membrane by rapid reduction of excitatory effects of potassium in cardiac tissue membrane | 1,000 to 2,000 mg calcium gluconate over two to five minutes 500 to 1,000 mg calcium chloride over two to five minutes May repeat dose after five minutes if electrocardiography changes persist or recur; then every 30 to 60 minutes as needed | No effect on serum potassium levels Continuous cardiac monitoring required Adverse effects of calcium chloride include phlebitis and tissue necrosis Adverse effects of calcium chloride and calcium gluconate include hypercalcemia, arrhythmia, bradycardia, hypotension, and cardiac arrest |
| Regular insulin | Drives extracellular potassium into the cells | 10 units intravenous bolus and 25 g of glucose (50 mL of 50% dextrose); administer 50 g (100 mL of 50% dextrose) of glucose if initial glucose level < 100 mg per dL (5.55 mmol per L); administer additional 25 g of glucose (50 mL of 50% dextrose) if serum glucose < 70 mg per dL (3.89 mmol per L) Glucose administration may not be needed if initial glucose > 200 mg per dL (11.10 mmol per L) Monitor glucose hourly for at least three hours | 0.6 to 1.2 mEq per L at one hour Onset of action: < 15 minutes Duration of effect: two hours Synergistic effect if administered with beta agonists Adverse effects include hypoglycemia; monitor every 30 minutes in the first hour, then hourly, and treat with dextrose if serum glucose < 70 mg per dL |
| Beta2- adrenergic agonists | Drive extracellular potassium into the cells | 10 to 20 mg via nebulizer | 0.6 mEq per L within 30 minutes after 10-mg inhaled dose 1.0 mEq per L one hour after 20-mg inhaled dose Duration of effect: two hours Synergistic effect if given with insulin Adverse effects include tachycardia |
| Sodium bicarbonate* | Drives extracellular potassium into the cells | Intravenous, continuous: 150 mEq in 1 L of 5% dextrose in water over two to four hours; normal saline may increase serum potassium and should not be used Intravenous, intermittent: 50 mEq over five minutes | Only beneficial in patients with metabolic acidosis; do not use in patients with end-stage renal disease Serum potassium decrease by 2 mEq per L for every 10 mEq per L increase in bicarbonate levels Adverse effects: hypernatremia, cardiac failure |
| Diuretics | Remove potassium from the body | No universally recommended dosage for acute or chronic hyperkalemia Typical initial dosages for edema: Furosemide: 20 to 40 mg once or twice per day (intravenous or orally) Bumetanide: 0.5 to 1 mg once or twice per day (intravenous or orally) Torsemide: 10 to 20 mg once per day orally | Effect dependent on urine output; unpredictable kaliuretic effect Use with caution in patients who are hypovolemic or with renal impairment; if needed for severe acute hyperkalemia, consider a crystalloid infusion before administration of dose Adverse effects: hypovolemia, acute kidney injury |
| Gastrointestinal cation exchanger | |||
| Sodium zirconium cyclosilicate (Lokelma) | Removes potassium from the body | Acute: 10 g orally three times per day for 48 hours (total of six doses) Chronic: 10 g orally three times per day for 48 hours, followed by 10 g once per day; can adjust to 5 g once every other day to 15 g once per day | Decrease by 0.67 mEq per L at 48 hours Administer other oral medications at least two hours before or after a dose Adverse effect: urinary tract infection (1.1%), edema (0.9%) |
| Patiromer (Veltassa) | Removes potassium from the body | Acute and chronic: 8.4 g orally per day; titrate up to 16.8 to 25.2 g per day | Decrease serum potassium by 0.70 mEq per L after four weeks Administer other oral medications three hours before or after a dose Adverse effects include constipation (7.6%), diarrhea (4.5%), hypomagnesemia (7.1%) |
| Sodium polystyrene sulfonate | Removes potassium from the body | 15 g orally one to four times per day 30 to 50 g rectally every six hours in retention enema | Serum potassium reduction from 0.8 to 1.4 mEq per L based on dose and duration Avoid concomitant administration with oral medications; significant drug interactions may occur Adverse effects include ischemic colitis, intestinal necrosis, intestinal perforation |
*—Only use in patients with metabolic acidosis.
LONG-TERM TREATMENT
Several potassium binders are available for chronic hyperkalemia. Sodium polystyrene sulfonate has been associated with hospitalizations due to serious adverse gastrointestinal events such as intestinal ischemia and thrombosis (HR = 1.9; 95% CI, 1.1 to 3.4).66,67 Patiromer (Veltassa) has been shown to reduce serum potassium by –0.70 mEq per L (95% CI, –0.48 to –0.91 mEq per L) at four weeks. Sodium zirconium cyclosilicate (Lokelma) has been shown to reduce serum potassium by –0.67 mEq per L (95% CI, –0.45 to –0.89 mEq per L) at 48 hours.68 Sodium zirconium cyclosilicate decreases serum potassium faster (−0.17 mEq per L; 95% CI, –0.05 to –0.30 mEq per L) at one hour, although both patiromer and sodium zirconium cyclosilicate may be useful in patients who are acutely hyperkalemic.68–71 These binders are also more expensive than sodium polystyrene sulfonate. The optimal agent has not been identified for patients with CKD.72 In patients with chronic hyperkalemia related to ACE inhibitors or ARB therapy, potassium binders can be used to safely continue renin-angiotensin-aldosterone system blockade for its benefits on CKD, heart failure, and myocardial infarction, but their long-term outcomes are being studied.43,52
This article updates a previous article on this topic by Viera and Wouk.46
Data Sources: PubMed was searched using the terms hyperkalemia, hypokalemia, potassium disorders, potassium disturbance, electrocardiogram, calcium, insulin, beta agonist, sodium polystyrene sulfonate, patiromer, sodium zirconium cyclosilicate, diuresis, dialysis, chronic kidney disease, cardiac arrest, low-potassium diet, dietary potassium, potassium mortality, prevalence, incidence, pseudohyperkalemia, and inpatient. Also searched were the Trip Database, Essential Evidence Plus, New England Journal of Medicine, JAMA Network, and the Cochrane database. Search dates: December 2021, February 2022, March 2022, May 2022, June 2022, and November 2022.
The authors acknowledge David A Klein, MD, MPH, for providing excellent commentary and professional review in the preparation of this manuscript.
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the U.S. Department of Defense, Uniformed Services University of the Health Sciences, or the U.S. Department of the Air Force.