Adnexal and ovarian masses (including cysts) are common and may require surgery. The U.S. Food and Drug Administration has approved the Risk of Ovarian Malignancy Algorithm (ROMA) test to assess the presurgical likelihood of ovarian cancer in an adult with an ovarian mass for whom surgery is planned. This test combines two serologic biomarkers (human epididymis protein 4 and cancer antigen 125) with menopausal status to produce a numerical score that identifies patients as having low or high risk. Two other multimarker serum tests (OVA1 and Overa), each of which checks for five serum markers, have also been approved by the U.S. Food and Drug Administration for the same indication.
The American College of Obstetricians and Gynecologists recommends that the risk of malignancy be evaluated before surgery because outcomes are improved when patients with cancer have surgery performed by a gynecologic oncologist.1

| Test | Indication | Population | Cost* |
|---|---|---|---|
| ROMA | Assesses likelihood of ovarian cancer in patients with planned surgery | Patients older than 18 years with an adnexal mass who have not yet been referred to an oncologist | $260 |
ROMA = Risk of Ovarian Malignancy Algorithm.
*—Reimbursement rate according to the 2023 Centers for Medicare and Medicaid Services clinical laboratory fee schedule.6
Accuracy
The ROMA test’s performance has been evaluated only in specialty referral populations with a higher risk of ovarian malignancy and as a stand-alone test rather than in conjunction with clinical assessment, as the test was intended to be used.
The diagnostic accuracy of the ROMA compared with human epididymis protein 4 or cancer antigen 125 testing alone was evaluated in a meta-analysis of 32 studies (Table 1).2 Studies included women with ovarian cancer or benign gynecologic disease requiring surgery, with tissue pathology as the reference standard. Blood samples were obtained before treatment of malignancy, if diagnosed. Performance characteristics were similar between premenopausal and postmenopausal patients. Cutoff values for the ROMA test were variable across studies, ranging from a score of 0.74 to 1.31 for premenopausal patients and 1.44 to 2.77 for postmenopausal patients.2 According to the manufacturer, a ROMA score of 1.31 or greater (premenopause) or 2.77 or greater (postmenopause) reflects a high likelihood of malignancy.3
TABLE 1. Pooled Performance Characteristics of Tests to Predict Ovarian Cancer Risk
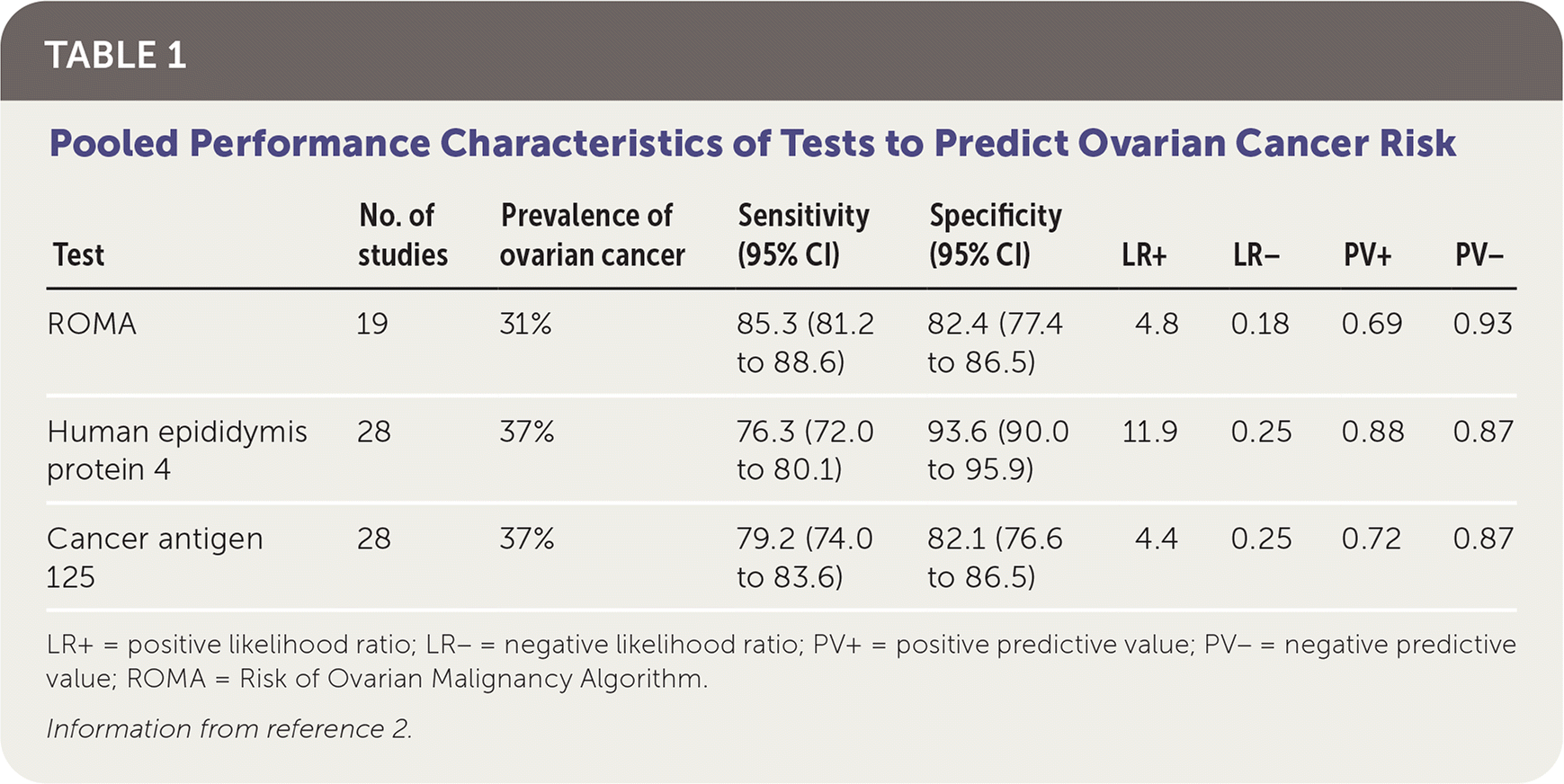
| Test | No. of studies | Prevalence of ovarian cancer | Sensitivity (95% CI) | Specificity (95% CI) | LR+ | LR− | PV+ | PV− |
|---|---|---|---|---|---|---|---|---|
| ROMA | 19 | 31% | 85.3 (81.2 to 88.6) | 82.4 (77.4 to 86.5) | 4.8 | 0.18 | 0.69 | 0.93 |
| Human epididymis protein 4 | 28 | 37% | 76.3 (72.0 to 80.1) | 93.6 (90.0 to 95.9) | 11.9 | 0.25 | 0.88 | 0.87 |
| Cancer antigen 125 | 28 | 37% | 79.2 (74.0 to 83.6) | 82.1 (76.6 to 86.5) | 4.4 | 0.25 | 0.72 | 0.87 |
LR+ = positive likelihood ratio; LR− = negative likelihood ratio; PV+ = positive predictive value; PV− = negative predictive value; ROMA = Risk of Ovarian Malignancy Algorithm.
Information from reference 2.
Benefit
Based on expert opinion and consensus, the American College of Obstetricians and Gynecologists recommends the integration of cancer antigen 125 or serum biomarker panels such as the ROMA test into clinical decision-making to determine the need for consultation with a gynecologic oncologist.1 Limited evidence has shown that patients with ovarian cancer have better outcomes, including living a median of five to eight months longer, when treated by a gynecologic oncologist.4
Harms
The ROMA test has not been well validated in a low-risk, primary care population and should not be used to determine whether a patient should proceed with surgery. The positive predictive value of this test will be poor in a population with a very low pretest probability, which could increase unnecessary testing and surgery.
The ROMA test also does not consider additional risk factors that should raise suspicion for ovarian cancer such as a personal or family history of the disease. A negative test result could inappropriately reassure a high-risk patient if not combined with a clinical assessment of risk, potentially delaying an ovarian cancer diagnosis. The ROMA test should not be used to screen for ovarian cancer in asymptomatic patients without an adnexal mass.
Cost
Citing a lack of reliable evidence demonstrating improved quality of life or decreased mortality in patients with ovarian cancer, the Centers for Medicare and Medicaid Services does not cover the cost of the three available multimarker tests.5 The ROMA test is variably covered by commercial insurance and costs $260. The similar biomarker tests OVA1 and Overa cost $900.6
Bottom Line
Family physicians encounter patients with adnexal masses who require surgery, some of whom may have ovarian cancer. The ROMA test can assess the presurgical likelihood of malignancy in patients who already have a planned surgery. Guidelines support referral of patients at high risk of malignancy to a gynecologic oncologist. The ROMA test should not be used for screening because the ability to draw conclusions about the test’s diagnostic performance in the primary care setting is limited.

